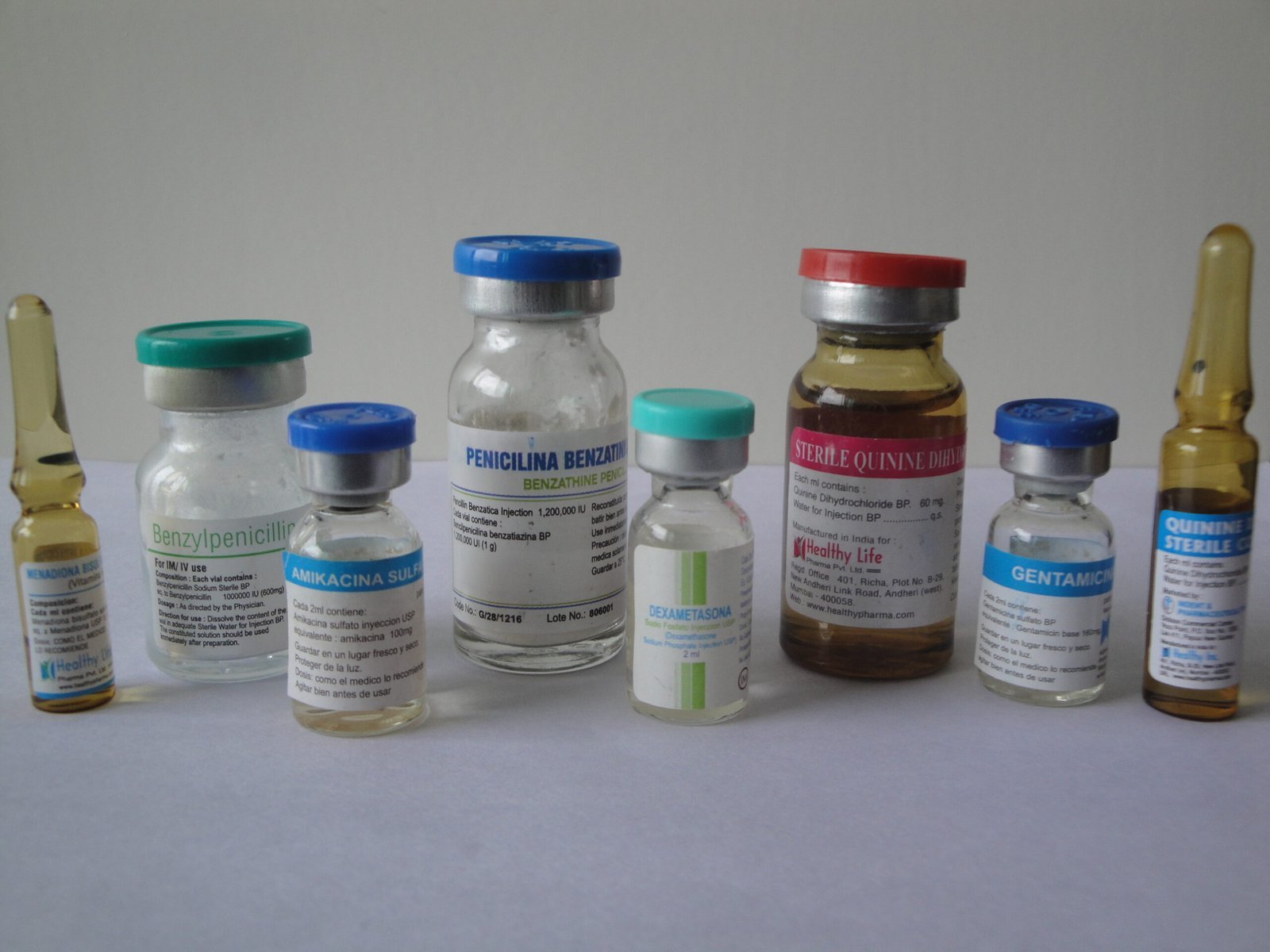
Product List
Showing 1–500 of 1289 results
Acarbose Tablet
FreeAcarbose Tablet 25 mg
Each Film coated tablet contains :
Acarbose BP 25 mgAcarbose Tablets50 mg
Each Filmcoated tablet contains :
Acarbose 50 mgUsage :- control high blood sugar in people with type 2 diabetes
Category :- Anti Diabetic Drugs
Therapeutic category :- Anti Diabetic Drugs
Acebrophylline (SR) & Montelukast Tablets
FreeProduct Composition & Strength
We supply this product as a Sustained-Release, Film-Coated Tablet, engineered to provide synchronized therapeutic levels with convenient once-daily dosing.
Active Ingredient Strength Primary Clinical Function Acebrophylline (SR) USP/BP 200 mg The Airway Opener: Provides 24-hour bronchodilation and thins thick mucus. Montelukast (as Sodium) USP/BP 10 mg The Inflammation Blocker: Prevents asthma triggers and reduces lung swelling. Excipients Hydrophilic Matrix Controlled-Release System: Engineered to provide a smooth plasma curve, minimizing heart palpitations. Acebrophylline (SR) Montelukast & Fexofenadine Tablets
FreeProduct Composition & Strength
We supply this product as a Sustained-Release, Film-Coated Tablet, engineered to provide a steady-state therapeutic concentration over a full 24-hour cycle.
Active Ingredient Strength Primary Clinical Function Acebrophylline (SR) USP/BP 200 mg The Broncho-Mucolytic: Relaxes airways and thins mucus for 24-hour breathing comfort. Montelukast (as Sodium) USP/BP 10 mg The LTRA: Prevents chronic inflammation and asthma triggers in the lungs. Fexofenadine HCl USP/BP 120 mg The Antihistamine: Provides non-sedating relief from upper respiratory allergic symptoms. Acebrophylline Tablets
FreeProduct Composition & Strength
We supply this product as both an Immediate-Release Tablet for acute management and a Sustained-Release (SR) Tablet for 24-hour protection.
Active Ingredient Strength Primary Clinical Function Acebrophylline USP/BP 100 mg The Standard Relief Unit: Taken twice daily to manage active symptoms of bronchitis and asthma. Acebrophylline USP/BP 200 mg SR The 24-Hour Anchor: A sustained-release format that provides consistent airway stability with once-daily dosing. Excipients Hydrophilic Matrix Controlled-Release System: Engineered to provide a smooth plasma concentration curve, minimizing the risk of “Theophylline-like” side effects. Aceclofenac ,Paracetamol & Trypsin-Chymotrypsin 150000AU Tablets
FreeProduct Composition & Strength
We supply this product as a Film-Coated, Enteric-Shielded Tablet to ensure the enzymes are protected from stomach acid and absorbed in the intestine.
Active Ingredient Strength Primary Clinical Function Aceclofenac USP/BP 100 mg The Anti-Inflammatory: Reduces joint pain and physical swelling of the tissues. Paracetamol USP/BP 325 mg The Analgesic: Provides rapid relief from acute pain and manages fever. Trypsin-Chymotrypsin 150,000 AU The Enzyme Payload: Resolves inflammation and clears edema to accelerate tissue repair. Aceclofenac , Paracetamol and Serratiopeptidase Tablets
FreeProduct Composition & Strength
We supply this product as a Film-Coated, Enteric-Shielded Tablet to ensure the enzyme remains active through the stomach acid.
Active Ingredient Strength Primary Clinical Function Aceclofenac USP/BP 100 mg The Anti-Inflammatory: Reduces joint and tissue inflammation and stiffness. Paracetamol USP/BP 325 mg The Analgesic: Provides rapid relief from acute pain and manages fever. Serratiopeptidase 15 mg The Enzyme Payload: Resolves inflammation and clears edema to accelerate tissue repair. Aceclofenac , Paracetamol and Tizanidine Tablets
FreeProduct Composition & Strength
We supply this product as a Film-Coated Tablet, engineered for synchronized absorption to ensure the muscle relaxant and pain reliever reach therapeutic levels at the same time.
Active Ingredient Strength Primary Clinical Function Aceclofenac USP/BP 100 mg The Anti-Inflammatory: Reduces joint and tissue inflammation and stiffness. Paracetamol USP/BP 325 mg The Analgesic: Provides rapid relief from acute pain and manages fever. Tizanidine (as HCl) USP/BP 2 mg The Muscle Relaxant: Relieves painful muscle spasms and improves physical mobility. Aceclofenac & Paracetamol Tablets
FreeProduct Composition & Strength
We supply this product as a Film-Coated Tablet, engineered for rapid disintegration and synchronized absorption.
Active Ingredient Strength Primary Clinical Function Aceclofenac USP/BP 100 mg The Anti-Inflammatory: Targets tissue inflammation and reduces joint stiffness. Paracetamol USP/BP 325 mg / 500 mg The Analgesic: Provides rapid relief from acute pain and manages fever. Excipients Proprietary Matrix Rapid-Release System: Engineered to ensure both APIs reach therapeutic levels within 30–60 minutes. Aceclofenac & Thiocolchicoside Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet, packed in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute stability of the semi-synthetic Thiocolchicoside component.
Active Ingredient Strength Primary Clinical Use Aceclofenac BP/Ph.Eur.
Thiocolchicoside INN/Ph.Eur.100 mg
4 mgStandard Orthopedic Dose: Twice-daily therapy for moderate-to-severe acute back pain and torticollis. Aceclofenac BP/Ph.Eur.
Thiocolchicoside INN/Ph.Eur.100 mg
8 mgSevere Trauma Dose: Maximum therapy for severe, immobilizing orthopedic spasms (strictly twice daily). Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Magnesium Stearate Diluent / Superdisintegrant (Engineered for immediate gastric dissolution and rapid onset of relief) Aceclofenac Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Amber Glass Ampoules. We utilize advanced solubilizing agents to ensure the drug remains stable in liquid form.
Active Ingredient Strength (Standard) Therapeutic Role Aceclofenac BP/IP 150 mg / ml Analgesic / Anti-inflammatory Excipients Q.S. Benzyl Alcohol / Propylene Glycol Preservative / Co-solvent *Pack Sizes: Tray of 5 Ampoules (1ml each) or 10 Ampoules.
Aceclofenac Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets (Standard) or Sustained Release (SR) Tablets.
Active Ingredient Strength Release Type Role Aceclofenac IP/BP/USP 100 mg Immediate Release Standard Adult Dose Aceclofenac IP/BP/USP 200 mg Sustained Release (SR) Once Daily Dosing Excipients Q.S. — Disintegrant / Binder Aceclofenac, Paracetamol & Chlorzoxazone Tablets
FreeProduct Composition & Strength
We supply this product as a Film-Coated Tablet, engineered for synchronized absorption to ensure the muscle relaxant and pain reliever reach therapeutic levels at the same time.
Active Ingredient Strength Primary Clinical Function Aceclofenac USP/BP 100 mg The Anti-Inflammatory: Reduces joint and tissue inflammation and stiffness. Paracetamol USP/BP 325 mg The Analgesic: Provides rapid relief from acute pain and manages fever. Chlorzoxazone USP/BP 250 mg / 500 mg The Muscle Relaxant: Relieves painful muscle spasms and improves physical mobility. Acenocoumarol Tablets
FreeProduct Composition & Strength
We supply this product in multiple strengths to allow for the precise, micro-gram level titration required for safe anticoagulation.
Active Ingredient Strength Primary Clinical Function Acenocoumarol USP/BP/Ph.Eur. 1 mg / 2 mg The Titration Unit: Used for fine-tuning the maintenance dose to keep the INR within the target range (usually 2.0–3.0). Acenocoumarol USP/BP/Ph.Eur. 3 mg / 4 mg The Loading/Anchor Dose: Used for initial therapy or for patients requiring higher systemic anticoagulation. Excipients Proprietary Matrix Bio-Stable System: Engineered to ensure consistent oral absorption and predictable pharmacokinetics. Acetaminophen & Tramadol Hydrochloride Tablets
FreeProduct Composition & Strength
We supply this product in the globally standardized “325/37.5” ratio, optimized for maximum efficacy with minimal opioid-related side effects.
Active Ingredient Strength Primary Clinical Function Acetaminophen (Paracetamol) USP/BP 325 mg The Rapid Starter: Provides quick onset of analgesia and antipyretic (fever-reducing) effects. Tramadol Hydrochloride USP/BP 37.5 mg The Sustained Blocker: Provides moderate-to-severe pain relief via opioid and neurotransmitter pathways. Excipients Proprietary Matrix Bio-Synchronized System: Engineered to ensure both APIs reach therapeutic plasma levels simultaneously for a synergistic effect. Acetazolamide Tablet
FreeAcetazolamide Tablets IP 250 mg
Each uncoated tablet Contains : Acetazolamide IP 250 mg
Excipients q.sAcetazolamide Tablets BP 250 mg
Each uncoated tablet Contains : Acetazolamide BP 250 mg
Excipients q.sUsage :- Prevent and reduce the symptoms of altitude sickness
Category :- Diuretic drugs
Therapeutic category :- Diuretic drugs
Acetyl Salicylic Acid Tablets
FreeProduct Composition & Strength
We supply this product in two distinct clinical ranges: the “Cardio-Shield” range and the “Analgesic-Power” range.
Active Ingredient Strength Primary Clinical Function Acetylsalicylic Acid USP/BP 75 mg / 100 mg The Cardio-Shield: Once-daily “Baby Aspirin” for long-term prevention of heart attack and stroke. Acetylsalicylic Acid USP/BP 300 mg The Emergency Loading Dose: Used immediately during a suspected heart attack to break up clots. Acetylsalicylic Acid USP/BP 500 mg The Analgesic Anchor: For the relief of headache, toothache, and musculoskeletal pain. Acitretin Tablets
FreeProduct Composition & Strength
We supply this product in standardized strengths to allow for precise clinical titration, packed in high-barrier Alu-Alu blister strips.
Active Ingredient Strength Primary Clinical Function Acitretin USP/Ph.Eur. 10 mg The Titration Unit: Used to start therapy or for maintenance in patients sensitive to retinoids. Acitretin USP/Ph.Eur. 25 mg The Therapeutic Anchor: The standard dose for achieving rapid clearance of severe skin lesions. Excipients Proprietary Matrix Bio-Stable System: Engineered to ensure consistent oral absorption (approx. 60% bioavailability when taken with food). Activated Charcoal Tablets
FreeProduct Composition & Strength
We supply this product as a High-Density, Compressible Tablet, engineered to disintegrate rapidly in the stomach for maximum surface area exposure.
Active Ingredient Strength Primary Clinical Function Activated Charcoal USP/BP/Ph.Eur. 250 mg The GI Relief Unit: Used primarily for over-the-counter (OTC) management of gas and bloating. Activated Charcoal USP/BP/Ph.Eur. 500 mg The Emergency Payload: High-potency format for clinical detoxification and poisoning management. Excipients Binder/Disintegrant Rapid-Discharge Matrix: Engineered to ensure the charcoal particles disperse immediately upon contact with gastric fluid. Adrenaline Injection I.P.
FreeComposition
Each ml. Contains:
Adrenaline Bitartrate I.P.
Eq. To. Adrenaline…………………….1 mg.
Water For Injec9on I.P………..………..Q.Pcd pharma franchise:-
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Adrenaline Injections
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Amber Glass Ampoules. The formulation includes stabilizers to prevent rapid oxidation.
Active Ingredient Strength (Standard 1:1000) Therapeutic Role Adrenaline Bitartrate BP/USP Eq. to Adrenaline 1 mg / ml Vasopressor / Bronchodilator Excipients Q.S. Sodium Metabisulfite / Sodium Chloride Antioxidant / Isotonicity *Pack Sizes: Tray of 10 Ampoules (1ml each) or Box of 50/100 Ampoules (Hospital Pack).
Adrenochrome Monosemicarbazone Tablet
FreeAdrenochrome Monosemicarbazone compound Tablets
each uncoated tablet contains:
Adronochrome Monosemi Carbazone (In House) (0.33 mg)
Menadione Sodium Bisulphite IP (10 mg)
Sodium Ascorbate IP eq. To Ascorbic Acid IP (100 mg)
Rutin NF (50 mg)
Dibasic Calium Phosphate IP (125 mg)
excipient (In House) (- QS)Afatinib Tablets
FreeProduct Composition & Strength
We supply this product in multiple strengths to allow for precise dose titration based on patient tolerability.
Active Ingredient Strength Primary Clinical Function Afatinib (as Dimaleate) USP/Ph.Eur. 20 mg / 30 mg The Dose-Adjustment Unit: Used for patients requiring a lower dose due to gastrointestinal or skin toxicity. Afatinib (as Dimaleate) USP/Ph.Eur. 40 mg The Standard Therapeutic Anchor: The globally validated starting dose for first-line treatment of EGFR+ NSCLC. Excipients Proprietary Matrix Bio-Stable Coating: Engineered to protect the sensitive dimaleate salt and ensure consistent systemic absorption. Agomelatine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Bioavailability Tablet, packed in high-barrier Alu-Alu blister strips to ensure the stability of the API.
Active Ingredient Strength Primary Clinical Function Agomelatine USP/Ph.Eur. 25 mg The Therapeutic Anchor: The globally validated dose for restoring circadian rhythm and treating major depression. Excipients Proprietary Matrix Bio-Synchronized Release: Engineered to ensure a rapid $T_{max}$ (approx. 1-2 hours) to align with the body’s natural melatonin surge. Albendazole Tablet
FreeADZOLE 200
Albendazole Tablets
Each Uncoated tablet Contains:
Albendazole USP 200 mg
Excipients q.sAlbendazole Tablet
s IP 400 mg
Each Uncoated tablet Contains:
Albendazole IP 400 mg
Colour : Sunset YellowUltiworm
Albendazole Tablets 400 mg
Each Uncoated chewable tablet contains:
Albendazole BP 400 mgZENBEST – 400 / ALBENDAZOLE TABLETS 400 MG Each Uncoated Tablets Contains: Albendazole BP 400 mg Excipients – QS
Albendazole Tablets IP 400 mg
Each Uncoated Chewable Tablet Contains: Albendazole IP 400 mg
Sunset yellow –
Excipients – QS
orange –
Usage :- certain infections caused by worms such as pork tapeworm and dog tapewormCategory :- Anthelmintic drugs
Therapeutic category :- Antiparasitic Anthelmintic,Antiprotozoal,
Alendronate Tablet
FreeAlenfos
Alendronate Tablets 70 mg
Each tablet contains :
Alendronate Sodium
Eq. To Alendronate Sodium anhydrous 70 mgAlfacalcidol Softgel Capsules
FreeProduct Composition & Strength
We supply this product as a Liquid-Filled Soft Gelatin Capsule, packed in highly protective, opaque blister strips to prevent the extreme light-degradation associated with vitamin D analogues.
Active Ingredient Strength Primary Clinical Use Alfacalcidol BP/EP 0.25 mcg (Micrograms) Initial Titration / Pediatric / Maintenance Dosing Alfacalcidol BP/EP 0.5 mcg (Micrograms) Standard Adult Dialysis / Osteoporosis Dosing Alfacalcidol BP/EP 1.0 mcg (Micrograms) Severe Hypocalcemia / High-PTH Suppression Excipients Refined Arachis (Peanut) Oil / BHA Lipid Carrier System / Antioxidant Stabilizer *Pack Sizes: 10×10 Opaque Alu-PVC Blisters or Alu-Alu Blisters. (Opaque packaging is an absolute necessity to maintain microgram API stability).
Alfuzosin & Dutasteride Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Bi-Layer or Capsule-in-Tablet format, packed in high-barrier Alu-Alu blister strips to ensure the stability of the moisture-sensitive Dutasteride.
Active Ingredient Strength Primary Clinical Function Alfuzosin HCl (Extended-Release) USP/BP 10 mg The Rapid Reliever: Provides immediate improvement in urine flow by relaxing prostate muscles. Dutasteride USP/BP 0.5 mg The Long-Term Shaper: Reduces the physical size of the prostate to prevent the need for surgery. Excipients Hydrophilic Matrix Controlled-Release System: Engineered to provide synchronized 24-hour therapeutic coverage with once-daily dosing. Alfuzosin Hydrochloride Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Extended-Release (ER) Tablet, packed in high-barrier Alu-Alu blister strips to ensure API stability.
Active Ingredient Strength Primary Clinical Function Alfuzosin Hydrochloride USP/BP 10 mg The Standard Therapeutic Anchor: The globally validated once-daily dose for moderate-to-severe BPH symptoms. Excipients Extended-Release Matrix Controlled-Delivery System: Engineered to provide synchronized 24-hour therapeutic coverage, eliminating the need for multiple daily doses. Allopurinol Tablet
FreeAlloric – 100 Tablets
Allopurinol Tablets IP 100 mg
Each Uncoated tablet contains :
Allopurinol IP 100 mgAllopurinol Tablets BP 300 mg
Each uncoated tablet contains :
Allopurinol BP 300 mgAllylestrenol Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Bio-Stable Tablet, packed in high-barrier Alu-Alu blister strips to ensure hormonal stability.
Active Ingredient Strength Primary Clinical Function Allylestrenol USP/BP/Ph.Eur. 5 mg The Standard OB-GYN Anchor: The globally validated dose for maintaining the corpus luteum and supporting the early placenta. Excipients Proprietary Matrix Bio-Synchronized System: Engineered to ensure rapid oral absorption and high plasma concentration to provide immediate uterine stability. Alpha Ketoanalogue Tablets
FreeProduct Composition & Strength
We supply this product as a Multi-Mineral, Amino Acid Complex, carefully balanced to meet the Nutritional Requirements of Uremic Patients.
Component (Active) Amount per Tablet Clinical Function Alpha-Keto-Isoleucine / Leucine / Valine / Phenylalanine Calcium Salts The Protein Builders: Converted into essential amino acids using waste nitrogen. Alpha-Hydroxy-Methionine Calcium Salt The Sulfur Source: Provides the essential hydroxy-analog of methionine. L-Lysine / L-Threonine / L-Tryptophan Acetate/Pure Form Essential Amino Acids: Direct supplementation of AA that the body cannot synthesize. L-Histidine / L-Tyrosine Pure Form Conditional Amino Acids: Critical for maintaining nitrogen balance in renal failure. Calcium Content ~50 mg (approx) Phosphate Binder Support: Provides calcium which helps in managing hyperphosphatemia. Alprazolam & Sertraline Hydrochloride Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Film-Coated Tablet, packed in high-barrier Alu-Alu blister strips to ensure the stability of the benzodiazepine component.
Active Ingredient Strength Primary Clinical Function Sertraline (as HCl) USP/BP 25 mg / 50 mg The Antidepressant Anchor: Provides long-term correction of serotonin levels to treat depression and OCD. Alprazolam USP/BP 0.25 mg / 0.5 mg The Anxiolytic Bridge: Provides rapid relief from acute anxiety and prevents panic spikes. Excipients Proprietary Matrix Bio-Synchronized System: Engineered to ensure the rapid release of Alprazolam for immediate relief while Sertraline maintains steady-state plasma levels. Alprostadil Tablets
FreeProduct Composition & Strength
We supply this product in high-purity oral and specialized dispersible formats, primarily for hospital and specialist use.
Active Ingredient Strength Primary Clinical Function Alprostadil USP/BP/Ph.Eur. 0.5 mg The Neonatal Unit: Used in controlled hospital settings for maintaining ductal patency in infants with congenital heart defects. Alprostadil USP/BP/Ph.Eur. 1 mg The Vascular/Urology Anchor: Used for severe peripheral vascular disease or refractory erectile dysfunction. Excipients Proprietary Matrix Bio-Stable System: Engineered to prevent the rapid degradation of the sensitive prostaglandin molecule during transit. Altretamine Capsules
FreeAltretamine capsules are antineoplastic (chemotherapy) medications used primarily for the palliative treatment of advanced ovarian cancer. They are most commonly available in a 50 mg strength.
Aluminium hydroxide Magnesium Trisilicate Tablet
FreeCompound Magnesium Trisilicate Tablets BP
Aluminium Hydroxide & magnesium Trisilicate tablets
Each Uncoated tablet contains :
Magnesium Trisilicate BP 250 mg
Dried Aluminium Hydroxide BP 120 mg
Peppermint FlavourDRIED ALUMINIUM HYDROXIDE GE+MAGNESIUM TRISILICATE TABLETS
Each uncoated chewable tablet contains: Magnesium Trisilicate IP 500 mg
Dried Aluminium Hydroxide Gel IP 250 mg
Colour: Sunset Yellow –
Excipients – QSAluminium Hydroxide & Magnesium Trisilicate Tablet NFI
Each uncoated Chewable Tablet Contains:
Dried Aluminium Hydroxide Gel IP 120 mg
Magnesium Trisilicate IP 250 mg
Peppermint Oil IP 0.003 mlALUMINUM HYDROXIDE GEL + MAGNESIUM TRISILICATE
Each Uncoated chewable tablet contains:
Magnesium Trisilicate IP 500 mg
Dried Aluminium Hydroxide gel IP 250 mgDRIED ALUMINIUM HYDROXIDE GE + MAGNESIUM TRISILICATE TABLETS Each uncoated chewable tablet contains: Magnesium Trisilicate IP 500 mg
Dried Aluminium Hydroxide Gel IP 250 mg
Colour: Sunset Yellow –
Excipients – QS
Usage: – Relieve the symptoms of indigestion, heartburn, or gastro esophageal reflux disorderCategory: – Antacid – Anti Ulcer drugs
Therapeutic category: – Antacid
Ambrisentan Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Potency Tablet, packed in high-barrier Alu-Alu blister strips to ensure API stability.
Active Ingredient Strength Primary Clinical Function Ambrisentan USP/Ph.Eur. 5 mg The Initiation Unit: The standard starting dose for adults to establish tolerance and initial pulmonary dilation. Ambrisentan USP/Ph.Eur. 10 mg The Maximum Maintenance Payload: For patients requiring additional symptomatic improvement after the 5 mg dose. Excipients Proprietary Matrix Bio-Optimized System: Engineered to ensure a consistent $T_{max}$ (peak concentration) of approx. 2 hours for reliable daily protection. Amikacin injection manufacturer
Freecomposition
Amikacin Sulphate 100 mg Injection (2ML VIAL)
Amikacin Sulphate 250 mg Injection (2ML VIAL)
Amikacin Sulphate 500 mg Injection (2ML VIAL)
Pcd pharma franchise:-
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy In
Amikacin Sulphate Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Liquid in Glass Vials or Ampoules. The solution is clear, colorless to pale yellow, and requires no reconstitution (ready-to-use).
ant Active Ingredient Strength (Standard) Therapeutic Role Amikacin Sulphate USP/BP 100 mg / 2 ml Pediatric Dose Amikacin Sulphate USP/BP 250 mg / 2 ml Intermediate Dose Amikacin Sulphate USP/BP 500 mg / 2 ml Adult Standard Dose Excipients Q.S. Sodium Citrate / Sodium Metabisulfite Buffer / Antioxid *Pack Sizes: Tray of 10 Ampoules or Box of 1/10 Vials.
Amikacin Sulphate injection
FreeComposition
Amikacin Sulphate 100 mg Injection
Each 2 ml Contains : Amikacin Sulphate I.P.
Equi. To. Amikacin ……….. 100 mg
Sodium Citrate I.P. ……… 0.5 % W/V
Sodium Metabisulphate I.P. 0.13 %W/V
Methyl Paraben I.P 0.08 % W/V
Propyl Paraben I.P 0.02 % W/V
Water for Injection I.P. Q.SAmikacin Sulphate 250 mg Injection
Each 2 ml Contains : Amikacin Sulphate I.P.
Equi. To. Amikacin ……….. 250 mg
Sodium Citrate I.P. ……… 1.25 % W/V
Sodium Metabisulphate I.P . 0.66 %W/V
Methyl Paraben I.P 0.08 % W/V
Propyl Paraben I.P 0.02 % W/V
Water for Injection I.P. Q.S.Amikacin Sulphate 500 mg Injection
Each 2 ml Contains :Amikacin Sulphate I.P.
Equi. To. Amikacin ……….. 500 mg
Sodium Citrate I.P. ……… 2.5 % W/V
Sodium Metabisulphate I.P.. 0.66 %W/V
Methyl Paraben I.P 0.08 % W/V
Propyl Paraben I.P 0.02 % W/V
Water for Injection I.P. Q.SUsage: –It is used to treat various bacterial infections
Category: – antibiotics injection
Therapeutic category: – antibiotics medicine
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Amikacin veterinary injection
FreeComposition
Amikacin 250 mg VET INJ. 50 ML
Usage: –Here are some general guidelines for the usage of Amikacin in veterinary medicine
Category: – veterinary injection
Therapeutic category: –veterinary medicine
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Amiloride & Hydrochlorothiazide Tablets
FreeProduct Composition & Strength
We supply this product in the globally standardized strength ratio, packed in high-barrier Alu-PVC or Alu-Alu blister strips.
Active Ingredient Strength Primary Clinical Function Amiloride (as HCl) USP/BP 5 mg The Potassium Guard: Prevents the loss of potassium, protecting against heart arrhythmias and muscle weakness. Hydrochlorothiazide USP/BP 50 mg The Volume Reducer: Excretes excess sodium and water to reduce the load on the heart and vessels. Excipients Proprietary Matrix Steady-State Release System: Engineered to ensure a consistent diuretic effect over a full 24-hour cycle. Aminosalicylic Acid Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Delayed-Release (DR) Tablet, packed in moisture-resistant Alu-Alu blister strips.
Active Ingredient Strength Primary Clinical Function 4-Aminosalicylic Acid USP/BP 500 mg The Standard Titration Unit: Allows for gradual dose escalation to manage gastrointestinal side effects. 4-Aminosalicylic Acid USP/BP 1000 mg The Maintenance Payload: High-dose format for adult MDR-TB regimens requiring up to 8–12g daily. Excipients Acid-Resistant Polymer Enteric Shield System: Engineered to bypass the stomach and release the API in the upper intestine for maximum absorption. Amiodarone Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Amber Glass Ampoules. The formulation utilizes specific co-solvents to solubilize the lipophilic Amiodarone molecule.
Active Ingredient Strength (Standard) Therapeutic Role Amiodarone HCl BP/USP 150 mg / 3 ml (50 mg/ml) Antiarrhythmic Agent Vehicle / Solvents Q.S. Polysorbate 80 / Benzyl Alcohol Solubilizing Agents *Pack Sizes: Tray of 5 Ampoules or 10 Ampoules.
Amiodarone injection
FreeComposition
Amiodarone 150 mg / 3 ml ( leaflet inside ) ( 24 months shelf life )
Pcd pharma franchise:-
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Amiodarone Tablet
FreeAmiodarone Tablets IP 100 mg
Each Uncoated Tablet contains:
Amiodarone Hydrochloride IP 100 mg
AMODAR – 200
Amiodarone Tablets IP 200 mg
Each Uncoated Tablet contains:
Amiodaron Hydrochloride IP 200 mg
Amodar – 200
Amiodarone Tablets BP 200 mg
Each Uncoated tablet contains:
Amiodarone Hydrochloride BP 200 mg
Usage: – Restore normal heart rhythm and maintain a regular, steady heartbeat.
Category: – Antiarrhytmics drugs
Therapeutic category: – Antiarrhythmics
Amisulpride Tablets
FreeProduct Composition & Strength
We supply this product in a full range of strengths to facilitate both low-dose “Disthymia” protocols and high-dose “Schizophrenia” maintenance.
Active Ingredient Strength Primary Clinical Function Amisulpride USP/BP 50 mg / 100 mg The Negative Symptom Unit: Used to treat social withdrawal, depression, and lack of initiative. Amisulpride USP/BP 200 mg The Transition Anchor: Used for moderate psychosis or as a step-up dose during titration. Amisulpride USP/BP 400 mg The Positive Symptom Payload: High-potency dose for acute psychotic episodes and hallucinations. Amitriptyline & Chlordiazepoxide Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Film-Coated Tablet, packed in high-barrier Alu-Alu blister strips to ensure the stability of the benzodiazepine component.
Active Ingredient Strength Primary Clinical Function Amitriptyline (as HCl) USP/BP 12.5 mg / 25 mg The Antidepressant Anchor: Elevates mood and energy levels by modulating neurotransmitter reuptake. Chlordiazepoxide USP/BP 5 mg / 10 mg The Anxiolytic Shield: Provides rapid relief from nervousness, agitation, and muscle tension. Excipients Proprietary Matrix Bio-Synchronized System: Engineered to ensure the rapid onset of the anxiolytic effect while the antidepressant effect builds over time. Amitriptyline Hydrochloride Tablet
FreeAmitriptyline Hydrochloride Tablets IP 10 mg
Each suger coated tablets contains: Amitriptyline Hydrochloride IP 10 mg
– Excipients –Amitriptyline Hydrochloride Tablets USP 10 mg
Each Sugarcoated tablet contains :
Amitriptyline Hydrochloride USP 10 mgAmitriptyline Hydrochloride Tablets IP 25 mg
Each Sugarcoated tablet contains :
Amitriptyline Hydrochloride IP 25 mgAmitriptyline Tablets BP 25 mg
Each Sugarcoated tablet contains:
Amitriptyline Hydrochloride BP 25 mgAmitriptyline Hydrochloride Tablets IP 75 mg
Each Sugarcoated tablet contains :
Amitriptyline Hydrochloride IP 75 mgUsage: – treat mental/mood problems such as depression
Category: – Anti psychotic + CNS Drugs
Therapeutic category: – Anti psychotic,Antidepressant,CNS Drugs,Anti Anxiety
Amlodipine & Enalapril Maleate Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets to protect the Enalapril Maleate from moisture-induced degradation and to ensure the physical integrity of the combination.
Active Ingredient Strength (Standard) Therapeutic Role Amlodipine (as Besylate) BP/USP 5 mg / 10 mg Calcium Channel Blocker (CCB) Enalapril Maleate BP/USP 5 mg / 10 mg / 20 mg ACE Inhibitor Excipients Q.S. Sodium Stearyl Fumarate / Lactose Lubricant & Stabilizer Amlodipine & Hydrochlorothiazide Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Film-Coated Tablet, engineered for synchronized 24-hour hemodynamic control.
Active Ingredient Strength Primary Clinical Function Amlodipine (as Besylate) USP/BP 5 mg / 10 mg The Vasodilator: Reduces the resistance of blood vessels to lower systemic pressure. Hydrochlorothiazide USP/BP 12.5 mg / 25 mg The Volume Reducer: Excretes excess sodium and water to reduce circulatory load. Excipients Proprietary Matrix Steady-State System: Engineered to ensure consistent plasma concentrations of both drugs to prevent “dips” in BP control. Amlodipine Besylate & Losartan Potassium Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets. The formulation is optimized for 24-hour efficacy, allowing for convenient once-daily dosing.
Active Ingredient Strength (Standard) Therapeutic Role Amlodipine Besylate BP/USP 5 mg Calcium Channel Blocker (CCB) Losartan Potassium BP/USP 50 mg Angiotensin Receptor Blocker (ARB) Excipients Q.S. Microcrystalline Cellulose / Starch Stabilizing Core Matrix Amlodipine With Lisinopril Dehydrate Tablets
FreeProduct Composition & Strength
We supply this product as Uncoated or Film Coated Tablets. The use of Lisinopril in its Dihydrate form ensures superior stability and predictable dissolution profiles.
Active Ingredient Strength (Standard) Therapeutic Role Amlodipine (as Besylate) BP/USP 5 mg / 10 mg Calcium Channel Blocker Lisinopril (as Dihydrate) BP/USP 5 mg / 10 mg / 20 mg ACE Inhibitor Excipients Q.S. Calcium Phosphate / Mannitol Stabilizing Agents Amodiaquine Tablets
FreeAmodiaquine Hydrochloride Tablets USP 100 mg
Each Uncoated Tablet Contains:
Amodiaquine Hydrochloride USP
eq. To Amodiaquine 100 mgAmodiaquine Hydrochloride Tablets USP 200 mg
Each Uncoated Tablet contains
Amodiaquine Hydrochloride USP
Eq. To Amodiaquine 200 mgAmodiaquine Hydrochloride Tablets USP 600 mg
Each Uncoated Tablet Contains:
Amodiaquine Hydrochloride USP
eq. To Amodiaquine 600 mgUsage: – Treat to malaria
Category: – Antimalarial drugs
Therapeutic category: – Antimalarial,
Amoxapine Tablets
FreeProduct Composition & Strength
We supply this product in a full range of strengths to allow for precise clinical titration, packed in high-barrier Alu-Alu blister strips.
Active Ingredient Strength Primary Clinical Function Amoxapine USP/BP 25 mg / 50 mg The Titration Unit: Used for gradual dose escalation to minimize initial sedative effects. Amoxapine USP/BP 100 mg The Standard Anchor: The primary maintenance dose for outpatients with moderate depression. Amoxapine USP/BP 150 mg The High-Potency Payload: For hospitalized patients with severe psychotic or endogenous depression. Amoxicillin & Potassium Clavulanate Dispersible Tablets
FreeProduct Composition & Strength
We supply this product in the globally standardized 7:1 ratio, packed in high-barrier Alu-Alu blister strips to ensure the stability of the moisture-sensitive Clavulanate.
Active Ingredient Strength Primary Clinical Function Amoxicillin (as Trihydrate) USP/BP 200 mg / 400 mg The Growth Inhibitor: Destroys the bacterial cell wall across a broad range of pathogens. Potassium Clavulanate USP/BP 28.5 mg / 57 mg The Resistance Shield: Neutralizes bacterial enzymes, allowing Amoxicillin to function. Excipients Sweetened Matrix Pediatric-Friendly System: Engineered with pleasant fruit flavors to ensure 100% child compliance. Amoxicillin & Potassium Clavulanate Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet, packed exclusively in highly secure, ultra-low moisture Alu-Alu blister strips (often containing integrated desiccants) to ensure the absolute stability of the incredibly hygroscopic Clavulanate salt.
Active Ingredient Strength Primary Clinical Function Amoxicillin Trihydrate USP/Ph.Eur.
Potassium Clavulanate USP/Ph.Eur.500 mg
125 mg
(Total: 625 mg)Standard Adult Dose: Therapy for moderate RTIs, dental infections, and skin infections. Amoxicillin Trihydrate USP/Ph.Eur.
Potassium Clavulanate USP/Ph.Eur.875 mg
125 mg
(Total: 1000 mg)High-Dose Adult Therapy: Aggressive intervention for severe pneumonia, severe sinus infections, and bite wounds. Excipients Microcrystalline Cellulose / Sodium Starch Glycolate / Colloidal Silicon Dioxide / Magnesium Stearate Diluent / Superdisintegrant / Moisture Scavenger (Engineered for immediate gastric dissolution) *Pack Sizes: 1×6 or 1×10 Alu-Alu Blisters (Optimized specifically for strict 5-to-7 day acute antibiotic regimens).
Amoxicillin Capsules
FreeAmoxicillin capsules are common penicillin-type antibiotics typically available in 250 mg and 500 mg strengths.
Amoxicillin Sodium Injection 250mg
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. It must be reconstituted with Sterile Water for Injection prior to use.
Active Ingredient Strength (Pediatric Dose) Therapeutic Role Amoxicillin Sodium USP/BP Equivalent to 250 mg Amoxicillin Broad Spectrum Antibiotic Excipients None (Pure Sterile Powder) – – *Pack Sizes: Tray of 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Amoxicillin Sodium Injection 500mg
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. It must be reconstituted with Sterile Water for Injection prior to use.
Active Ingredient Strength (Standard Adult Dose) Therapeutic Role Amoxicillin Sodium USP/BP Equivalent to 500 mg Amoxicillin Broad Spectrum Antibiotic Excipients None (Pure Sterile Powder) – – *Pack Sizes: Tray of 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Amoxicillin Trihydrate Dispersible Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Fruit-Flavored Tablet, packed in high-barrier Alu-Alu blister strips to ensure the stability of the moisture-sensitive API.
Active Ingredient Strength Primary Clinical Function Amoxicillin (as Trihydrate) USP/BP 125 mg The Pediatric Starter: Ideal for infants and toddlers requiring low-dose respiratory or ear infection therapy. Amoxicillin (as Trihydrate) USP/BP 250 mg The Junior Anchor: The standard dose for older children and adolescents for broad-spectrum anti-infective coverage. Excipients Sweetened Matrix Taste-Masking System: Engineered with pleasant flavors (Strawberry/Pineapple/Orange) to overcome the natural bitterness of the antibiotic. Amoxycillin Injection
FreeComposition
Amoxycillin 1gm + Clavulanic Acid 200mg
with WFI Dry Powder Injection
Amoxycillin 1000mg, Potassium
Clavulante 200mg 1.2gm Injection
Pcd pharma franchise:-
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Amoxycillin , Cloxacillin And Lactic Acid Bacillus Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Film-Coated Tablet or Capsule, packed in high-barrier Alu-Alu blister strips to ensure the stability of the live probiotic spores.
Active Ingredient Strength Primary Clinical Function Amoxycillin (as Trihydrate) USP/BP 250 mg The Growth Inhibitor: Destroys the bacterial cell wall across a broad range of pathogens. Cloxacillin (as Sodium) USP/BP 250 mg The Defense Breaker: Specifically neutralizes bacterial enzymes, making the treatment effective against “Staph” infections. Lactic Acid Bacillus (LAB) 1.5 Billion Spores The Biological Shield: Replenishes healthy gut bacteria to prevent diarrhea and boost immunity. Amoxycillin & Cloxacillin Capsules
FreeAmoxycillin and cloxacillin capsules (commonly known as co-amoxiclox) are combination antibiotics used to treat infections where bacteria might produce enzymes that resist standard penicillin. They are typically available in a 500 mg total strength (250 mg / 250 mg)
Amoxycillin & Cloxacillin veterinary injection
FreeComposition
Amoxycillin & Cloxacillin VET INJ. 4.5GM
Usage: –Here are some common usages for these antibiotics in veterinary medicine:
Category: – veterinary injection
Therapeutic category: – veterinary medicine
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Amoxycillin & Dicloxacillin Capsules
FreeAmoxycillin and dicloxacillin capsules are combination antibiotics designed to broaden the spectrum of treatment while protecting the medicine from bacterial resistance. They are typically available in a 500 mg total strength (often 250 mg / 250 mg)
Amoxycillin & Dicloxacillin Dispersible Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Dispersible Tablet, packed in high-barrier Alu-Alu blister strips to ensure the stability of the moisture-sensitive APIs.
Active Ingredient Strength Primary Clinical Function Amoxycillin (as Trihydrate) USP/BP 125 mg / 250 mg The Growth Inhibitor: Destroys the bacterial cell wall across a broad range of common pathogens. Dicloxacillin (as Sodium) USP/BP 125 mg / 250 mg The Enzyme Bodyguard: Specifically neutralizes bacterial defenses, making the treatment effective against “Staph” infections. Excipients Sweetened Matrix Pediatric-Friendly System: Engineered with non-bitter, pleasant flavors (Pineapple/Orange) to ensure child compliance. Amoxycillin & Dicloxacillin veterinary injection
FreeComposition
Amoxycillin & Dicloxacillin VET INJ. 4 GM
Usage: – used to treat staphylococcal infections in veterinary medicine
Category: – veterinary injection
Therapeutic category: – veterinary medicine
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Amoxycillin & Potassium Clavulanate Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The ratio is critically balanced (usually 5:1) for optimal pharmacokinetics in intravenous use.
Strength Variant Amoxicillin Content (as Sodium) Clavulanic Acid Content (as Potassium) Target Patient 1.2 g Vial 1000 mg (1 g) 200 mg Adults (Severe) 600 mg Vial 500 mg 100 mg Adults / Children 300 mg Vial 250 mg 50 mg Pediatric 150 mg Vial 125 mg 25 mg Infants *Pack Sizes: Tray of 1 Vial or Box of 1/10 Vials with Water for Injection (WFI).
amoxycillin & Sulbactam veterinary injection
FreeComposition
amoxycillin sodium & Sulbactam sodium VET 3 GM
Usage: – veterinary medicine is often used to treat a variety of bacterial infections in animals
Category: – veterinary injection
Therapeutic category: –veterinary medicine
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Amoxycillin Capsules
FreeAmoxycillin capsules are widely used penicillin-type antibiotics. They are most frequently prescribed in 250 mg and 500 mg strengths.Amoxycillin Dispersible Kid Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Fruit-Flavored Tablet, packed in high-barrier Alu-Alu blister strips to ensure the stability of the moisture-sensitive API.
Active Ingredient Strength Primary Clinical Function Amoxycillin (as Trihydrate) USP/BP 125 mg The Pediatric Starter: Ideal for infants and toddlers requiring low-dose respiratory or ear infection therapy. Amoxycillin (as Trihydrate) USP/BP 250 mg The Junior Anchor: The standard dose for older children and adolescents for broad-spectrum anti-infective coverage. Excipients Sweetened Matrix Taste-Masking System: Engineered with pleasant flavors (Strawberry/Pineapple/Orange) to overcome the natural bitterness of the antibiotic. Ampicillin & Cloxacillin Capsules
FreeAmpicillin and cloxacillin capsules (often called co-ampiclox) are combination antibiotics used to treat infections where the bacteria may produce enzymes that destroy standard penicillin. They are most commonly available in a 500 mg total strength (250 mg / 250 mg)
Ampicillin & Cloxacillin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. It must be reconstituted with Sterile Water for Injection prior to use.
Active Ingredient Strength (Standard 1:1 Ratio) Therapeutic Role Ampicillin Sodium USP/BP 250 mg Broad Spectrum Coverage Cloxacillin Sodium USP/BP 250 mg Anti-Staphylococcal Shield Total Vial Strength 500 mg Pediatric / Mild Infection (Adult Variant) 500 mg Ampicillin + 500 mg Cloxacillin (1 g Total) Standard Adult Dose Excipients None (Pure Sterile Powder) – – *Pack Sizes: Tray of 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Ampicillin & Cloxacillin veterinary injection
FreeComposition
Ampicillin & Cloxacillin VET INJ. 3 GM
Ampicillin & Cloxacillin VET INJ. 4.5 GM
Usage: – used antibiotics in veterinary medicine, often in combination, to treat a variety of bacterial infections in animals.
Category: – veterinary injection
Therapeutic category: – veterinary medicine
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Ampicillin & Dicloxacillin Capsules
FreeAmpicillin and dicloxacillin capsules are combination antibiotics designed to provide broad-spectrum coverage while resisting degradation by certain bacterial enzymes. They are commonly available in a 500 mg total strength (typically 250 mg / 250 mg).
Ampicillin and Flucloxacillin Capsules
FreeAmpicillin and flucloxacillin capsules (often referred to by the co-drug name co-fluampicil) are combination antibiotics used to treat infections where the specific bacteria is unknown or suspected to be a penicillin-resistant strain. They are typically available in a 250 mg/250 mg strength.
Ampicillin Capsules
FreeAmpicillin capsules are a penicillin-type antibiotic used to treat various bacterial infections. They are most commonly available in 250 mg and 500 mg strengths.
Ampicillin Injection 500mg
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. It must be reconstituted with Sterile Water for Injection prior to use.
Active Ingredient Strength (Standard Pediatric/Mild Dose) Therapeutic Role Ampicillin Sodium USP/BP Equivalent to 500 mg Ampicillin Broad Spectrum Antibiotic Excipients None (Pure Sterile Powder) – – *Pack Sizes: Tray of 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Ampicillin Sulbactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The 2:1 ratio is clinically optimized for pharmacokinetics.
Active Ingredient Strength (Standard 1.5g Vial) Strength (High Dose 3g Vial) Ampicillin Sodium USP/BP 1000 mg (1 g) 2000 mg (2 g) Sulbactam Sodium USP/BP 500 mg (0.5 g) 1000 mg (1 g) Total Vial Content 1.5 g 3.0 g Excipients None (Pure Sterile Powder) None (Pure Sterile Powder) *Pack Sizes: Tray of 10 Vials, or Box of 1 Vial with Water for Injection (WFI).
Anastrozole Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Cytotoxic Tablet, packed in high-barrier Alu-Alu blister strips to ensure the stability of the active API.
Active Ingredient Strength Primary Clinical Function Anastrozole USP/Ph.Eur. 1 mg The Standard Oncology Anchor: The globally validated dose for both adjuvant and first-line treatment of advanced breast cancer. Excipients Proprietary Matrix High-Containment Coating: Engineered with an impenetrable film-coat to prevent the release of cytotoxic dust, ensuring safety for healthcare workers. Antacid / Anti Ulcerant Tablets
FreeAluminium Hydroxide & Magnesium Trisilicate
Each Uncoated chewable tablet contains: Dried Aluminium Hydroxide gel IP 250 mg
Magnesium Trisilicate IP 500 mgAluminium Hydroxide & Magnesium Trisilicate Tablet NFI
Each uncoated Chewable Tablet Contains:
Dried Aluminium Hydroxide Gel IP 120 mg
Magnesium Trisilicate IP 250 mg
Peppermint Oil IP 0.003 mlApixaban Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Bioavailability Tablet, packed in high-barrier Alu-Alu blister strips to ensure the stability of the active API.
Active Ingredient Strength Primary Clinical Function Apixaban USP/Ph.Eur. 2.5 mg The Dose-Reduction Unit: Used for patients with specific risk factors (Age ≥80, Weight ≤60kg, or high Creatinine) to minimize bleeding. Apixaban USP/Ph.Eur. 5 mg The Therapeutic Anchor: The standard maintenance dose for stroke prevention in Atrial Fibrillation and DVT treatment. Excipients Proprietary Matrix Rapid-Dissolution System: Engineered to ensure a consistent $T_{max}$ of approx. 3 to 4 hours for reliable protection. Apremilast tablets
FreeProduct Composition & Strength
We supply this product in a standardized titration pack format or individual strengths to ensure patient safety during the initial phase.
Active Ingredient Strength Primary Clinical Function Apremilast USP/Ph.Eur. 10 mg / 20 mg The Titration Unit: Used during the first 5 days of therapy to minimize gastrointestinal side effects. Apremilast USP/Ph.Eur. 30 mg The Therapeutic Anchor: The standard maintenance dose for long-term control of Psoriasis and Psoriatic Arthritis. Excipients Proprietary Matrix Bio-Synchronized Release: Engineered to ensure a consistent $T_{max}$ of approx. 2.5 hours for steady-state immune modulation. Aripipralzole Tablets
FreeProduct Composition & Strength
We supply this product in a wide range of strengths to allow for precise clinical titration, packed in moisture-resistant Alu-Alu blister strips.
Active Ingredient Strength Primary Clinical Function Aripiprazole USP/Ph.Eur. 2 mg / 5 mg The Titration/Adjunct Unit: Used for initial dosing or as an add-on for Major Depressive Disorder. Aripiprazole USP/Ph.Eur. 10 mg / 15 mg The Standard Anchor: The primary maintenance dose for Schizophrenia and Bipolar Disorder. Aripiprazole USP/Ph.Eur. 20 mg / 30 mg The High-Potency Payload: For acute manic episodes or treatment-resistant cases under specialist care Arteether Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Oily Solution in Amber Glass Ampoules. The oily base (refined Arachis Oil or Ethyl Oleate) ensures depot formation in the muscle.
Active Ingredient Strength (Standard) Therapeutic Role α-β Arteether 150 mg / 2 ml Adult Dose (Standard) α-β Arteether 75 mg / 1 ml Pediatric Dose Vehicle Q.S. Refined Arachis Oil / Ethyl Oleate Oily Base (Sustained Release) *Pack Sizes: Tray of 3 Ampoules (Full Course) or Box of 10 Ampoules.
Artemether & Lumefantrine Tablets
FreeArtmefarin – ADULT
Artemether & Lumefantrine tablets
Each Uncoated tablet contains:
Artemether 80mg
Lumefantrine 480 mg
Excipients q.sArtmefarin – Disp
Artemether & Lumefantrine tablets
Each Uncoated Dispersible tablet contains:
Artemether 20 mg
Lumefantrine 120 mg
Excipients q.sArtmefarin – DS
Artemether & Lumefantrine Tablets
Each Film Tablet contains:
Artemether 40mg
Lumefantrine 240 mg
Excipients q.s
Approved Colour UsedARTMEFARIN / ARETEMETHER & LUMEFANTRINE TABLETS
Each uncoated tablet: – ARETEMETHER 20 mg
– LUMEFANTRINE 120 mgArtemether & Lumefantrine Tablets
Each Uncoated tablet contains:
Artemether 80mg
Lumefantrine 480 mg
Excipients q.sartemether & lumefantrine tablets 20 / 120 mg
Each Uncoated Dispersible tablet Contains: –
artemether In House 20 mg
Lumefantrine In House 120 mg
titanium dioxide –
Excipients – QSUsage: – Treat to malaria
Category: – Antimalarial drugs
Therapeutic category: – Antimalarial,
Artemether 40mg Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Oily Solution in Amber Glass Ampoules. The oily base ensures a depot effect for sustained release.
Active Ingredient Strength (Standard) Therapeutic Role Artemether IP/Ph.Int 40 mg / 1 ml Pediatric / Low Body Weight Dose Artemether IP/Ph.Int 80 mg / 1 ml Adult Standard Dose Vehicle Q.S. Refined Arachis Oil / Ethyl Oleate Oily Base (Depot) *Pack Sizes: Tray of 6 Ampoules or 10 Ampoules (Standard Course).
ARTEMETHER INJECTION
FreeComposition
Each ml. Contains: Artemether I.P. 80 mg.
Arachis Oil I.P. Q.S 1 ml
Usage: – Treat to malaria
Category: – Antimalarial injection
Therapeutic category: – Antimalarial,
Artemether-80 Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Oily Solution in Amber Glass Ampoules. The 80mg strength is specifically formulated to minimize the injection volume for adult patients while delivering a potent therapeutic dose.
Active Ingredient Strength (Adult Dose) Therapeutic Role Artemether IP/Ph.Int 80 mg Potent Antimalarial Vehicle Q.S. to 1 ml Refined Arachis Oil / Ethyl Oleate *Pack Sizes: Tray of 6 Ampoules or 10 Ampoules (Standard Adult Course).
Artesunate 120mg Injection
FreeProduct Composition & Strength
We supply this product as a Combi-Pack containing the Lyophilized Powder and special solvents required for activation.
Component Content Function Vial (Powder) Artesunate 120 mg (Sterile Lyophilized) Active Antimalarial Ampoule 1 Sodium Bicarbonate Injection (5%) – 1ml Activator / Solvent (Solubilizes the acid) Ampoule 2 Sodium Chloride Injection (0.9%) – 5ml Diluent (Adjusts volume/tonicity) *Pack Sizes: Single Combi-Pack or Tray of 5 Combi-Packs.
Artesunate 30mg Injection
FreeProduct Composition & Strength
We supply this product as a Combi-Pack containing the Lyophilized Powder and special solvents required for activation.
Component Content Function Vial (Powder) Artesunate 30 mg (Sterile Lyophilized) Active Antimalarial Ampoule 1 Sodium Bicarbonate Injection (5%) – 0.5ml Activator (Solubilizes the acid) Ampoule 2 Sodium Chloride Injection (0.9%) – 2.5ml Diluent (Adjusts volume/tonicity) *Pack Sizes: Single Combi-Pack or Tray of 5/10 Combi-Packs.
Artesunate 60mg Injection
FreeProduct Composition & Strength
We supply this product as a Combi-Pack containing the Lyophilized Powder and special solvents required for activation.
Component Content Function Vial (Powder) Artesunate 60 mg (Sterile Lyophilized) Active Antimalarial Ampoule 1 Sodium Bicarbonate Injection (5%) – 0.5ml/1ml Activator (Solubilizes the acid) Ampoule 2 Sodium Chloride Injection (0.9%) – 2.5ml/5ml Diluent (Adjusts volume/tonicity) *Pack Sizes: Single Combi-Pack or Tray of 5 Combi-Packs.
Artesunate Amodiaquine Tablets
FreeGlamosunate Tablets Combipak
Artesunate Tablets + Amodiaquine Hydrochloride Tablets USP
Each Filmcoated tablet contains:
Artesunate IP 50 mg
Each Tablet contains :
Amodiaquine Hydrochloride USP
Eq. To Amodiaquine 200 mg
Usage: – Treat severe MalariaCategory: – Antimalarial drugs
Therapeutic category: – Antimalarial,
Artesunate Mefloquine Tablets
FreeArsugin M Combipack
Artesunate Tablets + Mefloquine Tablets
a) Each Film Coated Tablet Contains:
Artesunate 200 mg
b) Each Film Coated Tablet Contains:
Mefloquine Hydrochloride BP
eq. To Mefloquine 250 mgUsage: – Treat severe Malaria
Category: – Antimalarial drugs
Therapeutic category: – Antimalarial,
Artesunate Tablets
FreeARTESUNATE TABLETS 25 MG
each uncoated tablet contains:
artesunate IP 25 mg
excipients q.s.Armax – 50
Artesunate Tablets 50 mg
Each Filmcoated tablet contains:
Artesunate 50 mgArtesunate Tablets 100 mg
Each uncoated tablet contains:
Artesunate IP 100 mg
Excipients – QSArtesunate Tablets 150 mg
Each Uncoated tablet contains:
Artesunate 150 mg
Excipients – q.sArtesunate Tablets 200 mg
Each Filmcoated tablet contains:
Artesunate IP 200 mgARTESUNATE TABLETS INT. PH.+PYRIMETHAMINE AND
SULPHADOXINE- AThree Tablets Of
– Each uncoated tablet contains:
– SULFADOXINE IP 500 MG
– Pyrimethamine IP 25 MG
– BThree Tablet Of
– Each Film coated tablet contains:
– Artesunate IP 200 MGARTESUNNATE + SULPHADOXINE AND
PYRIMETHAMINE IP COB
Each kit contains:Artisunate tab+sulphadoxine and Pyrimethamine tab
three tab of artesunate 150 mg and two tablets of sulphadoxine and pyrimethamine ip 525 mg
CMB
ARTESUNATE + SULPHADOXINE & PYRIMETHAMINE IP-
5 TO 8 YEARS 3 TABLETS OF ARTESUNATE 100MG AND
1 TABLET OF PYRIMETHAMINE AND SULPHADOXINE
TABLETS IP 750 & 37.5MGCMB
EACH KIT CONTAINSARTESUNATE + SULPHADOXINE & PYRIMETHAMINE IP – ADULT 15 YEARS & ABOVE
[ 3 TABLETS OF ARTESUNATE 200 MG AND 3 TABLET OF PYRIMETHAMINE AND SULPHADOXINE TABLETS IP 500 & 25 MG]
CMB
ARTESUNATE + SULPHADOXINE & PYRIMETHAMINE IP – INFANT BELOW 1 YEAR
[ 3 TABLETS OF ARTESUNATE 25 MG AND 2 TABLETS OF SULPHADOXINE & PYRIMETHAMINE TABLETS IP 125 & 6.25 MG] CMB Artesunate Tablets + Pyrimethamine & Sulphadoxine Tablets IP Age 1 to 4 Years CMB
3 TABLETS OF ARTESUNATE AND 1 TABLET OF PYRIMETHAMINE & SULPHADOXINE Each Combipack Contains:
ARTESUNATE TABLETS 50 MG
Each film coated tablet contains:
ARTESUNATE IP 50 mgPYRIMETHAMINE AND SULPHADOXINE TABLETS IP
ARTESUNATE TABLETS + PYRIMETHAMINE & SULPHADOXINE TABLETS IP Age 9 to 14 years CMB
3 TABLETS OF ARTESUNATE AND 2 TABLETS OF PYRIMETHAMINE & SULPH
ARTESUNATE TABLETS 150 MG
Each Uncoated tablet contains:
Artesunate 150 mg
Excipients – q.sPYRIMETHAMINE AND SULPHADOXINE TABLETS IP
Supp Each uncoated tablet contains: Sulphadoxine IP 500 mg
Pyrimethamine IP 25 mg
Excipients – QSArsugin M Combipack
Artesunate Tablets + Mefloquine Tablets
a Each Film Coated Tablet Contains:
Artesunate 200 mg
b Each Film Coated Tablet Contains:
Mefloquine Hydrochloride BP
eq. To Mefloquine 250 mgArsugin SP Combipack
Artesunate Tablets +
Sulfadoxine and Pyrimethamine Tablets USP
a Each Film Coated Tablet Contains:
Artesunate 200 mg
b Each Uncoated Tablet Contains:
Sulfadoxine USP 500 mg
Pyrimethamine USP 25 mgUsage: – Treat severe Malaria
Category: – Antimalarial drugs
Therapeutic category: – Antimalarial,
Ascorbic Acid Tablets
FreeAscorbic Acid Tablets IP 100 mg
Each Uncoated tablet contains :
Ascorbic Acid IP 100 mgAscorbic Acid Tablet IP 300 mg
Each Uncoated Tablet Contains:
Sodium Ascorbate IP
eq. To Ascorbic Acid 300 mgHealthy Vita C
(Ascorbic Acid tablets BP 500 mg)
Each Chewable uncoated tablet contains:
Ascorbic Acid BP 500 mg
Usage: – Treat or prevent low levels of vitamin C and to treat scurvyCategory: – Vitamin supplement
Therapeutic category: – Vitamin supplement
Aspirin Tablets
FreeProduct Composition & Strength
We supply this product in various formats, including Gastro-resistant (Enteric-coated), Dispersible, and standard Film-coated tablets.
Active Ingredient Strength Primary Clinical Function Aspirin (Acetylsalicylic Acid) 75 mg / 81 mg The Cardiac Shield: Low-dose daily therapy for the prevention of heart attack and stroke. Aspirin (Acetylsalicylic Acid) 150 mg Post-Surgical Maintenance: Used for patients with stents or high-risk vascular profiles. Aspirin (Acetylsalicylic Acid) 300 mg / 500 mg The Analgesic Anchor: High-dose relief for acute pain, migraine, and rheumatic fever. Atenolol & Chlorthalidone Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets. The combination is engineered to maintain therapeutic plasma levels over a 24-hour period, supporting once-daily dosing.
Active Ingredient Strength (Standard) Therapeutic Role Atenolol BP/USP 50 mg / 100 mg Cardioselective Beta-Blocker Chlorthalidone BP/USP 12.5 mg / 25 mg Long-acting Diuretic Excipients Q.S. Magnesium Stearate / Povidone Stabilizing Core Matrix Atenolol & S-Amlodipine Besylate Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Film-Coated Tablet, utilizing high-purity S-Amlodipine Besylate to ensure stability.
Active Ingredient Strength Primary Clinical Function Atenolol USP/BP 25 mg / 50 mg The Heart Rate Regulator: Controls the “Pump” by reducing heart rate and myocardial oxygen demand. S-Amlodipine (as Besylate) USP/BP 2.5 mg / 5 mg The Vessel Relaxant: Pure chiral isomer for maximum vasodilation with minimum side effects (edema). Excipients Proprietary Matrix Chiral-Stability System: Engineered to ensure the S-enantiomer remains stable and does not racemize during the shelf-life. Atenolol Tablets
FreeAtenolol Tablets IP 25 mg Each Uncoated Tablet
contains :- Atenolol IP 25 mg
Excipients – QSAtenolol Tablets IP 50 mg
Each Uncoated Tablet contains :
Atenolol IP 50 mg
Excipients q.sOtenol 50
Atenolol Tablets BP
Each Uncoated tablet contains :
Atenolol BP 50 mgOtenol
Atenolol Tablets BP
Each Uncoated tablet contains :
Atenolol BP 100 mgUsage: – Treat high blood pressure and irregular heartbeat
Category: – Hypertensive Cardiac drugs
Therapeutic category: – Cardiovascular Agent,Anti hypertensive
Atorvastatin & Aspirin and Clopidogrel Tablets
FreeProduct Composition & Strength
We supply this product typically in a Capsule-in-Capsule or Multi-Particulate Tablet format to ensure the chemical stability of the three distinct APIs.
Active Ingredient Strength Primary Clinical Function Atorvastatin (as Calcium) USP 10 mg / 20 mg The Lipid Stabilizer: Lowers cholesterol and prevents the growth of arterial blockages. Aspirin (Gastro-resistant) BP 75 mg / 150 mg The Primary Blood Thinner: Prevents initial platelet activation via the COX pathway. Clopidogrel (as Bisulphate) USP 75 mg The Secondary Blood Thinner: Provides high-level protection against stent thrombosis and recurrent stroke. Atorvastatin & Asprin Tablets
FreeProduct Composition & Strength
We supply this product as a Bi-Layered or Film-Coated Tablet, often utilizing specialized enteric coating for the Aspirin component to protect the stomach lining.
Active Ingredient Strength Primary Clinical Function Atorvastatin Calcium USP/BP 10 mg / 20 mg The Lipid Regulator: Controls cholesterol synthesis in the liver and stabilizes arterial walls. Aspirin (Gastro-resistant) BP 75 mg / 150 mg The Anti-Platelet Anchor: Provides low-dose blood thinning to prevent clot formation. Excipients Proprietary Matrix Gastro-Protective System: Engineered to ensure Aspirin dissolves only in the small intestine, minimizing gastric irritation. Atorvastatin Clopidogrel and Aspirin Capsules
FreeAtorvastatin, clopidogrel, and aspirin capsules are fixed-dose combination (FDC) medications used for the secondary prevention of cardiovascular events like heart attacks and strokes.
Atorvastatin Tablets
FreeAtorvastatin Tablet IP 10 mg
Each Film Coated Tablet Contains :
Atorvastatin Calcium IP
Eq. to Atorvastatin 10 mgAtorvastatin Tablets IP 20 mg
Each Filmcoated tablet contains :
Atorvastatin Calcium IP
Eq. To Atorvastatin 20 mgUsage: – Lower cholesterol
Category: – Antibiotic drugs
Therapeutic category: – Antilipidemic,Cardiovascular Agent
Atropine Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Clear Glass Ampoules.
Active Ingredient Strength (Standard) Therapeutic Role Atropine Sulphate USP/BP 0.6 mg / ml Pre-operative / Cardiac Dose Atropine Sulphate USP/BP 1 mg / ml Emergency / Antidote Dose Excipients Q.S. Sodium Chloride / Sulfuric Acid (pH adjustment) Isotonicity / Stabilizer *Pack Sizes: Tray of 10 Ampoules, 50 Ampoules, or 100 Ampoules (Hospital/Military Pack).
Atropine Sulphate injection
FreeComposition
Each ml Contains :
Atropine Sulphate I.P. ……… 1 mg
Water for Injec9on I.P. Q.S. 10 mlEach ml. Contains:
Atropine Sulphate I.P. ……..………. 0.6 mg,
Methyl Paraben I.P. ……………. 0.18% w/v
Propyl Paraben I.P. ……………. 0.02% w/v
(As Preserva9ve )
Water for Injec9on I.P ………… Q.S. 1mlUsage: – In some cases, atropine sulfate may be used in ophthalmology
Category: –antimuscarinic injection
Therapeutic category: – antimuscarinic drugs
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Atropine Sulphate veterinary injection
Freecomposition
Atropine Sulphate VET INJ. 100 ML
Atropine Sulphate VET INJ. 10 ML
Usage: – In some cases, atropine sulfate may be used in veterinary ophthalmology
Category: –atropine sulfate for veterinary injection
Therapeutic category: – atropine sulfate for veterinary injection
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Azathioprine Tablets
FreeATHEPRIN TABLETS
Azathioprine Tablets IP 50 mg
Each Uncoated Tablet Contains :
Azathioprine IP 50 mgUsage: – Prevent organ rejection in people who have received a kidney transplant
Category: – Arthritis drugs
Therapeutic category: – Arthritis
Azithromycin & Cefixime tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Film-Coated Tablet, packed in high-barrier Alu-Alu blister strips to prevent the degradation of the moisture-sensitive API.
Active Ingredient Strength Primary Clinical Function Cefixime (as Trihydrate) USP/BP 200 mg The Gram-Negative Specialist: Targets the cell walls of stubborn bacteria, highly effective in Typhoid and Bronchitis. Azithromycin (as Dihydrate) USP/BP 250 mg / 500 mg The Tissue-Penetrating Anchor: Provides prolonged activity in the lungs and soft tissues to eliminate intracellular pathogens. Excipients Proprietary Matrix Dual-Release System: Engineered to ensure synchronized absorption of both antibiotics despite their different chemical solubilities. Azithromycin Dispersible Tablet
FreeProduct Composition & Strength
We supply this product as Dispersible Tablets (DT) with advanced taste-masking technology.
Active Ingredient Strength Flavor Profile Role Azithromycin IP/BP/USP 100 mg Orange / Peppermint Pediatric Low Dose Azithromycin IP/BP/USP 250 mg Orange / Peppermint Pediatric Standard / Adult Excipients Q.S. Sweeteners Disintegrant / Flavor Azithromycin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Infusion in glass vials. It must be reconstituted and further diluted before administration.
Active Ingredient Strength (Standard Adult Dose) Therapeutic Role Azithromycin (as Dihydrate/Citrate) Equivalent to 500 mg Azithromycin Broad Spectrum Macrolide Excipients Q.S. Citric Acid / Sodium Hydroxide pH Adjuster / Solubilizer *Pack Sizes: Tray of 1 Vial, 5 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Azithromycin Tablets
FreeAzithromycin Dispersible Tablet 100mg
Each Disersible tablet cotain:
Azithromycin Dihydrate
Eq.To Anhydrous Azithromycin(100mg)
Std:IP(ACT)
Excipients(QS)(EXC)Azithromycin Tablets IP 250 mg
Each Filmcoated tablet contains:
Azithromycin Dihydrate IP
Eq. To Anhydrous Azithromycin 250 mg
Excipients q.sAzithromycin Tablets IP 500 mg
Each Filmcoated Tablet contains:
Azithromycin Dihydrate IP
Eq. To Anhydrous Azithromycin 500 mg
Excipients q.s
Colour : Ponceau 4RAzithromycin Tablets IP 500 mg
Each Filmcoated Tablet contains:
Azithromycin Dihydrate IP
Eq. To Anhydrous Azithromycin 500 mg
Excipients q.s
Colour : Ponceau 4RAzithromycin Tablets IP 1 gm
Each Film coated tablet contains:
Azithromycin Anhydrous IP 1 gm
As Azithromycin Dihydrate IPLAMITHROCIN / Azithromycin Tablets USP 500 Mg
Each film coated tablet contains:
– Azithromycin Dihydrate
eq. to Azithromycin USP (500 mg)
– Excipients (- QS)Usage: – chest infections
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic
Baclofen Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Disintegration Tablet, packed in moisture-resistant Alu-PVC or Alu-Alu blister strips.
Active Ingredient Strength Primary Clinical Function Baclofen USP/BP/Ph.Eur. 10 mg The Standard Titration Unit: The primary dose used for gradual escalation to determine patient tolerance and efficacy. Baclofen USP/BP/Ph.Eur. 20 mg The Maintenance Payload: High-potency dose for patients with severe spasticity from chronic spinal cord injuries. Excipients Proprietary Matrix Rapid-Release System: Engineered to ensure a predictable $T_{max}$ (time to peak concentration) of 1 to 2 hours for fast relief. Benfotiamine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Bioavailability Tablet, packed in high-barrier Alu-Alu or Alu-PVC blister strips.
Active Ingredient Strength Primary Clinical Function Benfotiamine USP/Ph.Eur. 100 mg / 150 mg The Neuropathy Anchor: Standard daily dose for the prevention of diabetic complications and nerve maintenance. Benfotiamine USP/Ph.Eur. 300 mg The Therapeutic Max-Payload: High-potency dose for clinical management of established Peripheral Neuropathy and Retinopathy. Excipients Proprietary Matrix Lipid-Enhanced System: Engineered to ensure maximum intestinal uptake and stable plasma concentrations. Benzathine Penicillin Injection 1.2 MIU
FreeProduct Composition & Strength
We supply this product as a Sterile Powder for Suspension in glass vials. It requires reconstitution with Sterile Water for Injection to form a milky white suspension.
Active Ingredient Strength (International Units) Therapeutic Role Benzathine Penicillin G USP/BP 1,200,000 IU (1.2 MIU) Standard Adult Prophylaxis Excipients Q.S. Lecithin / Sodium Citrate Suspending / Buffering Agents *Pack Sizes: Tray of 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Benzathine Penicillin Injection 2.4 MIU
FreeProduct Composition & Strength
We supply this product as a Sterile Powder for Suspension in glass vials. It requires reconstitution with Sterile Water for Injection to form a milky white suspension.
Active Ingredient Strength (International Units) Therapeutic Role Benzathine Penicillin G 1,200,000 IU (1.2 MIU) Pediatric / Standard Dose Benzathine Penicillin G 2,400,000 IU (2.4 MIU) Adult Syphilis / High Dose Benzathine Penicillin G 600,000 IU (0.6 MIU) Pediatric Prophylaxis Excipients Q.S. Lecithin / Sodium Citrate Suspending / Buffering Agents *Pack Sizes: Tray of 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Benzhexol Tablet
FreeProduct Composition & Strength
We supply this product as Uncoated Tablets. The chemical name Benzhexol and Trihexyphenidyl refer to the exact same molecule.
Active Ingredient Strength (Standard) Role Trihexyphenidyl HCl (Benzhexol) 2 mg Initial Dose / Mild EPS Trihexyphenidyl HCl (Benzhexol) 5 mg Forte / Maintenance Dose Excipients Q.S. Binder / Filler Benzthiazide & Triamterene Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Disintegration Tablet, packed in high-barrier Alu-PVC or Alu-Alu blister strips.
Active Ingredient Strength Primary Clinical Function Benzthiazide USP/BP 25 mg The Thiazide Payload: The primary driver for sodium and water excretion to lower blood pressure. Triamterene USP/BP 50 mg The Potassium Guardian: Physically prevents the loss of Potassium (K+) during the diuretic process. Excipients Proprietary Matrix Bio-Balance System: Engineered to ensure synchronized dissolution of both active APIs for maximum clinical synergy. Benzyl Penicillin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Buffered Powder for Injection in glass vials. The potency is measured in International Units (IU).
Active Ingredient Strength (IU) Mass Equivalent Therapeutic Role Benzyl Penicillin Sodium 1,000,000 IU (1 MU) ~600 mg Standard Dose Benzyl Penicillin Sodium 5,000,000 IU (5 MU) ~3000 mg High Dose (Meningitis) Benzyl Penicillin Sodium 10,000,000 IU (10 MU) ~6000 mg Severe Sepsis Buffering Agent Sodium Citrate – pH Stabilizer *Pack Sizes: Tray of 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Betahistine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Disintegration Tablet, packed in high-barrier Alu-Alu blister strips to ensure the stability of the active dihydrochloride salt.
Active Ingredient Strength Primary Clinical Function Betahistine Dihydrochloride USP/Ph.Eur. 8 mg / 16 mg The Standard Maintenance Anchor: Ideal for long-term management of vertigo and tinnitus. Betahistine Dihydrochloride USP/Ph.Eur. 24 mg / 48 mg The Therapeutic Payload: High-potency dose for acute phases of Meniere’s Disease and severe vestibular dysfunction. Excipients Proprietary Matrix Gastric-Gentle System: Engineered to ensure rapid absorption while minimizing the common “histamine-like” gastric irritation. Bethanecol Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Bioavailability Tablet, packed in secure Alu-PVC or Alu-Alu blister strips.
Active Ingredient Strength Primary Clinical Function Bethanechol Chloride USP/BP 10 mg The Pediatric & Titration Unit: Ideal for initial dosing or mild cases of gastric atony. Bethanechol Chloride USP/BP 25 mg The Urology Anchor: The standardized therapeutic dose for managing post-operative urinary retention. Excipients Proprietary Matrix Acid-Stable Formulation: Engineered to survive the gastric environment for consistent intestinal absorption. Biotin Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Compressed, High-Bioavailability Tablet, packed in moisture-resistant Alu-PVC or Alu-Alu blister strips.
Active Ingredient Strength Primary Clinical Function Biotin USP/BP (Vitamin H) 5 mg The Standard Maintenance Unit: The definitive therapeutic dose for treating brittle nails and mild hair thinning. Biotin USP/BP (Vitamin H) 10 mg The Therapeutic Max-Payload: High-potency dose utilized for severe deficiency, advanced dermatology protocols, and metabolic support. Excipients Proprietary Matrix Rapid-Release System: Engineered to ensure 100% water solubility for immediate intestinal absorption. Biperiden Tablets
FreeBiperiden Hydrochloride Tablets 2 mg
Each Filmcoated Tablet Contains:
Biperiden Hydrochloride BP 2 mgUsage: – stiffness, tremors, spasms, poor muscle control
Category: – Antiparkinson drugs
Therapeutic category: –
Bisacodyl Tablets
FreeBisacodyl Tablets IP 5 mg
Each Enteric tablet contains :
Bisacodyl IP 5 mgBISLAX
Bisacodyl Tablets BP 5 mg
Each Enteric tablet contains :
Bisacodyl BP 5 mgNovolax
Bisacodyl Tablets BP
Each Enteric Coated tablet contains :
Bisacodyl BP 5 mgU-LAX
Bisacodyl Tablets BP
Each Enteric coated tablet contains:
Bisacodyl BP 5 mgUsage: – Treat to constipation
Category: – Laxative drugs
Therapeutic category: –
Bisoprolol Tablets
FreeSolol
Bisoprolol Fumarate Tablets USP
Each Film Coated Tablet Contains:
Bisoprolol Fumarate USP 5 mgUsage: – Treat high blood pressure and heart failure
Category: – Anti Hypertensive / Cardiac Drugs
Therapeutic category: – Antihypertensive
Bleomycin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder in glass vials. Potency is measured in USP Units, not milligrams.
Active Ingredient Strength Therapeutic Role Bleomycin Sulphate USP/IP 15 Units (approx. 15 mg) Antineoplastic Antibiotic Excipients Nitrogen (Inert Atmosphere) None (Pure Powder) *Pack Sizes: Single Vial Box or Tray of 10 Vials.
Bortenat
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder in glass vials. It must be reconstituted with Normal Saline (0.9% NaCl).
Active Ingredient Strength Reference Brand Bortezomib (as Mannitol Ester) 2 mg / Vial Bortenat 2mg Bortezomib (as Mannitol Ester) 3.5 mg / Vial Bortenat 3.5mg Excipients Mannitol USP/BP Stabilizer *Pack Sizes: Single Vial Box with protective Cytotoxic shrink-wrapping.
Bortezomib Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder in glass vials. It contains Mannitol, which forms a stable boronic ester with Bortezomib for shelf stability.
Active Ingredient Vial Strength Primary Use Bortezomib (as Mannitol Boronic Ester) 2 mg / Vial Single Dose / Low BSA Bortezomib (as Mannitol Boronic Ester) 3.5 mg / Vial Standard Adult Dose Excipients Mannitol (Sterile) Stabilizer / Bulking Agent *Pack Sizes: Single Vial Box with protective Cytotoxic shrink-wrapping.
ortezomib injection is an antineoplastic (cancer) medication supplied as a sterile lyophilised powder (freeze-dried cake) for reconstitution. It is most commonly known by the brand name Velcade.
Bosentan Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Bioavailability Tablet, packed in moisture-resistant Alu-Alu blister strips to ensure the stability of the active API.
Active Ingredient Strength Primary Clinical Function Bosentan (as Monohydrate) USP/Ph.Eur. 62.5 mg The Initiation Unit: Utilized for the first 4 weeks of therapy to assess patient tolerance and liver response. Bosentan (as Monohydrate) USP/Ph.Eur. 125 mg The Maintenance Payload: The standardized therapeutic dose for long-term management of PAH and exercise capacity improvement. Excipients Proprietary Matrix Controlled Disintegration System: Engineered to ensure a predictable pharmacokinetic profile, matching the innovator brand’s $T_{max}$ (time to peak concentration). Bromhexine Hydrochloride Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Disintegration Tablet, packed in moisture-resistant Alu-PVC or Alu-Alu blister strips.
Active Ingredient Strength Primary Clinical Function Bromhexine Hydrochloride BP/USP 8 mg The Respiratory Anchor: The standardized therapeutic unit for adults and children over 12 for the treatment of productive cough. Excipients Proprietary Matrix Rapid Dissolution System: Engineered to ensure the tablet shatters instantly in the stomach for rapid systemic absorption and lung-tissue penetration. Bromocriptin Tablets
FreeBromocriptin Mesylate Tablets IP 2.5 MG
Each uncoated tablet contains
Bromocriptin Mesylate IP
eq.to Bromocriptine 2.5 mgUsage: – high levels of a natural substance prolactin in the body
Category: – Anti Parkinson drugs
Therapeutic category: – Anti Parkinson
Buclizine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Fast-Acting Tablet, packed in high-barrier Alu-PVC or Alu-Alu blister strips.
Active Ingredient Strength Primary Clinical Function Buclizine Hydrochloride BP/USP 25 mg The Universal Anchor: The standardized therapeutic unit for both appetite stimulation (adults/pediatrics) and motion sickness prevention. Excipients Proprietary Matrix High-Disintegration System: Engineered to ensure the tablet dissolves rapidly in the gastric environment for a fast onset of action (typically within 30–60 minutes). Buprenorphine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Compressed Sublingual Tablet, engineered for rapid mucosal absorption.
Active Ingredient Strength Primary Clinical Function Buprenorphine HCl USP/Ph.Eur. 0.4 mg / 2 mg The Pain & Induction Anchor: Low-dose format for chronic pain or the initial phase of addiction treatment. Buprenorphine HCl USP/Ph.Eur. 8 mg The Maintenance Payload: High-strength dose for long-term stabilization in Opioid Use Disorder. Excipients Sublingual Matrix Rapid Dissolution System: Engineered to dissolve completely under the tongue in < 2 minutes for maximum transmucosal bioavailability. Bupropion tablets
FreeProduct Composition & Strength
We supply this product in three distinct Release Profiles, packed in moisture-resistant Alu-Alu or HDPE containers to protect the stability of the hydrochloride salt.
Release Profile Strength Primary Clinical Function Bupropion XL (Extended Release) 150 mg / 300 mg The Depression Anchor: Once-daily dosing (24-hour release) for MDD and Seasonal Affective Disorder. Bupropion SR (Sustained Release) 150 mg The Smoking Cessation Specialist: Twice-daily dosing (12-hour release) for smoking cessation and MDD. Bupropion IR (Immediate Release) 75 mg / 100 mg The Titration Unit: Immediate-release format for precise dose escalation under specialist supervision. Buserelin Acetate ( GnRH Analogue) veterinary injection
FreeComposition
Buserelin Acetate ( GnRH Analogue) INJ. 2.5 ML
Buserelin Acetate ( GnRH Analogue) INJ. 5 ML
Buserelin Acetate ( GnRH Analogue) INJ. 10 ML
usage: –Here are some potential veterinary uses injection
Category: – veterinary injection
Therapeutic category: – veterinary injection
Cabergoline Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Compressed, Micro-Dose Tablet, packed in high-barrier Alu-Alu blister strips or specialized glass vials to ensure the stability of the active API.
Active Ingredient Strength Primary Clinical Function Cabergoline USP/Ph.Eur. 0.5 mg The Standard Specialty Anchor: The definitive therapeutic unit for managing prolactin-related infertility and pituitary tumors. Excipients Proprietary Matrix Micro-Gram Content Uniformity: Engineered utilizing Geometric Dilution techniques to ensure every 0.5 mg tablet contains the exact clinical dose across the entire batch. Calcium & Calcitriol Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Softgel or Tablet, packed in high-barrier Alu-Alu or Blister strips to protect the light-sensitive Calcitriol.
Active Ingredient Strength Primary Clinical Function Calcium Carbonate USP/BP 500 mg (200 mg Elemental) The Mineral Substrate: Provides the high-density elemental calcium needed for skeletal reinforcement. Calcitriol USP/BP 0.25 mcg The Active Hormone: The potent, pre-activated Vitamin D that drives 100% absorption efficiency. Excipients Oil-Based Matrix Softgel Stability: For softgel formats, we utilize a specialized oil-base to ensure the fat-soluble Calcitriol remains fully dissolved and bioavailable. Calcium & Chloecalciferol Tablets
FreeCALCIUM & CHLOECALCIFEROL TABLETS
each film coated tablet contains:
Vitamin D3 250 IU
calcium carbonate IP 1250 mg
Equivalent to Elemental calcium 500 mg
excipients q.s.Calcium Acetate Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Disintegration Tablet, packed in moisture-resistant Alu-Alu or HDPE bottles.
Active Ingredient Strength Primary Clinical Function Calcium Acetate USP/Ph.Eur. 667 mg The Dialysis Anchor: Each 667mg tablet provides 169mg of elemental calcium, the standardized dose for maximum phosphate binding. Excipients Proprietary Matrix Rapid Dissolution System: Engineered to ensure the tablet shatters instantly in the stomach to maximize contact with dietary phosphorus during a meal. Calcium Carbonate with Vitamin D3 Tablets
FreeCalcium Carbonate With Vitamin D3
Each Uncoated Tablet contains :
Calcium Carbonate 1250 Eq Elemental Calcium IP (500 mg )
Cholecalciferol IP (250 IU)
Excipient (Inhouse ) (-QS)Calcium Carbonate With Vitamin D3Tablets
Each Uncoated Chewable Tablet Contains:
Calcium Carbonate IP 625 mg
Vitamin D3 IP 200 IUCalcium Gluconate Tablets IP 500 mg
Each Uncoated Tablet Contains:
Calcium Gluconate IP 500 mgCAL4U TABLETS
Calcium carbonate with Vitamin D3 Tablets
Each Film Coated Tablet Contains:
Calcium Carbonate IP 625 mg
eq. To Calcium 250 mg
Vitamin D3 125 IU
Shelf life: 1 yearSHREYCAL TABLETS
(Calcium, Minerals with Vitamin D3 Tablets )
Each Filmcoated Tablets Contains:
Calcium Citrate USP 1000 mg
Eq To Elemental Calcium 211 mg
Vitamin D3 IP 200 IU
Magnesium Hydroxide IP Eq To Elemental Magnesium 100 mg
Zinc Sulphate USP Eq To Elemental Zinc 4 mg
Colour : Titanium Dioxide IP
Shelf Life: 2 years
Dosage: As Directed by PhysicianCalcium Gluconate Tablets IP 500 mg
Each Uncoated Tablet Contains:
Calcium Gluconate IP 500 mgUsage: – Treat by low calcium levels
Category: – Generic Drugs
Therapeutic category: – Generic Drugs
Calcium Citrate With Vitamin D3 Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets (typically White or Off-White, Oblong/Caplet shape). The coating makes the large tablet easier to swallow.
Active Ingredient Strength (Standard) Therapeutic Role Calcium Citrate USP/BP 1000 mg (Eq. to 210 mg Elemental Ca) Bone Mineralization Vitamin D3 (Cholecalciferol) USP/BP 200 IU / 400 IU / 1000 IU Absorption Enhancer (High Strength Variant) 1200 mg Citrate + 1000 IU D3 Severe Osteoporosis Excipients Q.S. Starch / Magnesium Stearate / HPMC Disintegrant / Coating Calcium Citrate, Vitamin D3, Methylcobalamin, Folic Acid & Pyridoxine HCl Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Potency Tablet, packed in moisture-resistant Alu-Alu or Alu-PVC blister strips to protect the sensitive B-vitamins.
Active Ingredient Strength Primary Clinical Function Calcium Citrate USP/BP 1000 mg The Structural Payload: Provides elemental calcium for bone mineral density (BMD) restoration. Vitamin D3 (Cholecalciferol) IP 200 IU / 400 IU The Absorption Catalyst: Facilitates active intestinal transport of calcium. Methylcobalamin USP/JP 1500 mcg The Nerve Restorer: Active B12 for nerve fiber regeneration and red blood cell formation. Folic Acid USP/BP 1.5 mg The DNA/Heme Synthesis Anchor: Essential for cell division and lowering homocysteine. Pyridoxine HCl (Vit B6) USP 3 mg The Metabolic Co-factor: Supports neurotransmitter synthesis and protein metabolism. Calcium Citrate,Vitamin D3, Magnesium, Zinc Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets (typically White or Blue, Oblong). The coating masks the metallic taste of Zinc and Magnesium.
Active Ingredient Strength (Standard) Therapeutic Role Calcium Citrate USP/BP 1000 mg (Eq. to 210 mg Elemental Ca) Bone Mineralization Vitamin D3 (Cholecalciferol) 200 IU / 400 IU Absorption Enhancer Magnesium Hydroxide USP/BP 100 mg (Eq. to Elemental Mg) D3 Activation / Bone Structure Zinc Sulphate Monohydrate USP/BP 4 mg / 10 mg (Eq. to Elemental Zn) Collagen Synthesis Excipients Q.S. Croscarmellose Sodium / HPMC Disintegrant / Coating Calcium Dobesilate Capsules
FreeCalcium dobesilate capsules are vasoprotective medications primarily used to treat chronic venous insufficiency and diabetic retinopathy. They are most commonly available in a 500 mg strength
Calcium Gluconate Injection
FreeProduct Composition & Strength
We supply this product as a Supersaturated Sterile Solution in 10ml ampoules.
Active Ingredient Concentration Elemental Calcium Content Calcium Gluconate Monohydrate USP/BP 10% w/v (100 mg / ml) 0.465 mEq/ml (Approx 9 mg Ca++ / ml) Stabilizer Calcium D-Saccharate Prevents Crystallization *Pack Sizes: Tray of 10 Ampoules (10ml) or 50 Ampoules.
Calcium Gluconate Tablets
FreeProduct Composition & Strength
We supply this product as Uncoated Tablets or Chewable Tablets in bulk jars or blisters.
Active Ingredient Strength Elemental Calcium Role Calcium Gluconate IP/BP/USP 500 mg ~ 45 mg Pediatric / Maintenance Calcium Gluconate IP/BP/USP 1000 mg (1g) ~ 90 mg Adult Therapeutic Dose Excipients Q.S. — Binder / Sweetener Calcium Pantothanate Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Compressed, High-Purity Tablet, packed in moisture-resistant Alu-PVC or Alu-Alu blister strips to prevent the hygroscopic vitamin from degrading.
Active Ingredient Strength Primary Clinical Function D-Calcium Pantothenate USP/BP 50 mg / 100 mg The Wellness & Hair Anchor: Standard daily dose for nutritional support and maintenance of hair/skin health. D-Calcium Pantothenate USP/BP 250 mg / 500 mg The Therapeutic Payload: High-dose intervention for clinical B5 deficiency, acne management, and post-surgical wound healing. Excipients Proprietary Matrix Moisture-Lock Formulation: Engineered utilizing specialized binders to protect the vitamin from atmospheric humidity, ensuring a 36-year shelf life in Zone IVb tropical climates. Calcium with Vit. D3 & B12 veterinary injection
FreeComposition
Calcium with Vit. D3 & B12 VET INJ. 30 ML
Usage: – The dosage of the injection will depend on the specific needs of the animal
Category: – veterinary injection
Therapeutic category: – Osteoporosis or Bone Disorders
Candesartan Tablets
FreeCandesartan Tablets
Each Film Coated Tablet Contains:
Candesartan Cilexitil 16 mgUsage: – High blood pressure and heart failure
Category: – Anti Hypertensive / Cardiac drugs
Therapeutic category: – Hypertensive
Carbamazepine Tablets
FreeCarbamazepine Tablets IP 200 mg
Each Uncoated Tablet Contains :
Carbamazepine IP 200 mgCarbamezapine Tablets IP 200 mg
Each film Tablet Contains :
Carbamezapine IP 200 mg
excipients q.s.Cirocar – 200 Tablets
Carbamazepine Tablets BP
Each Uncoated Tablet contains :
Carbamazepine BP 200 mgUsage: – Treat to epilepsy
Category: – Hypertensive / Cardiac drugs
Therapeutic category: – Antiepileptic, Anticonvulsant
Carbazochrome Salicylate Injecton
FreeComposition
Each ml Contains :
Carbazochrome Salicylate Equivalent to
Adrenochrome Monosemicarbazone 5 mg
Phenol I.P. …………………… 0.5 %
( As Preserva9ve )
Water for Injec9on I.P. Q.S. 10 ml, 30 mlUsage: – use of medications can vary by country and region.
Category: –hemostatic injection
Therapeutic category: – hemostatic agent
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Carbazochrome Salicylate veterinary injection
FreeComposition
Carbazochrome Salicylate VET INJ. 10 ML
Carbazochrome Salicylate VET INJ. 30 ML
Usage: – Here are some potential usages of carbazochrome salicylate for veterinary injection
Category: – carbazochrome salicylate for veterinary injection
Therapeutic category: – carbazochrome salicylate for veterinary injection
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Carbidopa and Levodopa Tablets
FreeCarbidopa and Levodopa Tablets IP
Each uncoated tablet contains :
Levodopa IP 100 mg
Carbidopa IP
As Anhydrous Carbidopa 10 mgCombimet Tablets
Carbidopa and Levodopa Tablets IP
Each Uncoated tablet contains :
Carbidopa IP
Eq. To anhydrous Carbidopa 25 mg
Levodopa IP 250 mgLEVODOPA AND CARBIDOPA TABLETS 100 +10 MG
Each filmcoated tablet contains: Levodopa BP 100 mg
– Carbidopa BP –
– As Anhydrous Carbidopa BP 10 mg
– Titanium dioxide –
– Excipients – QSCarbimazole Tablets
FreeCarbimazole Tablets BP 10 Mg
Each film coated tablet contains: Carbimazole BP 10 mg Excipient – QS
Usage: – Treat an overactive thyroidCategory: – Thyroid Hormones drugs
Therapeutic category: – Thyroid Hormones
Carboplatin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Liquid Concentrate in glass vials. It is ready to dilute.
Active Ingredient Concentration Vial Size (Total Drug) Carboplatin USP/BP 10 mg / ml 150 mg (15 ml vial) Carboplatin USP/BP 10 mg / ml 450 mg (45 ml vial) Excipients Water for Injection / Mannitol – *Pack Sizes: Single Vial Box with protective Cytotoxic shrink-wrapping.
Carvedilol Tablets
FreeCarvedilol Tablets 3.125 mg
Each Uncoated tablet contains :
Carvedilol BP 3.125 mgCarvedilol Tablets 6.25 mg
Each Uncoated Tablet Contains:
Carvedilol BP 6.25 mgCarvy
Carvedilol Tablets 12.5 mg
Each Film Coated Tablet Contains:
Carvedilol BP 12.5 mgUsage: – Treat high blood pressure and heart failure
Category: – Anthelmintic drugs
Therapeutic category: –
Cefadroxil 500 mg Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet, packed in highly secure, moisture-resistant Alu-Alu or High-Density Alu-PVC blister strips to ensure the absolute stability of the beta-lactam ring.
Active Ingredient Strength Primary Clinical Function Cefadroxil Monohydrate USP/Ph.Eur. 500 mg (Equivalent to anhydrous Cefadroxil) Global Standard: Twice-daily adult therapy for pharyngitis, skin infections, and UTIs. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Macrogol / Magnesium Stearate Diluent / Superdisintegrant / Film-Coating (Engineered for rapid gastric dissolution and high bioavailability) Cefadroxil Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Monohydrate Tablet, packed in high-barrier Alu-PVC or Alu-Alu blister strips to ensure the stability of the active API.
Active Ingredient Strength Primary Clinical Function Cefadroxil (as Monohydrate) USP/Ph.Eur. 250 mg The Pediatric & ENT Anchor: Ideal for Pharyngitis, Tonsillitis, and mild skin infections in adolescents. Cefadroxil (as Monohydrate) USP/Ph.Eur. 500 mg The Dermatology & UTI Payload: High-efficacy therapeutic dose for Cellulitis, Impetigo, and bladder infections. Excipients Proprietary Matrix High-Density Granulation: Engineered to manage the bulk of the 500mg payload while ensuring rapid disintegration and consistent absorption. Cefdinir Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Bioavailability Tablet, packed in high-barrier Alu-Alu blister strips to ensure the stability of the active ingredient.
Active Ingredient Strength Primary Clinical Function Cefdinir USP/Ph.Eur. 300 mg The Respiratory & Skin Anchor: The definitive therapeutic unit utilized for CAP, Sinusitis, and Skin/Soft Tissue Infections (SSTI). Excipients Proprietary Matrix Crystalline Form Stability: Engineered utilizing specialized binders to maintain the Crystalline Form I of Cefdinir, ensuring maximum shelf-life and predictable intestinal absorption. Cefepime & Sulbactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 2:1.
Strength Variant Cefepime Content Sulbactam Content Ratio 1.5 g Vial 1000 mg (1 g) 500 mg (Sodium) 2:1 (Standard) Excipients L-Arginine Buffer / Stabilizer – *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefepime & Tazobactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 8:1.
Strength Variant Cefepime Content Tazobactam Content Ratio 1.125 g Vial 1000 mg (1 g) 125 mg (Sodium) 8:1 (Standard) Excipients L-Arginine Buffer / Stabilizer – *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefepime injection
FreeComposition
Each vial contains: Cefepime Hydrochloride equal to
Cefepime 1 gm, Tazobactam Sodium
Equivalent to Tazobactam 125 mg
Each vial contains: Cefepime Hydrochloride
equal to Cefepime 1 gm
Usage: – used to treat various bacterial infections.
Category: –Anti bacterial injection
Therapeutic category: – bacterial infections
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Cefepime Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. It contains L-Arginine as a buffering agent (instead of sodium salts).
Active Ingredient Strength Therapeutic Role Cefepime Hydrochloride USP/BP Equivalent to 1000 mg (1 g) Cefepime Severe Infection / Sepsis Cefepime Hydrochloride USP/BP Equivalent to 500 mg Cefepime Pediatric / Moderate Infection Buffer L-Arginine pH Stabilizer (Approx 725 mg per g) *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefixime & Potassium Clavulanate Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Moisture-Protected Tablet, packed exclusively in high-barrier Alu-Alu blister strips to ensure the stability of the highly sensitive Clavulanate payload.
Active Ingredient Strength Primary Clinical Function Cefixime (as Trihydrate) USP/Ph.Eur. 200 mg The 3rd-Gen Payload: Broad-spectrum bactericidal activity targeting resistant Gram-negative bacilli and Enteric pathogens. Potassium Clavulanate Diluted BP/USP 125 mg The Beta-Lactamase Shield: Irreversibly inhibits the enzymes that cause antibiotic resistance. Excipients Proprietary Matrix Moisture-Scavenging Formulation: Engineered specifically utilizing Low-Humidity Processing (RH < 20%). Clavulanic acid is extremely hygroscopic; our tablets use special desiccant-compatible excipients to prevent degradation. Cefixime and Clavulanic Acid Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Moisture-Protected Tablet, packed exclusively in high-barrier Alu-Alu blister strips to ensure the stability of the highly sensitive Clavulanate payload.
Active Ingredient Strength Primary Clinical Function Cefixime (as Trihydrate) USP/Ph.Eur. 200 mg The 3rd-Gen Payload: Broad-spectrum bactericidal activity targeting resistant Gram-negative bacilli and Enteric pathogens. Potassium Clavulanate Diluted BP/USP 125 mg The Beta-Lactamase Shield: Irreversibly inhibits the enzymes that cause antibiotic resistance. Excipients Proprietary Matrix Moisture-Scavenging Formulation: Engineered specifically utilizing Low-Humidity Processing (RH < 20%). Clavulanic acid is extremely hygroscopic; our tablets use special desiccant-compatible excipients to prevent degradation. Cefixime and Linezolid Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Potency Tablet, packed in high-barrier Alu-Alu blister strips to ensure the stability of the moisture-sensitive APIs.
Active Ingredient Strength Primary Clinical Function Cefixime (as Trihydrate) USP/Ph.Eur. 200 mg The Gram-Negative Anchor: Broad-spectrum bactericidal activity against H. influenzae, E. coli, and Salmonella. Linezolid USP/Ph.Eur. 600 mg The Gram-Positive Powerhouse: Bacteriostatic activity against multi-drug resistant Staphylococci and Streptococci. Excipients Proprietary Matrix Advanced Disintegration System: Engineered utilizing Super-Disintegrants to manage the large 800mg+ active payload, ensuring the tablet shatters rapidly for maximum absorption. Cefixime and Ofloxacin Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Bilayer or Co-Processed Tablet, packed in high-barrier Alu-Alu blister strips.
Active Ingredient Strength Primary Clinical Function Cefixime (as Trihydrate) USP/Ph.Eur. 200 mg The Extracellular Specialist: Targets the bacterial cell wall; highly effective against Gram-negative enteric pathogens. Ofloxacin USP/Ph.Eur. 200 mg The Intracellular Specialist: Targets DNA replication; excellent tissue penetration into the lungs, prostate, and urinary tract. Excipients Proprietary Matrix Bilayer Technology: Engineered to prevent physical interaction between the two APIs, ensuring maximum stability and a synchronized release profile in the gut. Cefixime Capsules 400mg
FreeCefixime 400mg capsules are third-generation cephalosporin antibiotics used for a variety of bacterial infections. While 200mg is a common dose, the 400mg strength is often used for once-daily dosing.
Cefixime Dispersible Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Rapidly Disintegrating (DT) Matrix, packed in high-barrier Alu-Alu blister strips.
Active Ingredient Strength Primary Clinical Function Cefixime Trihydrate USP/Ph.Eur. 100 mg / 200 mg The Pediatric & Enteric Anchor: Dispersible format for easy administration in children and patients with dysphagia. Excipients Sweetening & Flavoring Agents Compliance Engineering: Formulated with high-grade fruit flavors and non-cariogenic sweeteners to mask the bitter antibiotic taste, ensuring 100% pediatric compliance. Disintegrants Super-Disintegrant Blend Instant-Dispersible Matrix: Engineered to shatter and form a smooth, grit-free suspension in < 45 seconds when placed in 5-10 mL of water. Cefixime Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Bioavailability Tablet, packed exclusively in high-barrier Alu-Alu blister strips to ensure the stability of the trihydrate salt.
Active Ingredient Strength Primary Clinical Function Cefixime (as Trihydrate) USP/Ph.Eur. 200 mg The Standard Primary Care Anchor: Utilized for uncomplicated UTIs, Otitis Media, and Pharyngitis. Cefixime (as Trihydrate) USP/Ph.Eur. 400 mg The Enteric & Gonorrhea Payload: High-efficacy therapeutic dose for Typhoid Fever and uncomplicated Gonorrhea. Excipients Proprietary Matrix Solubility Enhancement: Engineered utilizing Laser Micronization of the API to ensure rapid dissolution and 100% bioequivalence to the innovator brand. Cefoperazone & Sulbactam 2Gm Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in 20ml glass vials.
Active Ingredient Strength Function Cefoperazone Sodium USP/BP 1000 mg (1 g) Anti-Pseudomonal Cephalosporin Sulbactam Sodium USP/BP 1000 mg (1 g) Beta-Lactamase Inhibitor Ratio 1 : 1 High Sulbactam Load *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with 10ml Water for Injection (WFI).
Cefoperazone & Sulbactam injection
FreeComposition
C 1.5 G INJECTION (VIAL) 20 ml vial
Cefoperazone Sodium 1000 mg + Sulbactam 500 mg
Usage: – used to treat severe bacterial infections caused by organisms susceptible to cefoperazone
Category: – cephalosporin antibiotic, injection
Therapeutic category: – cephalosporin antibiotic,
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Cefoperazone & Sulbactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 1:1.
Strength Variant Cefoperazone Content Sulbactam Content Ratio 1.5 g Vial 500 mg (Sodium) 500 mg (Sodium) 1:1 (Standard) 3.0 g Vial 1000 mg (1 g) 1000 mg (1 g) 1:1 (High Dose) Excipients None (Sterile Blend) – – *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefoperazone & Tazobactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 8:1.
Strength Variant Cefoperazone Content Tazobactam Content Ratio 1.125 g Vial 1000 mg (1 g) 125 mg (Sodium) 8:1 (Standard) Excipients None (Sterile Blend) – – *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
1.125 g Vial 1000 mg (1 g) 125 mg (Sodium) 8:1 (Standard) Excipients None (Sterile Blend) – – Cefoperazone Sodium Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. It contains approximately 34 mg (1.5 mEq) of sodium per gram.
Active Ingredient Strength (Standard) Therapeutic Role Cefoperazone Sodium USP/BP Equivalent to 1000 mg (1 g) Cefoperazone Adult Surgical / Biliary Dose Excipients None (Pure Sterile Powder) – *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefopodoxime Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Bioavailability Matrix, packed in high-barrier Alu-Alu blister strips.
Active Ingredient Strength Primary Clinical Function Cefpodoxime Proxetil USP/Ph.Eur. 100 mg The Pediatric & Titration Anchor: Ideal for Pharyngitis, Tonsillitis, and uncomplicated UTIs in adolescents and adults. Cefpodoxime Proxetil USP/Ph.Eur. 200 mg The Respiratory Macrodose: High-efficacy payload for Community-Acquired Pneumonia and Sinusitis. Excipients Proprietary Matrix Solubility Enhancement: Engineered utilizing micronized API and specialized surfactants to overcome the naturally poor solubility of the Proxetil salt. Cefotaxime Sodium inection
FreeComposition
Cefotaxime Sodium 1000mg Vial+wfi
Pcd pharma franchise:-
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Cefotaxime Sodium Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The sodium content is lower than many other cephalosporins (approx 2.2 mmol/g).
Active Ingredient Strength Primary Patient Cefotaxime Sodium USP/BP Equivalent to 1000 mg (1 g) Adults (Meningitis/Sepsis) Cefotaxime Sodium USP/BP Equivalent to 500 mg Pediatrics Cefotaxime Sodium USP/BP Equivalent to 250 mg Neonates / Infants Excipients None (Pure Sterile Powder) – *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefpirome & Sulbactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 2:1.
Strength Variant Cefpirome Content (as Sulfate) Sulbactam Content (as Sodium) Ratio 1.5 g Vial 1000 mg (1 g) 500 mg 2:1 (Standard) Excipients Sodium Carbonate Buffer – *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefpirome Sulfate Injection
FreeCefpirome sulfate injection is a fourth-generation cephalosporin antibiotic typically supplied as a sterile dry powder for intravenous (IV) or intramuscular (IM) administration
Cefpodoxime & Clavulanic acid Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Moisture-Protected Tablet, packed exclusively in high-barrier Alu-Alu blister strips to ensure the stability of the highly sensitive Clavulanate payload.
Active Ingredient Strength Primary Clinical Function Cefpodoxime Proxetil USP/Ph.Eur. 200 mg The 3rd-Gen Payload: Broad-spectrum bactericidal activity targeting resistant Gram-negative and Gram-positive pathogens. Potassium Clavulanate Diluted BP/USP 125 mg The Beta-Lactamase Shield: Irreversibly inhibits the enzymes that cause antibiotic resistance. Excipients Proprietary Matrix Moisture-Scavenging Formulation: Engineered specifically utilizing Low-Humidity Processing (RH < 20%). Clavulanic acid is extremely hygroscopic; our tablets use special desiccant-compatible excipients to prevent degradation. Cefpodoxime Proxetil Dispersible Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Rapidly Disintegrating (DT) Matrix, packed in high-barrier Alu-Alu blister strips.
Active Ingredient Strength Primary Clinical Function Cefpodoxime Proxetil USP/Ph.Eur. 50 mg / 100 mg The Pediatric & Titration Anchor: Dispersible format for easy administration in children and elderly patients with swallowing difficulties. Excipients Sweetening & Flavoring Agents Bitter-Shield Technology: Engineered with molecular masking agents (e.g., Cyclodextrins) to neutralize the intense bitterness of the API, ensuring 100% pediatric compliance. Disintegrants Croscarmellose Sodium / SSG Instant-Dispersible Matrix: Engineered to shatter and form a uniform suspension in < 60 seconds when placed in 5-10 mL of water. Cefpodoxime-Potassium-Clavulanate-Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Moisture-Protected Tablet, packed exclusively in high-barrier Alu-Alu blister strips to ensure the stability of the highly sensitive Clavulanate payload.
Active Ingredient Strength Primary Clinical Function Cefpodoxime Proxetil USP/Ph.Eur. 200 mg The 3rd-Gen Payload: Broad-spectrum bactericidal activity targeting resistant Gram-negative and Gram-positive pathogens. Potassium Clavulanate Diluted BP/USP 125 mg The Beta-Lactamase Shield: Irreversibly inhibits the enzymes that cause antibiotic resistance. Excipients Proprietary Matrix Moisture-Scavenging Formulation: Engineered specifically utilizing Low-Humidity Processing (RH < 20%). Clavulanic acid is extremely hygroscopic; our tablets use special desiccant-compatible excipients to prevent degradation. Cefprozil Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Monohydrate Tablet, packed in high-barrier Alu-Alu blister strips to prevent hydrolytic degradation.
Active Ingredient Strength Primary Clinical Function Cefprozil USP/Ph.Eur. 250 mg The Pediatric & Mild Infection Anchor: Ideal for Pharyngitis, Tonsillitis, and mild skin infections. Cefprozil USP/Ph.Eur. 500 mg The Respiratory & SSTI Macrodose: Utilized for Acute Sinusitis, secondary infections of Bronchitis, and complicated skin infections. Excipients Proprietary Matrix Hydrolysis-Resistant Formulation: Engineered utilizing high-density granulation to protect the monohydrate API from environmental moisture. Ceftazidime & Sulbactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is either 2:1 or 1:1 depending on the market requirement.
Strength Variant Ceftazidime Content Sulbactam Content Ratio 1.5 g Vial 1000 mg (1 g) 500 mg (Sodium) 2:1 (Standard) 2.25 g Vial 1125 mg 1125 mg 1:1 (High Potency) Excipients Sodium Carbonate Buffer – *Pack Sizes: Tray of 1 Vial or Box of 1/10 Vials with Water for Injection (WFI).
Ceftazidime & Tazobactam Injection
FreeCeftazidime and tazobactam injection is a combination antibiotic used primarily in hospital settings for serious, multi-drug resistant bacterial infections. It is typically available as a sterile dry powder for reconstitution.
Product Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 8:1.
Strength Variant Ceftazidime Content Tazobactam Content Target Patient 1.125 g Vial 1000 mg (1 g) 125 mg (Sodium) Standard Adult Dose 281.25 mg Vial 250 mg 31.25 mg Pediatric Dose Excipients Sodium Carbonate Buffer Solubilizer Ceftazidime Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. It contains Sodium Carbonate as a solubilizer.
Active Ingredient Strength Primary Patient Ceftazidime Pentahydrate USP/BP Equivalent to 1000 mg (1 g) Adults (Standard Dose) Ceftazidime Pentahydrate USP/BP Equivalent to 250 mg Pediatrics / Neonates Excipients Sodium Carbonate Gas-Releasing Buffer *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Ceftiofur Sterile veterinary injection
FreeComposition
Ceftiofur Sodium Sterile Powder STERILE 1 GM
Usage: – used to treat bacterial skin infections, abscesses, and wounds in various animals.
Category: – veterinary injection
Therapeutic category: –veterinary medicine
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Ceftizoxime veterinary invection
FreeComposition
Ceftizoxime 2500 MG
Usage: –that is used in veterinary medicine to treat bacterial infections in animals
Category: – veterinary invection
Therapeutic category: –veterinary medicine
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Ceftriaxone & Sulbactam injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 2:1.
Strength Variant Ceftriaxone Content Sulbactam Content Target Patient 1.5 g Vial 1000 mg (1 g) 500 mg (Sodium) Adult Standard Dose 750 mg Vial 500 mg 250 mg Pediatric / Moderate Dose 375 mg Vial 250 mg 125 mg Infant / Neonate Dose Ratio 2 : 1 – Optimal Synergy *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Ceftriaxone & Sulbactam veterinary injection
FreeCompotion
Ceftriaxone & Sulbactam VET INJ. 3 GM
Ceftriaxone & Sulbactam VET INJ. 4.5 GM
Usage: – used in veterinary medicine to treat bacterial infections in animals.
Category: – veterinary injection
Therapeutic category: –veterinary medicine
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Ceftriaxone & Tazobactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 8:1.
Strength Variant Ceftriaxone Content Tazobactam Content Ratio 1.125 g Vial 1000 mg (1 g) 125 mg (Sodium) 8:1 (Standard) 562.5 mg Vial 500 mg 62.5 mg Pediatric Excipients None (Sterile Blend) – – *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Ceftriaxone 1gm Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials.
Active Ingredient Strength Standard Use Ceftriaxone Sodium USP/BP Equivalent to 1000 mg (1 g) Ceftriaxone Adult Standard Dose Sodium Content Approx 83 mg (3.6 mEq) Per 1g Vial *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Ceftriaxone 500mg Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials.
Active Ingredient Strength Target Use Ceftriaxone Sodium USP/BP Equivalent to 500 mg Ceftriaxone Pediatrics / IM Gonorrhea Sodium Content Approx 41.5 mg (1.8 mEq) Low Sodium Load *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Ceftriaxone injection
FreeComposition
Cefotaxime Sodium 1000mg Vial+wfi
Ceftriaxone 1000mg Vial+wfi
Ceftriaxone 250mg Vial+wfi
Ceftriaxone 500mg Vial+wfi
Ceftriaxone 250mg & Sulbactam 125mg Vial+wfi
Ceftriaxon 1000mg & Sulbactam 500mg Vial+wfi
Ceftriaxone 1000mg & Tazobactam 125mg Vial+wfi
Usage: – It may be used for the treatment of complicated or severe urinary tract infections.
Category: – Ceftriaxone injection
Therapeutic category: –various bacterial infections
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Ceftriaxone Sodium veterinary injection
FreeComposition
Ceftriaxone Sodium Vet INJ. 3 GM
Ceftriaxone Sodium Vet INJ. 4 GM
Usage: – used to treat various bacterial infections in animals
Category: – veterinary injection
Therapeutic category: – veterinary medicine
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Cefuroxime & Sulbactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 2:1.
Strength Variant Cefuroxime Content Sulbactam Content Primary Use 1.5 g Vial 1000 mg (1 g) 500 mg (Sodium) Adult Therapy / Surgery 1.125 g Vial 750 mg 375 mg Moderate Infection 375 mg Vial 250 mg 125 mg Pediatrics *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefuroxime Axetil Tablets
FreeProduct Composition & StrengthWe supply this product as a Precision-Blended, Amorphous Solid Dispersion Tablet, packed exclusively in secure, moisture-resistant Alu-Alu blister strips.
Active Ingredient Strength Primary Clinical Function Cefuroxime Axetil USP/Ph.Eur. 250 mg The Pediatric & ENT Anchor: Utilized for Acute Otitis Media, Pharyngitis, and uncomplicated UTIs. Cefuroxime Axetil USP/Ph.Eur. 500 mg The Respiratory Macrodose: Utilized for Community-Acquired Pneumonia, Bronchitis, and early-stage Lyme Disease. Excipients Proprietary Matrix Amorphous Solid Dispersion Technology: Engineered to maintain the API in its highly-soluble amorphous state for maximum clinical cure rates. Cefuroxime Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials.
Active Ingredient Strength Primary Use Cefuroxime Sodium USP/BP Equivalent to 750 mg Cefuroxime Standard Adult Dose Cefuroxime Sodium USP/BP Equivalent to 1500 mg (1.5 g) Severe Infections / Surgery Excipients None (Pure Sterile Powder) – *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefuroxime Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Amorphous Solid Dispersion Tablet, packed exclusively in secure, moisture-resistant Alu-Alu blister strips to ensure the stability of the hygroscopic prodrug.
Active Ingredient Strength Primary Clinical Function Cefuroxime Axetil USP/Ph.Eur.
(Equivalent to Cefuroxime)250 mg The Pediatric & ENT Anchor: The definitive unit utilized for the treatment of Acute Otitis Media, Pharyngitis, and uncomplicated Urinary Tract Infections. Cefuroxime Axetil USP/Ph.Eur. 500 mg The Respiratory Macrodose: The high-efficacy therapeutic payload required for the management of Community-Acquired Pneumonia, Bronchitis, and early-stage Lyme Disease. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Sodium Lauryl Sulfate / Colloidal Silicon Dioxide / Premium Polymeric Film Diluent / Superdisintegrant / Solubilizer / Glidant / Film-Coating (Engineered specifically utilizing Amorphous Solid Dispersion Technology. Because crystalline Cefuroxime Axetil has near-zero bioavailability, our tablets are engineered to maintain the API in its highly-soluble amorphous state for maximum clinical cure rates). *Pack Sizes: 1×6, 1×10, or 10×10 Alu-Alu Blisters (Optimized specifically for strict 5-to-10 day acute curative regimens).
Celecoxib Capsules
FreeCelecoxib capsules are selective non-steroidal anti-inflammatory drugs (NSAIDs) used to manage pain and inflammation. They are commonly available in strengths of 50 mg, 100 mg, 200 mg, and 400 mg.
Cephalexin Capsules
FreeCephalexin capsules are a first-generation cephalosporin antibiotic used to treat various bacterial infections. They are typically available in strengths of 250 mg, 500 mg, and sometimes 333 mg or 750 mg.
Cephalothin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. It contains approximately 2.8 mEq (64 mg) of Sodium per gram.
Active Ingredient Strength Therapeutic Role Cephalothin Sodium USP/BP Equivalent to 1000 mg (1 g) Cephalothin Surgical Prophylaxis / Skin Infection Excipients Sodium Bicarbonate Buffer / pH Adjuster *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cetrizine Tablets
FreeLevocetirizine Tablets IP 5 mg
Each Uncoated Tablet Contains:
Levocetirizine IP 5 mg
Excipients q.sCitrin – 10 Cetrizine Tablets 10 mg
Each Filmcoated tablet contains:
Cetrizine Dihydrochloride BP 10 mgUsage: – Relieve runny nose
Category: – Anti Allergic Drugs
Therapeutic category: – Antihistamine, Anti Allergic
Chlorambucil Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Low-Dose Cytotoxic Tablet, packed exclusively in light-shielded, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical survival of the sensitive aromatic mustard payload.
Active Ingredient Strength Primary Clinical Function Chlorambucil USP/Ph.Eur. 2 mg The Standard Hematology Anchor: The definitive unit utilized for chronic, daily maintenance and precise titration in CLL and low-grade lymphomas. Chlorambucil USP/Ph.Eur. 5 mg The Pulse-Dosing Payload: High-efficacy therapeutic payload utilized for “Pulse” therapy regimens or initial aggressive induction phases. Excipients Lactose Anhydrous / Colloidal Silicon Dioxide / Magnesium Stearate / Hypromellose / Titanium Dioxide / Premium Polymeric Film Diluent / Glidant / Lubricant / Protective Cytotoxic Shield (Engineered specifically utilizing an impenetrable film coat. This ensures the highly toxic, mutagenic API never comes into contact with the skin of the pharmacist or caregiver handling the medication). *Pack Sizes: 25-Tablet or 30-Tablet Bottles/Blisters (Optimized for oncology-specific treatment cycles).
Chloramphenicol Sodium Succinate Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in glass vials. It must be reconstituted before use.
Active Ingredient Strength Therapeutic Role Chloramphenicol Sodium Succinate USP/BP Equivalent to 1000 mg (1 g) Chloramphenicol Broad Spectrum Antibiotic Excipients None (Pure Sterile Powder) – *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Chlordiazepoxide Hydrochloride Tablets
FreeProduct Composition & Strength
We supply this product as Sugar Coated Tablets (often Green or Yellow). The robust coating is essential to mask the intensely bitter taste of the API and protect the light-sensitive drug.
Active Ingredient Strength (Standard) Therapeutic Role Chlordiazepoxide HCl USP/BP 10 mg Mild Anxiety / Elderly / Taper End Chlordiazepoxide HCl USP/BP 25 mg Severe Anxiety / Alcohol Detox Loading Excipients Q.S. Sucrose / Talc / Calcium Carbonate Sugar Coating (Light Barrier) Chlordiazepoxide Hydrochloride Tablets
FreeChlordiazepoxide Hydrochloride Tablets BP
Each Filmcoated Tablet Contains:
Chlordiazepoxide HCl BP 10 mgUsage: – relieve anxiety and to control agitation caused by alcohol withdrawal
Category: – Anti Anxiety Drugs
Therapeutic category: –
Chlorine Tablets
FreeProduct Specifications & Strength
We supply these tablets in High-Density Industrial Configurations, packed in moisture-proof, UV-stabilized HDPE drums to ensure the stability of the active chlorine content during international transit.
Chemical Grade Available Strength Primary Application Calcium Hypochlorite (Tablets) 65% Available Chlorine The Standard Industrial Grade: Primarily utilized for commercial swimming pool sanitation and general environmental disinfection. Calcium Hypochlorite (High-Purity) 70% Available Chlorine The Municipal Water Anchor: The elite-tier concentration required for large-scale potable water treatment and disaster-relief water purification kits. Physical Form 20g (Small Tabs) / 200g (Giant Tabs) Engineered specifically utilizing High-Tonnage Hydraulic Pressing to ensure the tablets do not crumble during transport, maintaining a consistent surface area for uniform dissolution. *Pack Sizes: 2kg/5kg Pails or 25kg/45kg/50kg Industrial HDPE Drums.
Chloroquine Phosphate Injection
FreeComposition
Each ml Contains : Chloroquine Phosphate I.P. …. 64.5 mg
Equivalent Chloroquine base …. 40.0 mg
Chlorobutol I.P. ………………. 0.5 % ( As Preserva9ve )
Water for Injec9on I.P. Q.S. 2 ml, 5 mlUsage: – Prevent and treat malaria
Category: – Antimalarial drugs
Therapeutic category: – Antimalarial
Pcd pharma franchise:-
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Chloroquine Phosphate Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Amber Glass Vials/Ampoules.
Active Ingredient Salt Strength Base Equivalent Chloroquine Phosphate USP/BP 64.5 mg / ml Equivalent to 40 mg Chloroquine Base Total Vial Content 30 ml Multi-dose Vial 1200 mg Base Total Excipients Chlorocresol (Preservative) Water for Injection *Pack Sizes: Tray of 10 Ampoules (5ml) or Multi-dose Vials (30ml).
Chloroquine Phosphate Tablets
FreeCHLOROQUINE PHOSPHATE TABLETS IP 100 MG
Each film coated tablet contains: – CHLOROQUINE PHOSPHATE IP 160 MG Eq. to Chloroquine base 100 MGM QUEEN – 250
Chloroquine Phosphate Tablets IP 250 mg
Each Sugarcoated Tablet Contains
Chloroquine Phosphate IP 250 mgMosquin
Chloroquine phosphate Tablets BP
Each Uncoated tablet contains :
Chloroquine Phosphate BP 160 mg
Eq. To Chloroquine Base 100 mgMqueen -250
Chloroquine Phosphate Tablets BP
Each Sugarcoated tablet contains :
Chloroquine Phosphate BP 250 mgChloroquine Tablets
FreeCHLOROQUINE PHOSPHATE TABLETS IP 100 MG Each film coated tablet contains: CHLOROQUINE PHOSPHATE IP 160 MG Eq. to Chloroquine base 100 MG
Mqueen -250
Chloroquine Phosphate Tablets BP
Each Sugarcoated tablet contains :
Chloroquine Phosphate BP 250 mgMosquin
Chloroquine phosphate Tablets BP
Each Uncoated tablet contains :
Chloroquine Phosphate BP 160 mg
Eq. To Chloroquine Base 100 mgM QUEEN – 250
Chloroquine Phosphate Tablets IP 250 mg
Each Sugarcoated Tablet ContainsChloroquine Phosphate IP 250 mg
Usage: – Prevent and treat malaria
Category: – Antimalarial drugs
Therapeutic category: – Antimalarial
Chlorpheniramine Tablets
FreeChlorpheniramine Maleate Tablets IP 4 mg
Each Uncoated tablet contains :
Chlorpheniramine Maleate IP 4 mgUsage: – Uses Hay fever, and the common cold
Category: – Anti Allergic drugs
Therapeutic category: – Antiasthmatic
Chlorpromazine and Trihexyphenidyl Tablets
FreeProduct Composition & Strength
We supply this product in Precision-Blended configurations, often customized for institutional titration, packed in secure, moisture-resistant Alu-PVC or Alu-Alu blister strips.
Active Ingredient Common Strengths Primary Clinical Function Chlorpromazine HCl USP/Ph.Eur. 25 mg / 50 mg / 100 mg The Antipsychotic Anchor: High-potency D2 blockade to control positive symptoms of psychosis and induce behavioral calm. Trihexyphenidyl HCl USP/Ph.Eur. 2 mg The Anti-Tremor Shield: Centrally acting anticholinergic required to neutralize drug-induced Parkinsonism and muscle rigidity. Excipients Lactose Monohydrate / Maize Starch / Povidone K-30 / Magnesium Stearate / Polymeric Film Coat Diluent / Binder / Disintegrant / Lubricant (Engineered specifically utilizing High-Shear Wet Granulation to ensure 100% uniformity of the 2mg Trihexyphenidyl payload alongside the larger Chlorpromazine dose, guaranteeing consistent motor protection). *Pack Sizes: 10×10 Blisters or Bulk Jars of 1000 (Optimized specifically for high-volume psychiatric hospital dispensing).
Chlorpromazine Hydrochloride Tablets
FreeChlorpromazine Hydrochloride Tablets IP 25 mg
Each Sugarcoated tablets contains :
Chlorpromazine Hydrochloride IP 25 mgCHLORPROMAZINE HYDROCHLORIDE TABLETS IP 50 MG Each sugar coated Tablet Contains:
chlorpromazine hydrochloride IP 50 mg Excipients q.s.Chlorpromazine Tablets IP 100 mg
Each Sugar Coated Tablet Contains:
Chlorpromazine Hydrochloride IP 100 mgChlorpromazine Tablets IP 200 mg
Each Sugarcoated tablet contains :
Chlorpromazine Hydrochloride IP 200 mg Excipients -QSUsage: – treat the symptoms of schizophrenia
Category: – Anti Psychotic Drugs + CNS
Therapeutic category: –Chlorthalidone Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Bioavailability Tablet, packed in moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the stability of the micronized active ingredient.
Active Ingredient Strength Primary Clinical Function Chlorthalidone USP/BP 6.25 mg / 12.5 mg The Contemporary Starting Dose: Highly preferred for modern “Low-Dose” hypertension therapy to maximize pressure control while minimizing electrolyte loss. Chlorthalidone USP/BP 25 mg The High-Potency Payload: Utilized for resistant hypertension or the management of edema associated with heart failure or hepatic cirrhosis. Excipients Microcrystalline Cellulose / Colloidal Silicon Dioxide / Magnesium Stearate / Pregelatinized Starch Diluent / Glidant / Lubricant / Binder (Engineered specifically utilizing Micronized API Technology to ensure absolute uniformity in the 6.25mg and 12.5mg ultra-low-dose tablets, guaranteeing consistent renal response across every batch). Chlorzoxazone ,Ibuprofen & Paracetamol Tablets
FreeProduct Composition & Strength
We supply this product as a High-Density, Precision-Compressed Tablet, packed exclusively in secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the stability of the large multi-API payload.
Active Ingredient Strength Primary Clinical Function Chlorzoxazone USP/BP 250 mg The Muscle Relaxant Anchor: Targets the spinal cord to inhibit the reflexes driving the muscle spasm. Ibuprofen USP/BP 400 mg The Anti-Inflammatory Payload: Blocks prostaglandin synthesis to reduce tissue swelling and peripheral pain. Paracetamol (Acetaminophen) USP/BP 325 mg The Central Analgesic Booster: Synergistically raises the pain threshold for rapid symptomatic relief. Excipients Microcrystalline Cellulose / Sodium Starch Glycolate / Povidone K-30 / Magnesium Stearate / Polymeric Film Coat Diluent / Superdisintegrant / Binder / Lubricant (Engineered specifically utilizing High-Pressure Rotary Compression to manage the massive ~1000mg tablet weight while ensuring the tablet remains swallowable and shatters instantly in gastric fluid). *Pack Sizes: 10×10 Blisters (Optimized specifically for 5-day to 10-day acute injury recovery cycles).
Cholecalciferol (Vitamin D3) Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Oily Solution in glass ampoules.
Active Ingredient Strength Vehicle Cholecalciferol USP/BP (Vitamin D3) 300,000 IU / 1 ml (7.5 mg) Oily Base (Arachis or Sesame Oil) Cholecalciferol USP/BP (Vitamin D3) 600,000 IU / 1 ml (15 mg) Oily Base (Arachis or Sesame Oil) Excipients BHT / Benzyl Alcohol Antioxidant / Preservative *Pack Sizes: Tray of 5 Ampoules, 10 Ampoules, or 50 Ampoules.
Cholecalciferol Granules Sachet
FreeCHOLECALCIFEROL GRANULES each sachet of 1 gm contains: cholecalciferol IP 60000 IU Excipients
Usage: – Dietary supplement
Category: – Vitamin Supplement
Therapeutic category: – Vitamin Supplement
Chymotrypsin Tablets
FreeCirozyme – 1 Tablets
Trypsin – Chymotrypsin Tablets
Each Enteric coated tablet contains :100000 Armour units of enzymatic activity supplied by a purified concentrate
which has specific trypsin & chymotrypsin activity in a ratio of approximately six to one
Usage: – Redness and Swelling with pockets of infection (abscesses),ulcers, surgery, or critical illness
Category: – Anti Allergic drugs
Therapeutic category: – Allergic drugs
CILNIDIPINE & TELMISARTAN TABLETS
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Bilayer or Co-Processed Tablet, packed exclusively in secure, high-barrier Alu-Alu blister strips to ensure the stability of the moisture-sensitive Telmisartan and light-sensitive Cilnidipine.
Active Ingredient Strength Primary Clinical Function Telmisartan USP/Ph.Eur. 40 mg The Hormonal Stabilizer: Blocks the RAAS system to provide 24-hour systemic pressure control and metabolic benefits (PPAR-gamma activation). Cilnidipine USP/Ph.Eur. 10 mg The Vascular Relaxant: Provides immediate N-type and L-type calcium channel blockade to lower peripheral resistance without edema. Excipients Meglumine / Sorbitol / Sodium Hydroxide / Magnesium Stearate / Titanium Dioxide (Opaque Coating) Solubilizer / Diluent / PH Modifier / Light Shield (Engineered specifically utilizing Alkaline Matrix Solubilization. Telmisartan requires a highly alkaline environment to dissolve, while Cilnidipine is light-sensitive. Our bilayer technology ensures each API is released at its peak kinetic window). Cilostazol Tablets
FreeCilostazol Tablets 50 mg
Each Uncoated Tablet Contains:
Cilostazol 50 mgCilostazol Tablets 100 mg
Each Uncoated Tablet contains :
Cilostazol 100 mgUsage: – improve the symptoms of a certain blood flow problem in the legs
Category: – Anti Hypertensive / Cardiac Drugs
Therapeutic category: – Anti Hypertensive
Cimetidine Tablets
FreeGlatidine 200
Cimetidine Tablets USP 200 mg
Each Uncoated tablet contains :
Cimetidine USP 200 mgGlatidine 400
Cimetidine Tablets USP 400 mg
Each Uncoated tablet contains :
Cimetidine USP 400 mgUsage: – Treat and prevent certain types of stomach ulcer
Category: – Antacid & Anti ulcer drugs
Therapeutic category: – Antiulcer,Antacid
CINITAPRIDE TABLETS
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Low-Dose Gastric Tablet, packed exclusively in secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the hydrogen tartrate salt.
Active Ingredient Strength Primary Clinical Function Cinitapride Hydrogen Tartrate 1 mg The Global Prokinetic Anchor: The definitive therapeutic unit utilized to accelerate gastric emptying and increase esophageal tone in patients with chronic GERD or dyspepsia. Excipients Lactose Monohydrate / Maize Starch / Povidone K-30 / Magnesium Stearate / Hypromellose / Titanium Dioxide (Film Coat) Diluent / Binder / Lubricant / Protective Barrier (Engineered specifically utilizing High-Shear Wet Granulation to ensure 100% uniformity of the microscopic 1mg payload. The premium film coat protects the tablet from atmospheric moisture and masks the bitter taste of the tartrate salt). *Pack Sizes: 10×10 Blisters (Optimized specifically for 15-day to 30-day chronic GI dispensing regimens).
Cinnarizine & Dimenhydrinate Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Immediate-Release Tablet, packed exclusively in secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the active antihistamine salts.
Active Ingredient Strength Primary Clinical Function Cinnarizine BP/USP 20 mg The Peripheral Stabilizer: Stops the influx of calcium into the inner ear hair cells to dampen the physical sensation of vertigo. Dimenhydrinate BP/USP 40 mg The Central Antiemetic: Blocks H1 and muscarinic receptors in the brain to prevent nausea and motion-induced vomiting. Excipients Microcrystalline Cellulose / Maize Starch / Talc / Magnesium Stearate / Croscarmellose Sodium Diluent / Binder / Glidant / Superdisintegrant (Engineered specifically utilizing High-Shear Dry Blending to ensure 100% uniformity of the active APIs, providing a rapid disintegration profile that delivers relief within 30 minutes of ingestion). *Pack Sizes: 10×10 Blisters (Optimized specifically for short-course acute vertigo regimens).
Cinnarizine Tablets
FreeCingin
Cinnarizine Tablets 25 mg
Each Uncoated Tablet contains :
Cinnarizine 25 mgUsage: – Travel sickness
Category: – Anti Allergic Drugs
Therapeutic category: – Anti Allergic
Ciprofloxacin Tablets
FreeCiprofloxacin Dispersible Tablets
Each Uncoated Dispersible tablet contains:
Ciprofloxacin Hydrochloride IP
Eq. Ciprofloxacin 100 mg
Approved colour & FlavourCiprofloxacin Tablets IP 250 mg
Each Film coated tablet contains:
Ciprofloxacin Hydrochloride IP
Eq.To Ciprofloxacin Anhydrous 250 mg
Color : Titanium Dioxide IPCiprofloxacin Tablets IP 500 mg
Each Filmcoated tablet contains :
Ciprofloxacin Hydrochloride IP
Eq.To Ciprofloxacin Anhydrous 500 mg
Excipients q.sCiproctan – 250 Tablets
Ciprofloxacin Tablets BP 250 mg
Each Filmcoated tablet contains :
Ciprofloxacin Hydrochloride BP
Eq.To Ciprofloxacin Anhydrous 250 mgCiproctan
Ciprofloxacin Tablets BP 500 mg
Each Filmcoated tablet contains :
Ciprofloxacin Hydrochloride BP
Eq.To Ciprofloxacin Anhydrous 500 mgUsage: – Treat or prevent certain infections caused by bacteria such as pneumonia
Category: – Antibiotic Drugs
Therapeutic category: – Antibiotic
Ciprofloxacin Tinidazole Tablets
FreeOctum – TZ
Each Filmcoated tablet contains :
Ciprofloxacin HCl BP
Eq.To Ciprofloxacin Anhydrous 500 mg
Tinidazole BP 600 mgUsage: – Bacterial & Parasitic infections
Category: – Antibiotic Drugs
Therapeutic category: – Antibiotic
Ciprofloxacin veterinary injection
FreeComposition
Ciprofloxacin veterinary INJ. 50 ML
Usage: – Treat or prevent certain infections caused by bacteria such as pneumonia
Category: – Antibiotic injection
Therapeutic category: – Antibiotic injection
Citalopram Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Bioavailability Tablet, packed exclusively in secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the chemical stability of the hydrobromide salt.
Active Ingredient Strength Primary Clinical Function Citalopram Hydrobromide USP/Ph.Eur. 10 mg / 20 mg The Primary Care Anchor: The definitive starting and maintenance doses utilized for the vast majority of patients with moderate depression or panic disorder. Citalopram Hydrobromide USP/Ph.Eur. 40 mg The Severe Refractory Payload: The maximum therapeutic dose utilized for severe depression. (Note: Doses above 40mg are strictly avoided due to cardiac risks). Excipients Lactose Monohydrate / Maize Starch / Croscarmellose Sodium / Magnesium Stearate / Hypromellose / Titanium Dioxide (Film Coat) Diluent / Binder / Superdisintegrant / Lubricant / Opaque Coating (Engineered specifically utilizing High-Shear Wet Granulation to ensure 100% uniformity of the active API, providing a smooth, fast-acting dissolution profile that matches the originator brand). *Pack Sizes: 10×10 Blisters or 28-Tablet Calendar Packs (Optimized for monthly psychiatric compliance).
Citicoline Sodium Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Glass Ampoules or Vials.
Active Ingredient Strength Volume Citicoline Sodium USP/BP 500 mg 2 ml Ampoule (250 mg/ml) Citicoline Sodium USP/BP 1000 mg (1 g) 4 ml Ampoule (250 mg/ml) Excipients Water for Injection / pH Adjusters Preservative Free (Single Use) *Pack Sizes: Tray of 5 Ampoules, 10 Ampoules, or 50 Ampoules.
Citicoline Tablets
FreeCITICOLINE -1000</p>
CITICOLINE CONTROLLED RELEASE TABLETS 1000 MG
CITICOLINE SODIUMEQ TO CITICOLINE 1000 MG
ERYTHROCYINE & BRILLIANT BLUE FCF
EXCIPIENTS QSUsage: – Help memory loss due to aging
<strong>Category: – Antiparkinson drugs
Therapeutic category: – Nootropic, CNS Drugs
Clarithromycin Tablets
FreeClarithromycin Tablets USP 500 mg
Each Filmcoated tablet contains :
Clarithromycin USP 500 mgUsage: – Chest infections
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic
Clindamycin & clotrimazole Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Non-Greasy Vaginal Tablet (V-Tab), packed exclusively in highly secure, moisture-resistant Alu-Alu or Alu-PVC blister strips, often accompanied by a specialized applicator for sterile insertion.
Active Ingredient Strength Primary Clinical Function Clindamycin Phosphate USP/Ph.Eur. 100 mg The Antibacterial Anchor: The definitive therapeutic unit utilized to eradicate anaerobic bacteria and restore the natural vaginal flora (Lactobacilli). Clotrimazole USP/Ph.Eur. 200 mg The Antifungal Payload: The massive imidazole payload required to penetrate deep into the mucosal layers and destroy recurrent yeast colonies. Excipients Adipic Acid / Sodium Bicarbonate / Lactose / Maize Starch / Magnesium Stearate Effervescent Matrix / Diluent / Disintegrant / Lubricant (Engineered specifically utilizing an Effervescent Vaginal Base. Upon contact with minimal vaginal moisture, the tablet releases microscopic amounts of CO2 to rapidly disperse the active APIs across the entire vaginal vault, ensuring no “untreated zones” remain) *Pack Sizes: 3-Tablet or 7-Tablet Strips (Optimized specifically for the strict, short-course clinical curative regimens).
Clindamycin Capsules
FreeClindamycin capsules are lincosamide antibiotics primarily used to treat serious bacterial infections, including those of the skin, lungs, and internal organs. They are most commonly formulated as Clindamycin Hydrochloride.
Clindamycin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Glass Ampoules or Vials.
Active Ingredient Strength Volume Clindamycin Phosphate USP/BP 300 mg 2 ml Ampoule (150 mg/ml) Clindamycin Phosphate USP/BP 600 mg 4 ml Ampoule (150 mg/ml) Excipients Benzyl Alcohol / EDTA Preservative / Stabilizer *Pack Sizes: Tray of 5 Ampoules, 10 Ampoules, or 50 Ampoules.
Clindipine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Light-Shielded Cardiovascular Tablet, packed exclusively in highly secure, opaque Alu-Alu blister strips to ensure the absolute chemical stability of the highly photosensitive active ingredient.
Active Ingredient Strength Primary Clinical Function Cilnidipine USP/Ph.Eur. 5 mg The Geriatric & Titration Anchor: The definitive starting dose utilized for elderly patients or those with mild essential hypertension to prevent sudden hypotensive episodes. Cilnidipine USP/Ph.Eur. 10 mg The Global Maintenance Standard: The standardized daily therapeutic payload utilized for the vast majority of adult patients requiring chronic 24-hour blood pressure control. Cilnidipine USP/Ph.Eur. 20 mg The Severe Refractory Macrodose: Utilized strictly for severe, uncontrolled hypertension where the 10mg dose has failed to achieve target blood pressure metrics. Excipients Microcrystalline Cellulose / Lactose Monohydrate / Croscarmellose Sodium / Hypromellose / Magnesium Stearate / Opaque Polymeric Film (Titanium Dioxide base) Diluent / Densifier / Superdisintegrant / Binder / Lubricant / Protective Light Shield (Engineered specifically utilizing a dense, titanium dioxide-heavy film coat. Cilnidipine is intensely sensitive to UV and visible light; this opaque coating guarantees 100% of the active molecule survives to reach the patient’s systemic circulation) *Pack Sizes: 10×10 Opaque Alu-Alu Blisters (Optimized specifically for strict 30-day chronic cardiology dispensing regimens).
Clobazam Tablets
FreeClobazam Tablet 5 mg
Each Uncoated Tablet Contains :
Clobazam IP 5 mgClobazam Tablet 10 mg
Each Uncoated Tablet Contains :
Clobazam IP 10 mgUsage: – Control seizures
Category: – Hypertensive / Cardiac drugs
Therapeutic category: – Anti convulsant , Antiepileptic
Clofazimine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Micronized Lipid-Matrix Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or heavy-gauge Alu-Alu blister strips to ensure the absolute chemical stability of the sensitive phenazine dye.
Active Ingredient Strength Primary Clinical Function Clofazimine USP/Ph.Eur. 50 mg The Standard MDT Maintenance Unit: The globally standardized daily dose utilized in the WHO Multi-Drug Therapy (MDT) regimen for Multibacillary Leprosy. Clofazimine USP/Ph.Eur. 100 mg The MDR-TB & Acute ENL Macrodose: The heavy therapeutic payload utilized for the aggressive suppression of acute ENL reactions or as a core component of the standardized short-course MDR-TB regimen. Excipients Microcrystalline Cellulose / Propylene Glycol / Gelatin / Sodium Lauryl Sulfate / Magnesium Stearate / Heavy Opaque Polymeric Film Diluent / Lipid-Solvent Matrix / Binder / Surfactant / Lubricant / Protective Light Shield (Engineered specifically utilizing Advanced API Micronization & Solid-Lipid Tableting. Because Clofazimine is practically insoluble in water, the API is micro-milled and suspended in a specialized surfactant matrix to ensure it absorbs perfectly in the gut without requiring a soft-gelatin encapsulation process) *Pack Sizes: 10×10 Blisters or Bottles of 100 (Optimized specifically for strict, continuous, multi-year infectious disease dispensing regimens).
Clomifene citrate Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Light-Shielded Fertility Tablet, packed exclusively in highly secure, opaque Alu-PVC or heavy-gauge Alu-Alu blister strips to ensure the absolute chemical stability of the photosensitive SERM.
Active Ingredient Strength Primary Clinical Function Clomifene Citrate USP/Ph.Eur. 50 mg The Standard Ovulatory Anchor: The definitive starting dose utilized for the first cycle of ovulation induction to assess ovarian response and minimize the risk of dangerous hyperstimulation. Clomifene Citrate USP/Ph.Eur. 100 mg The Refractory Macrodose: The heavy therapeutic payload utilized strictly for subsequent cycles in patients who failed to ovulate (clomifene-resistant PCOS) on the 50mg dose. Excipients Lactose Monohydrate / Maize Starch / Pregelatinized Starch / Magnesium Stearate / Opaque Polymeric Film (Titanium Dioxide base) Diluent / Binder / Disintegrant / Lubricant / Protective Light Shield (Engineered specifically utilizing a dense, titanium dioxide-heavy film coat. Clomifene degrades under direct UV/visible light; this opaque coating guarantees 100% of the active hormone modulator survives from the blister pack to the patient’s stomach) *Pack Sizes: 10-Tablet or 5-Tablet Blisters (Optimized specifically for the strict, 5-day cyclical dosing regimen utilized in reproductive endocrinology).
Clonazepam Tablets
FreeClonazepam Tablets IP 0.5 mg
Each Uncoated Tablet contains :
Clonazepam IP 0.5 mgClonazepam Tablets USP 2 MG
Each Uncoated Tablet Contains:
Clonazepam USP 2 mgUsage: – Control seizures or fits due to epilepsy, involuntary muscle spasms, panic disorder and sometimes restless legs syndrome
Category: – Anti Hypertensive / Cardiac drugs
Therapeutic category: – Anti convulsant , Antiepileptic
Clonidine Tablets
FreeClonidine Tablets BP 75 mcg
Each Uncoated tablet contains :
Clonidine Hydrochloride BP 75 mcgClonidine Tablets BP 150 mcg
Each Uncoated tablet contains :
Clonidine Hydrochloride BP 150 mcgUsage: – Treat high blood pressure (hypertension)
Category: – Anti Hypertensive / Cardiac drugs
Therapeutic category: – Antiplatelets
Clopidogrel & Aspirin Capsules
FreeClopidogrel and Aspirin capsules are a combination of two antiplatelet medications (blood thinners) used primarily to prevent heart attacks and strokes in patients with high cardiovascular risk.
Clopidogrel Tablets
FreeClopidogrel Tablets IP
Each Film Coated Tablet Contains:
Clopidogrel Bisulphate IP
eq. To Clopidogrel 75 mgClopidogrel Tablets USP 75 mg
Each Filmcoated tablet contains :
Clopidogrel Bisulphate USP
Eq. To Clopidogrel 75 mgPlaxina
Clopidogrel With Aspirin Tablets
Each Film Coated Tablet Contains:
Clopidogrel Bisulphate USP
E.Q To Clopidogrel 75 mg
Aspirin BP 75 mgUsage: – Prevents Platelets
Category: – Anti Hypertensive / Cardiac drugs
Therapeutic category: – Antiplatelets
Clopidogrel, Aspirin Tablets Clopidogrel, Aspirin Tablets
FreeA Clopidogrel + Aspirin tablet is a dual antiplatelet therapy (DAPT) used primarily to prevent serious cardiovascular events like heart attacks and strokes
Clotrimazole Vaginal Tablets
FreeClotry Clotrimazole Vaginal Tablets IP 100 mg
Each Uncoated tablet contains:
Clotrimazole IP 100 mgClotry
Clotrimazole Vaginal Tablets USP
Each Uncoated tablet contains:
Clotrimazole USP 100 mgClotry – 500
Clotrimazole Vaginal Tablets USP
Each Uncoated tablet contains :
Clotrimazole USP 500 mgUsage: – Treat yeast infections of the vagina.
Category: – AntiFungal drugs
Therapeutic category: – Anti fungal
Cloxacillin Capsules
FreeCloxacillin capsules are penicillin-type antibiotics used specifically to treat infections caused by staphylococci (staph infections) that produce an enzyme called penicillinase.
Cloxacillin 1gm Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials.
Active Ingredient Strength Primary Use Cloxacillin Sodium USP/BP Equivalent to 1000 mg (1 g) Cloxacillin Severe Infection / Surgery Sodium Content Approx 2.3 mEq (53 mg) per gram Monitor in Heart Failure *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Clozapine Tablets
FreeCirocloz Tablets
Clozapine Tablets USP 100 mg
Each Uncoated tablet contains :
Clozapine USP 100 mgUsage: – Treat certain mental/mood disorders
Category: – Antipsychotic / CNS Drugs
Therapeutic category: –
Co -Trimoxazole Tablets Tablet
FreeCo- Trimoxazole Tablets IP
Trimethoprime & Sulphamethoxazole Tablets IP
Each Uncoated tablet contains :
Trimethoprime IP 20 mg
Sulphamethoxazole IP 100 mgTrimethoprim & sulphamethoxazole tablets IP Co -trimoxazole Tablets IP
Each film coated tablets : Excipients q.s
Trimethoprim IP 80 mg
Sulphamethoxazole IP 400 mgTrimethoprim & sulphamethoxazole tablets IP
Each film coated tablets :
Trimethoprim IP 160 mg
Sulphamethoxazole IP 800 mgCo – Trimoxazole Tablets BP
Trimethoprim & Sulphamethoxazole Tablets BP
Each Uncoated Tablet contains :
Trimethoprim BP 80 mg
Sulfamethoxazole BP 400 mgTACOT – DS
Trimethoprim & Sulphamethoxazole Tablets BP
Each Uncoated tablet contains :
Trimethoprim BP 160 mg
Sulphamethoxazole BP 800 mgUsage: – Traveler’s diarrhea
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic
Colistimethate Sodium injection
FreeComposition
Each vial contains : Colistimethate Sodium IP 1MIU (IU
: International Units ) (As sterile Lyophilized Powder )
Each vial contains : Colistimethate Sodium IP 2MIU (IU
: International Units ) (As sterile Lyophilized Powder )
Pcd pharma franchise:-
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Corion
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in glass vials with a specific solvent.
Active Ingredient Strength (IU) Primary Use Chorionic Gonadotropin USP/BP 5000 IU Standard Ovulation Trigger Chorionic Gonadotropin USP/BP 10000 IU High Dose / IVF Trigger Chorionic Gonadotropin USP/BP 2000 IU Male Hypogonadism / Maintenance Solvent Sodium Chloride (Isotonic) 1 ml or 2 ml Ampoule *Pack Sizes: Tray of 1 Vial + Solvent, 5 Vials + Solvents.
Cyclophosphamide Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in glass vials.
Active Ingredient Strength Primary Use Cyclophosphamide USP/BP 200 mg Vial Low Dose / Autoimmune Cyclophosphamide USP/BP 500 mg Vial Standard Chemotherapy Cyclophosphamide USP/BP 1000 mg (1 g) Vial High Dose / Hematology Excipients Mannitol / Sodium Chloride Bulking Agent (For Cake Structure) *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials.
Cyclophosphamide Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Cytotoxic Prodrug Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips or heavy-gauge HDPE bottles to ensure the absolute chemical stability of the sensitive nitrogen mustard derivative and to physically protect caregivers from API exposure.
Active Ingredient Strength Primary Clinical Function Cyclophosphamide USP/Ph.Eur.
(as Cyclophosphamide Monohydrate)50 mg The Oncology & Autoimmune Macrodose: The definitive oral therapeutic unit utilized for chronic, daily continuous “metronomic” chemotherapy regimens and severe immunosuppressive maintenance. Excipients Lactose Monohydrate / Microcrystalline Cellulose / Starch / Magnesium Stearate / Heavy Sucrose or Polymeric Film-Coat Diluent / Densifier / Disintegrant / Lubricant / Protective Cytotoxic Shield (Engineered specifically utilizing a thick, impenetrable sugar or film coat to ensure the highly toxic API never comes into contact with the skin of the patient, pharmacist, or caregiver handling the pill) *Pack Sizes: Bottles of 50/100 or 10×10 Alu-Alu Blisters (Optimized specifically for strict chronic oncology and rheumatology dispensing regimens).
Cyclosporine tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, SMEDDS Liquid-Filled Soft Gelatin Capsule, packed exclusively in highly secure, moisture-resistant heavy-gauge Alu-Alu blister strips to ensure the absolute chemical and physical stability of the volatile microemulsion.
Active Ingredient Strength Primary Clinical Function Cyclosporine USP/Ph.Eur. (Modified) 25 mg / 50 mg The Micro-Titration Anchor: The definitive units utilized for extremely precise, weight-based dose adjustments during the critical early post-transplant phase, or for pediatric dosing. Cyclosporine USP/Ph.Eur. (Modified) 100 mg The Transplant Macrodose: The heavy therapeutic payload required to minimize the total pill burden for adult patients maintaining long-term solid organ graft survival. Excipients (SMEDDS Matrix) Polyoxyl 40 Hydrogenated Castor Oil / Polysorbate 80 / Propylene Glycol / Corn Oil Mono-di-triglycerides / Alpha-Tocopherol Microemulsion Vehicles & Surfactants / Co-Solvent / Lipophilic Carrier / Antioxidant (Engineered specifically to keep the massive lipophilic peptide fully dissolved inside the gelatin shell, instantly forming nanometer-sized droplets upon contact with stomach acid for maximum bioavailability) *Pack Sizes: 5×10 or 10×10 Alu-Alu Blisters (Optimized specifically for strict, lifelong immunosuppressive dispensing regimens).
Cyproheptadine Tablets
FreeCYPROHEPTADINE TABLETS IP 4 MG
Each uncoated tablets contains: Cyproheptadine Hydrochloride (- )
Eq to Anhydrous Cyproheptadine hclIP (4 mg)
Excipients (- QS)CYPROHEPTADINE TABLETS BP 4 MG
Each Uncoated Tablet Contains::
Cyproheptadine Hydrochloride Equivalent to Anhydrous Cyproheptadine Hydrochloride BP 4 mg
Excipients – QSCYPROHEPTADINE TABLETS BP 4 MG
Each Sugar Coated Tablet Contains:
Cyproheptadine Hydrochloride Equivalent to Anhydrous Cyproheptadine Hydrochloride BP 4 mg
Colour : Sunset yellow, Titanium Dioxide. –
Excipients – QSUsage: – Relieve the itching of allergic skin conditions, and to treat hives, including hives caused by exposure to cold temperatures and by rubbing the skin
Category: – Anti Allergic Drugs
Therapeutic category: – Antihistamine,Anti Allergic
Cytarabine Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution (Liquid) or Lyophilized Powder.
Active Ingredient Strength Format Cytarabine USP/BP 100 mg / 5 ml (20 mg/ml) Ready-to-use Liquid Cytarabine USP/BP 500 mg / 10 ml (50 mg/ml) Ready-to-use Liquid Cytarabine USP/BP 1000 mg (1 g) Vial Lyophilized Powder or Conc. Liquid Excipients Water for Injection Preservative-Free (Available for IT use) *Pack Sizes: Tray of 1 Vial, 5 Vials, or Box of 1/10 Vials.
Dabigatran Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Tartaric Acid-Cored Pellet Formulation, packed exclusively in highly specialized, moisture-blocking Alu-Alu blister strips with integrated desiccants to ensure the absolute chemical survival of the intensely hygroscopic prodrug.
Active Ingredient Strength Primary Clinical Function Dabigatran Etexilate Mesylate USP/Ph.Eur. 75 mg / 110 mg The Renal & Geriatric Anchor: The strictly mandated dose reductions utilized for elderly patients (over 80 years old), patients with moderate renal impairment, or those at an elevated risk of hemorrhagic bleeding. Dabigatran Etexilate Mesylate USP/Ph.Eur. 150 mg The Cardiovascular Macrodose: The massive, globally standardized therapeutic payload required for the aggressive, daily prevention of stroke and systemic embolism in Non-Valvular Atrial Fibrillation (NVAF). Excipients Tartaric Acid (Core) / Hypromellose / Talc / Hydroxypropyl Cellulose / Dimethicone Acidic Microenvironment Generator / Polymer Binder / Glidant / Film-Coating (Engineered specifically utilizing advanced Fluid-Bed Pellet Coating. The tartaric acid core is isolated with a polymer layer, then coated with the Dabigatran API, and sealed again. This prevents the acid from prematurely degrading the drug before it reaches the patient’s stomach) *Pack Sizes: 10×10 Specialized Alu-Alu Blisters or Heavy-Gauge HDPE Bottles with Desiccant Caps (Optimized specifically for absolute moisture defense).
Dapsone Tablets
FreeDapsone Tablets IP 50 mg
Each Uncoated tablet contains :
Dapsone IP 50 mgUsage: – Type of skin disorder (dermatitis herpetiformis)
Category: – Anti Fungal Drugs
Therapeutic category: – Anti bacterial
Darifenacin Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Hydrophilic Matrix Prolonged-Release Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips or heavy-gauge HDPE bottles to ensure the absolute physical stability of the controlled-release polymer.
Active Ingredient Strength Primary Clinical Function Darifenacin Hydrobromide USP/Ph.Eur.
(Equivalent to Darifenacin base)7.5 mg (PR) The Standard Titration & Geriatric Anchor: The definitive, highly efficacious starting and maintenance dose utilized for the vast majority of patients to control OAB symptoms with minimal side effects. Darifenacin Hydrobromide USP/Ph.Eur. 15 mg (PR) The Severe Refractory Macrodose: High-efficacy therapeutic payload utilized strictly for patients who require greater symptom relief and have demonstrated high tolerability to the 7.5 mg dose. Excipients Hypromellose (HPMC) / Dibasic Calcium Phosphate / Magnesium Stearate / Polyethylene Glycol / Titanium Dioxide / Premium Polymeric Film Hydrophilic PR Matrix / Diluent / Lubricant / Plasticizer / Opaque Film (Engineered specifically utilizing high-viscosity Hypromellose. Upon contact with gastrointestinal fluids, the tablet hydrates to form a thick, gelatinous outer layer that strictly controls the diffusion of the highly soluble hydrobromide salt over a 24-hour period) *Pack Sizes: 10×10 Alu-Alu Blisters or Bottles of 30 (Optimized specifically for strict 30-day chronic urological dispensing regimens).
Daunorubicin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in red-capped glass vials.
Active Ingredient Strength Appearance Daunorubicin Hydrochloride USP/BP Equivalent to 20 mg Daunorubicin Red-Orange Lyophilized Cake Excipients Mannitol Bulking Agent *Pack Sizes: Tray of 1 Vial. (Cytotoxic Packaging).
Deferasirox Tablets
FreeProduct Composition & Strength
We supply this product across two distinct, Precision-Blended Engineering Platforms, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute physical and chemical stability of the active pharmaceutical ingredient.
Active Ingredient Strength & Form Primary Clinical Function Deferasirox USP/Ph.Eur. 125 mg / 250 mg / 500 mg (Dispersible) The Pediatric Suspension Standard: Engineered with micro-crystalline disintegrants to rapidly dissolve in water or apple juice for precise, weight-based pediatric dosing. Deferasirox USP/Ph.Eur. 90 mg / 360 mg (Film-Coated) The High-Compliance Macrodose: The highly densified, lactose-free therapeutic payload designed to be swallowed whole, minimizing GI distress for adult patients. (Note: 360mg FCT is clinically equivalent to 500mg DT). Excipients (FCT Matrix) Microcrystalline Cellulose / Crospovidone / Povidone K-30 / Poloxamer 188 / Magnesium Stearate / Premium Polymeric Film Diluent / Superdisintegrant / Binder / Solubilizer / Lubricant / Heavy Film-Coating (Engineered specifically utilizing advanced High-Shear Wet Granulation and Poloxamer Solubilization. Because the FCT tablet is highly densified, it requires elite surfactants to guarantee instantaneous release and absorption in the gastrointestinal tract without the need for pre-dispersion) *Pack Sizes: 10×10 Alu-Alu Blisters or Heavy-Gauge HDPE Bottles of 30 (Optimized specifically for strict, 30-day chronic hematology dispensing regimens).
Deferiprone Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Macrodose Tablet, packed exclusively in highly secure, heavy-gauge HDPE bottles to ensure the absolute physical and chemical stability of the massive active pharmaceutical payload.
Active Ingredient Strength Primary Clinical Function Deferiprone USP/Ph.Eur. 500 mg The Standard Titration Unit: The baseline therapeutic dose utilized for precise, weight-based titration in pediatric and lower-weight thalassemia patients. Deferiprone USP/Ph.Eur. 1000 mg The Hematology Macrodose: The massive therapeutic payload required to minimize the total daily pill burden for adult patients suffering from severe, refractory iron overload. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Povidone K-30 / Colloidal Silicon Dioxide / Magnesium Stearate / Premium Polymeric Film Diluent / Superdisintegrant / Binder / Glidant / Heavy Film-Coating (Engineered specifically utilizing advanced High-Shear Wet Granulation. Because a 1000mg tablet is inherently massive, it must be highly densified to remain swallowable, heavily coated to mask its severe bitterness, and packed with elite disintegrants to ensure it shatters instantly in the stomach) *Pack Sizes: Heavy-Gauge HDPE Bottles of 50 or 100 (Optimized specifically for massive, multi-pill daily chronic dispensing regimens).
Deflazacort Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Steroidal Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the glucocorticoid prodrug.
Active Ingredient Strength Primary Clinical Function Deflazacort USP/Ph.Eur. 6 mg The Pediatric & Titration Standard: The definitive unit utilized for weight-based dosing in pediatric Duchenne Muscular Dystrophy, slow tapering protocols, and chronic low-dose maintenance for rheumatoid arthritis. Deflazacort USP/Ph.Eur. 30 mg The Acute Flare-Up Macrodose: High-efficacy therapeutic payload utilized for the immediate, aggressive suppression of severe allergic anaphylaxis, acute asthma exacerbations, and violent autoimmune flare-ups. Excipients Lactose Monohydrate / Microcrystalline Cellulose / Croscarmellose Sodium / Magnesium Stearate / Colloidal Silicon Dioxide Diluent / Binder / Superdisintegrant / Lubricant / Glidant (Engineered utilizing Extreme Multi-Stage Geometric Dilution to ensure the steroidal payload is flawlessly uniform, manufactured under strict OEB 4 containment protocols to prevent cross-contamination and protect facility operators) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict titration, tapering, and chronic immunological dispensing regimens).
Desmopressin Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Extreme Microdose Peptide Tablet, packed exclusively in highly secure, moisture-resistant heavy-gauge HDPE bottles or Alu-Alu blister strips to ensure the absolute chemical stability of the fragile acetate salt.
Active Ingredient Strength Primary Clinical Function Desmopressin Acetate USP/Ph.Eur.
(Equivalent to Desmopressin base)0.1 mg The Titration & Pediatric Anchor: The definitive starting dose utilized for primary nocturnal enuresis in children or baseline control of Central Diabetes Insipidus. Desmopressin Acetate USP/Ph.Eur. 0.2 mg The Maintenance Macrodose: High-efficacy therapeutic payload utilized for severe, refractory polyuria and nocturia in adults. Excipients Lactose Monohydrate / Potato Starch / Povidone K-30 / Magnesium Stearate Diluent / Disintegrant / Binder / Lubricant (Engineered specifically utilizing Strict Geometric Dilution to ensure the microscopic peptide payload is flawlessly uniform, manufactured under stringent low-moisture conditions to prevent hydrolytic degradation of the fragile peptide bonds) *Pack Sizes: Heavy-Gauge HDPE Bottles of 30/100 or 10×10 Alu-Alu Blisters (Optimized specifically for strict chronic endocrine dispensing regimens).
Dexlansoprazole & Domperidone Capsules
FreeDexlansoprazole and Domperidone capsules are a combination medication primarily used for erosive esophagitis and Gastroesophageal Reflux Disease (GERD). This combination reduces stomach acid while also speeding up gastric emptying to prevent reflux.
Diazepam Tablets
FreeDiazepam Tablets IP 5 mg
Each Uncoated tablet contains :
Diazepam IP 5 mg
Excipients q.sDiazepam Tablets BP 5 mg
Each Uncoated tablet contains :
Diazepam BP 5 mgUsage: – Anxiety, alcohol withdrawal, and seizures
Category: – Anti Anxiety Drugs
Therapeutic category: – Anti Anxiety
Diclofenac Potassium Tablets
FreeProduct Composition & Strength
We supply this product as Sugar Coated or Film Coated Tablets (typically Reddish-Brown or White). The coating is essential to mask the intensely bitter and metallic taste of the potassium salt.
Active Ingredient Strength (Standard) Therapeutic Role Diclofenac Potassium USP/BP 25 mg Pediatric (Older Children) / Mild Pain Diclofenac Potassium USP/BP 50 mg Standard Acute Adult Dose Excipients Q.S. Maize Starch / Povidone / Sucrose Disintegrant / Coating Agent Diclofenac Potassium Tablets
FreeDiclofenacPotassium Tablets 50 mg
Each Sugarcoated tablet contains:
Diclofenac Potassium BP 50 mgDiclofenac Potassium and Paracetamol Tablets
Each uncoated Tablet contains: Diclofenac Potassium BP 50 mg
Paracetamol BP 650 mg
colour: Tartrazine –
Excipient – QSUsage: – Muscle aches, backaches, dental pain, menstrual cramps, and sports injuries
Category: – Anti Analgesic Painkiller Drugs
Therapeutic category: – NSAID, Arthritis, Painkiller
Diclofenac Sodium & Serratiopeptidase Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Multi-Particulate or Bilayer Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute stability of the sensitive biological enzyme.
Active Ingredient Strength Primary Clinical Function Diclofenac Sodium USP/Ph.Eur. 50 mg Potent peripheral NSAID for sustained localized pain and inflammation blockade. Serratiopeptidase IP/Ph.Eur. 10 mg or 15 mg (Enteric-Coated) Proteolytic enzyme engineered to drain localized post-surgical/traumatic edema. Excipients Methacrylic Acid Copolymer / Microcrystalline Cellulose / Magnesium Stearate Enteric Coating Polymer for Serratiopeptidase / Diluent / Lubricant *Pack Sizes: 10×10 Alu-Alu Blisters (Optimized specifically for 7-to-14 day post-operative or orthopedic recovery regimens).
Diclofenac Sodium Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Amber Glass Ampoules. We offer both the standard 3ml volume and the advanced 1ml (AQ) technology.
Active Ingredient Strength Technology Variant Diclofenac Sodium USP/BP 75 mg / 3 ml Standard (Propylene Glycol Base) Diclofenac Sodium USP/BP 75 mg / 1 ml AQ / Dynapar Generic (Painless, Water Base) Excipients Benzyl Alcohol / Mannitol Preservative / Stabilizer *Pack Sizes: Tray of 5 Ampoules, 10 Ampoules, or 50 Ampoules.
Diclofenac Sodium injection
FreeComposition
Diclofenac Sodium 25mg & Benzyl Alcohol 4.0 v/v
Diclofenac sodium 75mg/ml (1 ml Amp)
Each ml Contains : Diclofenac Sodium I.P ………… 25 mg
Benzyl Alcohol I.P. …………….40 mg
Water for Injec9on I.P. Q.S 3 mlPcd pharma franchise:-
Diclofenac Sodium Paracetamol Serratiopeptidase Tablets
FreeA tablet containing Diclofenac Sodium + Paracetamol + Serratiopeptidase is a triple-action medication formulated to provide comprehensive relief from pain, fever, and inflammation. It is widely used for musculoskeletal disorders, post-surgical recovery, and dental pain.
Diclofenac Sodium Tablets
FreeDiclofenac Sodium Tablets 25 MG Each Filmcoated Tablet contains :
Diclofenac Sodium BP 25 mg Excipient -QSDiclofenac Sodium Tablets BP 50 mg
Each Enteric Coated tablet contains :
Diclofenac Sodium BP 50 mgDiclofenac Sodium Tablets IP 50 mg
Each Enteric coated tablet contains :
Diclofenac Sodium IP 50 mgEach Uncoated tablet contains :
Diclofenac Sodium BP 50 mg
Enteric Coated
Paracetamol BP 500 mgDiclofenac sodium tablets 50 mg
each uncoated tablet contains :
Diclofenac Sodium BP 50 mg
colour -tartrazine In House –
Excipient – In House – QSDiclofenac sodium tablets 50 mg
each uncoated tablet contains :
Diclofenac Sodium BP 50 mg
colour -tartrazine In House –
Excipient – In House – QSDiclofenac sodium tablets 50 mg
each uncoated tablet contains :
Diclofenac Sodium BP 50 mg
colour -tartrazine In House –
Excipient – In House – QSDiclofenac sodium tablets 50 mg
each uncoated tablet contains :
Diclofenac Sodium BP 50 mg
colour -tartrazine In House –
Excipient – In House – QSDiclofenac Sodium Tablets 100 mg
each film coated tablet contains: Diclofenac Sodium BP (100 mg)
colour Titanium Dioxide (In House) (- )
Excipient (In House) (- QS)Usage: – Relieve pain, reduce swelling and ease inflammation
Category: – Anti Analgesic Painkiller Drugs
Therapeutic category: – NSAID, Arthritis, Painkiller
Dicyclomine & Mefenic Acid Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Bi-Layer FDC Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or heavy-gauge Alu-Alu blister strips to ensure the absolute chemical stability of both active pharmaceutical ingredients.
Active Ingredient Strength Primary Clinical Function Dicyclomine Hydrochloride USP/Ph.Eur. 10 mg The Anticholinergic Anchor: The potent antimuscarinic payload required to rapidly force smooth muscle relaxation and dry up hyper-secretions in the gut. Mefenamic Acid USP/Ph.Eur. 250 mg The Gynecological Analgesic Payload: The dual-action fenamate NSAID required to crush radiating inflammatory pain and block uterine prostaglandin receptors. Excipients Microcrystalline Cellulose / Sodium Starch Glycolate / Povidone K-30 / Colloidal Silicon Dioxide / Magnesium Stearate / Premium Enteric-Like Polymeric Film Diluent / Superdisintegrant / Binder / Glidant / Heavy Film-Coating (Engineered specifically utilizing Bi-Layer Compression Technology to physically separate the highly reactive Dicyclomine from the bulky Mefenamic Acid, ensuring rapid, synchronized gastric dissolution without the APIs degrading each other) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict, short-term acute gynecological and GI dispensing regimens).
Dicyclomine HCL Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Amber Glass Ampoules.
Active Ingredient Strength Volume Dicyclomine Hydrochloride USP/BP 20 mg 2 ml Ampoule (10 mg/ml) Excipients Sodium Chloride / Water for Injection Isotonic Vehicle *Pack Sizes: Tray of 5 Ampoules, 10 Ampoules, or 50 Ampoules.
Dicyclomine Hydrochloride veterinary injection
FreeComposition
Dicyclomine Hydrochloride VET INJ. 30ML
Usage: – Dicyclomine can be used to treat gastrointestinal disorders in animals
Category: – veterinary injection
Therapeutic category: – veterinary medicine
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Dicyclomine Tablets
FreeDicyclomine HCL Tablets IP 10 mg
Each uncoated tablet contains :
Dicyclomine HCL IP 10 mgDicyclomine HCl Tablet IP 20 mg
Each Uncoated Tablet Contains :
Dicyclomine HCl IP 20 mgUsage: – Irritable bowel syndrome
Category: –
Therapeutic category: –
Dienogest Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Microdose Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the highly potent steroidal API.
Active Ingredient Strength Primary Clinical Function Dienogest USP/Ph.Eur. 2 mg The Global Endometriosis Standard: The definitive therapeutic micro-dose utilized globally for the continuous, long-term suppression of pelvic pain, dysmenorrhea, and dyspareunia caused by endometriosis. Excipients Lactose Monohydrate / Microcrystalline Cellulose / Crospovidone / Povidone K-30 / Magnesium Stearate / Premium Polymeric Film Diluent / Binder / Superdisintegrant / Lubricant / Heavy Film-Coating (Engineered specifically utilizing advanced multi-stage geometric dilution to ensure the 2mg steroidal payload is flawlessly uniform, finished with an opaque film-coat to seal the potent hormone safely away from factory handlers) *Pack Sizes: 14/28-Tablet Calendar Blisters (Optimized specifically for daily, uninterrupted, long-term gynecological dispensing).
Diethyl Carbamazine Tablets
FreeDiethylcarbamazine Citrate Tablets IP 50 mg
Each Uncoated tablet contains :
Diethylcarbamazine Citrate IP 50 mgDiethyl Carbamazine Citrate Tablets IP 100 mg
Each Uncoated Tablet Contains: Diethylcarbamazine Citrate Tablets 100 mg
Usage: – Certain parasitic diseases caused by infection with roundworms
Category: – Anti Hypertensive/ Cardiac drugs
Therapeutic category: – Antiepileptic, Anticonvulsant
Digoxin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Glass Ampoules.
Active Ingredient Strength Concentration Digoxin USP/BP 500 mcg (0.5 mg) 250 mcg/ml (2 ml Ampoule) Excipients Propylene Glycol / Alcohol Solubilizers (Critical for stability) *Pack Sizes: Tray of 5 Ampoules, 10 Ampoules, or 50 Ampoules.
Digoxin Tablets
FreeDigoxin Tablets IP 0.25 mg
Each Uncoated tablet contains:
Digoxin IP 0.25 mgDIGOXIN TABLETS IP 62.5 MCG Each uncoated tabelt contiains: digoxin tablets IP 62. 5 mcg excipients q.s.
Usage: – treat heart failure
Category: – Generic drugs and Medicine
Therapeutic category: – Generic Drugs
Diloxanide Furoate Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Luminal-Dispersion Macrodose Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or heavy-gauge Alu-Alu blister strips to ensure the absolute chemical stability of the anti-parasitic ester.
Active Ingredient Strength Primary Clinical Function Diloxanide Furoate USP/Ph.Eur. 500 mg The Global Anti-Parasitic Standard: The definitive therapeutic macrodose utilized globally for the total luminal eradication of amoebic cysts in asymptomatic carriers and post-dysentery patients. Excipients Microcrystalline Cellulose / Sodium Starch Glycolate / Povidone K-30 / Colloidal Silicon Dioxide / Magnesium Stearate / Premium Polymeric Film Diluent / Superdisintegrant / Binder / Glidant / Film-Coating (Engineered specifically utilizing advanced high-shear granulation and elite superdisintegrants to ensure the massive 500mg tablet shatters and disperses uniformly across the intestinal lining, maximizing physical contact with the embedded cysts) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict, 10-day acute parasitology eradication regimens).
Diltiazem Tablets
FreeDIL- 60
Diltiazem Hydrochloride Tablets IP 60 mg
Each Filmcoated tablet contains :
Diltiazem Hydrochloride IP 60 mgUsage: – Treat high blood pressure
Category: – Hypertensive/ Cardiac drugs
Therapeutic category: –
Diphenoxylate & Atropine Sulfate Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Extreme Microdose FDC Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of both the opioid and the anticholinergic alkaloid.
Active Ingredient Strength Primary Clinical Function Diphenoxylate Hydrochloride USP/Ph.Eur. 2.5 mg The Anti-Diarrheal Anchor: The potent mu-opioid agonist required to physically halt hyperactive intestinal peristalsis. Atropine Sulfate USP/Ph.Eur. 0.025 mg (25 mcg) The Abuse Deterrent (Ultra-Microdose): The anticholinergic alkaloid engineered strictly to prevent deliberate opioid overdosage. Excipients Microcrystalline Cellulose / Sucrose / Maize Starch / Magnesium Stearate / Purified Talc Diluent / Binder / Disintegrant / Lubricant / Glidant (Engineered utilizing Extreme Multi-Stage Geometric Dilution to guarantee the microscopic 25 microgram Atropine payload is flawlessly distributed throughout the tablet, preventing lethal anticholinergic “hot spots”) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict, short-term acute gastrointestinal dispensing regimens).
Diphenylhydantoin Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Dissolution-Optimized Macrodose Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the highly reactive sodium salt.
Active Ingredient Strength Primary Clinical Function Diphenylhydantoin (Phenytoin) Sodium USP/Ph.Eur. 100 mg The Global Epilepsy Standard: The definitive therapeutic unit utilized globally for the chronic, daily maintenance and suppression of tonic-clonic and psychomotor seizures. Excipients Microcrystalline Cellulose / Lactose Monohydrate / Sodium Starch Glycolate / Povidone / Magnesium Stearate Diluent / Binder / Superdisintegrant / Glidant / Lubricant (Engineered specifically utilizing Strict Particle-Size Distribution (PSD) Control. Because the drug is highly insoluble, the API is micro-milled to an exact, mathematically defined micron size to guarantee a highly predictable, extended dissolution profile in the gut, preventing toxic “dose dumping”) *Pack Sizes: Bottles of 100 or 10×10 Blisters (Optimized specifically for strict chronic neurology dispensing regimens and therapeutic drug monitoring protocols).
Disulfiram Tablets
FreeDisulfiram Tablet IP 500 MG Each uncoated tablets cotains: Diulfiram IP 500 mg Excipient -QS
Usage: – Side effects and safety, interactions
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Donepezil Tablets
FreeDonepezil Tablets IP 5 mg
Each Film Coated Contains:
Donepezil HCl IP 5 mgUsage: – Treat dementia
Category: – Pharmaceutical Tablets
Therapeutic category: –
Doripenem Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in glass vials.
Active Ingredient Strength Primary Use Doripenem Monohydrate USP/BP Equivalent to 500 mg Doripenem Standard Adult Dose Doripenem Monohydrate USP/BP Equivalent to 250 mg Renal Impairment Excipients None (Pure Sterile Powder) – *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials.
Dothiepin Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Heavy-Coated Macrodose Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure absolute chemical stability and patient safety.
Active Ingredient Strength Primary Clinical Function Dothiepin (Dosulepin) Hydrochloride USP/Ph.Eur. 25 mg The Titration & Geriatric Standard: The definitive low-dose unit utilized for careful initial titration, daytime anxiety management, or for elderly patients highly sensitive to anticholinergic side effects. Dothiepin (Dosulepin) Hydrochloride USP/Ph.Eur. 75 mg The Nighttime Maintenance Macrodose: High-efficacy therapeutic payload utilized strictly at bedtime to induce heavy sleep and drive profound, long-term monoamine receptor down-regulation. Excipients Lactose Monohydrate / Microcrystalline Cellulose / Maize Starch / Colloidal Silicon Dioxide / Magnesium Stearate / Premium Opaque Polymeric Film Diluent / Binder / Disintegrant / Glidant / Heavy Film-Coating (Engineered specifically to form an impenetrable physical barrier over the tablet core. Dothiepin hydrochloride is profoundly bitter and can cause local oral anesthesia if it dissolves in the mouth; our elite coating guarantees seamless swallowing and zero taste disruption) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict, heavily monitored 30-day psychiatric dispensing regimens).
Doxepin Hydrochloride Capsules
FreeDoxepin Hydrochloride capsules are tricyclic antidepressants (TCAs) used to treat depression, anxiety, and sleep disorders (insomnia).
Doxofylline Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Macrodose Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the xanthine derivative.
Active Ingredient Strength Primary Clinical Function Doxofylline USP/Ph.Eur. 400 mg The Global Pulmonology Standard: The definitive therapeutic macrodose utilized globally for the rapid and sustained reversal of severe bronchoconstriction in adult asthma and COPD patients. Excipients Microcrystalline Cellulose / Sodium Starch Glycolate / Povidone K-30 / Colloidal Silicon Dioxide / Magnesium Stearate / Premium Polymeric Film Diluent / Superdisintegrant / High-Shear Binder / Glidant / Film-Coating (Engineered specifically utilizing advanced rapid-burst disintegrants to ensure the massive 400mg tablet dissolves quickly in the stomach, while the heavy film-coat completely masks the intensely bitter taste characteristic of all xanthine alkaloids) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict chronic respiratory dispensing regimens).
Doxycycline 100mg Capsules
FreeDoxycycline 100mg capsules are broad-spectrum tetracycline antibiotics used for bacterial infections, malaria prophylaxis, and severe acne.
Doxycycline Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in amber glass vials.
Active Ingredient Strength Primary Use Doxycycline Hyclate USP/BP Equivalent to 100 mg Doxycycline Standard Adult Dose Excipients Ascorbic Acid / Mannitol Stabilizer / Bulking Agent *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials.
Doxycycline Tablets
FreeDoxycycline Tablets 100 mg
Each Film Coated Tablet Contains:
Doxycycline Hydrochloride IP
eq. to Doxycycline 100 mgDispersible Doxycycline Tablets BP 100 mg
Each Uncoated Tablet Contains:
Doxycycline Monohydrate BP
eq. To Anhydrous Doxycycline 100 mgUsage: – Infections
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic
Dried Aluminium Hydroxide & Magnesium Trisilicate Tablet
FreeAluminium Hydroxide & Magnesium Trisilicate Tablet NFI
Each uncoated Chewable Tablet Contains:
Dried Aluminium Hydroxide Gel IP 120 mg
Magnesium Trisilicate IP 250 mg
Peppermint Oil IP 0.003 mlALUMINUM HYDROXIDE GEL + MAGNESIUM TRISILICATE
Each Uncoated chewable tablet contains:
Magnesium Trisilicate IP 500 mg
Dried Aluminium Hydroxide gel IP 250 mgCompound Magnesium Trisilicate Tablets BP
Aluminium Hydroxide & magnesium Trisilicate tablets
Each Uncoated tablet contains :
Magnesium Trisilicate BP 250 mg
Dried Aluminium Hydroxide BP 120 mg
Peppermint Flavour
Usage: – Relieve the symptoms of indigestion, heartburn, or gastro esophageal reflex disorderCategory: – Antacid – Anti Ulcer drugs
Therapeutic category: –
Dried Aluminium Hydroxide Gel, Magnesium Hydroxide & Dimethylpolysiloxane Tablets
FreeProduct Composition & Strength
We supply this product as Chewable Tablets (typically White or Pink, Mint/Orange Flavored). The chewable format is critical for increasing the surface area for acid neutralization and gas dispersion.
Active Ingredient Strength (Standard) Therapeutic Role Dried Aluminium Hydroxide Gel USP/BP 250 mg / 400 mg Sustained Antacid Magnesium Hydroxide USP/BP 250 mg / 400 mg Rapid Antacid Activated Dimethylpolysiloxane (Simethicone) USP/BP 50 mg / 25 mg Antiflatulent (Gas Relief) Excipients Q.S. Mannitol / Sorbitol / Flavor Sweetener / Coolant Dried Aluminium Hydroxide, Magnesium Hydroxide & Simethicone Tablets
FreeProduct Composition & Strength
We supply this product as Chewable Tablets, which is the optimal delivery format to ensure the active ingredients are finely dispersed before reaching the stomach.
Active Ingredient Strength Primary Clinical Function Dried Aluminium Hydroxide Gel 250 mg / 300 mg The Sustained Buffer: Provides long-lasting acid neutralization and forms a protective coating on the stomach lining. Magnesium Hydroxide 250 mg The Rapid Neutralizer: Provides immediate relief from heartburn and balances the digestive transit time. Simethicone 25 mg / 50 mg The Defoaming Agent: Physically breaks down gas bubbles to relieve bloating and abdominal discomfort. Flavors Mint / Fruit / Orange Palatability Matrix: Engineered to be pleasant-tasting to ensure high patient compliance for OTC use. Drotaverine & Aceclofenac Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Bi-Layer Macrodose FDC Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of both active pharmaceutical ingredients.
Active Ingredient Strength Primary Clinical Function Drotaverine Hydrochloride USP/Ph.Eur. 80 mg The Spasmolytic Anchor (Forte Dose): The massive isoquinoline payload required to rapidly force smooth muscle relaxation in the ureters, biliary tract, and uterus. Aceclofenac USP/Ph.Eur. 100 mg The Anti-Inflammatory Payload: The potent COX-2 preferential NSAID required to crush the radiating inflammatory pain and tissue swelling. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Povidone K-30 / Colloidal Silicon Dioxide / Magnesium Stearate / Premium Enteric-Like Polymeric Film Diluent / Superdisintegrant / Binder / Glidant / Heavy Film-Coating (Engineered specifically utilizing Bi-Layer Compression Technology to physically separate the two highly reactive APIs, guaranteeing maximum shelf-life and ensuring rapid, simultaneous gastric dissolution without the APIs degrading each other) *Pack Sizes: 10×10 Alu-Alu Blisters (Optimized specifically for strict, short-term acute emergency dispensing regimens).
Drotaverine & Mefenamic Acid Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Bi-Layer Macrodose FDC Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or heavy-gauge Alu-Alu blister strips to ensure the absolute chemical stability of both active pharmaceutical ingredients.
Active Ingredient Strength Primary Clinical Function Drotaverine Hydrochloride USP/Ph.Eur. 80 mg The Spasmolytic Anchor (Forte Dose): The massive isoquinoline payload required to rapidly force smooth muscle relaxation in the uterus, biliary tract, and intestines. Mefenamic Acid USP/Ph.Eur. 250 mg The Gynecological Analgesic Payload: The potent, dual-action fenamate NSAID required to crush radiating inflammatory pain and block uterine prostaglandin receptors. Excipients Microcrystalline Cellulose / Sodium Starch Glycolate / Povidone K-30 / Colloidal Silicon Dioxide / Magnesium Stearate / Premium Enteric-Like Polymeric Film Diluent / Superdisintegrant / High-Shear Binder / Glidant / Heavy Film-Coating (Engineered specifically utilizing Bi-Layer Compression Technology to physically separate the two active APIs, guaranteeing maximum shelf-life and ensuring rapid gastric dissolution without the APIs chemically degrading each other) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict, short-term acute gynecological and emergency dispensing regimens).
Drotaverine Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Amber Glass Ampoules.
Active Ingredient Strength Volume Drotaverine Hydrochloride USP/BP 40 mg 2 ml Ampoule (20 mg/ml) Excipients Sodium Metabisulfite Antioxidant *Pack Sizes: Tray of 5 Ampoules, 10 Ampoules, or 50 Ampoules.
Drotaverine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Rapid-Burst Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the highly bitter hydrochloride salt.
Active Ingredient Strength Primary Clinical Function Drotaverine Hydrochloride USP/Ph.Eur. 40 mg The Standard GI/Gynecology Anchor: The definitive therapeutic unit utilized for irritable bowel syndrome (IBS), routine dysmenorrhea, and mild gastrointestinal cramping. Drotaverine Hydrochloride USP/Ph.Eur. 80 mg (Forte) The Acute Colic Macrodose: Massive therapeutic payload utilized for the immediate, aggressive suppression of agonizing renal colic (kidney stones), biliary colic (gallstones), and severe cervical spasms during labor. Excipients Lactose Monohydrate / Maize Starch / Crospovidone / Talc / Magnesium Stearate / Premium Polymeric Film Diluent / Disintegrant / Superdisintegrant / Glidant / Heavy Film-Coating (Engineered specifically utilizing advanced “Rapid-Burst” superdisintegrants to ensure the tablet shatters instantly upon contact with gastric fluid, guaranteeing hyper-fast pain relief while masking the intense, bitter taste of the API) *Pack Sizes: 10×10 Blisters (Optimized specifically for rapid-response acute ward dispensing and outpatient pain management).
Duloxetine Capsules
FreeDuloxetine capsules are delayed-release (gastro-resistant) medications used to treat depression, anxiety, and chronic pain. Because the medicine can be destroyed by stomach acid, it is formulated as acid-resistant pellets inside a hard gelatin capsule.
Duloxetine Tablets
FreeDuloxetine Tablets 20 mg
Each enteric coated tablet contains:
Duloxetine Hydrochloride
eq. To Duloxetine 20 mgUsage: – Depression and anxiety
Category: – Antidepressant drugs
Therapeutic category: – Antidepressant
Dutasteride Capsules
FreeDutasteride capsules are most commonly available as 0.5 mg soft gelatin capsules. They contain the active drug dissolved in a liquid medium to ensure proper absorption
Dydrogesterone Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the retro-steroidal API.
Active Ingredient Strength Primary Clinical Function Dydrogesterone USP/Ph.Eur. 10 mg The Global Fertility & Maternal Standard: The definitive therapeutic unit utilized globally for luteal phase support, miscarriage prevention, and chronic endometriosis management. Excipients Lactose Monohydrate / Hypromellose / Maize Starch / Colloidal Silicon Dioxide / Magnesium Stearate / Premium Polymeric Film Diluent / Binder / Disintegrant / Glidant / Heavy Film-Coating (Engineered specifically utilizing high-shear wet granulation to ensure the 10mg steroidal payload is flawlessly uniform, finished with an opaque film-coat to lock the teratogen/hormone safely away from factory handlers) *Pack Sizes: 10×10 Blisters or Calendar Blisters of 14/28 (Optimized specifically for precise tracking during the menstrual cycle and IVF protocols).
Ebastine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Rapid-Dissolution Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the piperidine derivative API.
Active Ingredient Strength Primary Clinical Function Ebastine USP/Ph.Eur. 10 mg The Standard Allergy Anchor: The definitive once-daily unit utilized for the baseline control of Seasonal and Perennial Allergic Rhinitis (Hay Fever). Ebastine USP/Ph.Eur. 20 mg The Dermatology Macrodose: High-efficacy therapeutic payload utilized strictly for severe, refractory Chronic Idiopathic Urticaria (hives) or intensely severe allergic rhinitis. Excipients Microcrystalline Cellulose / Lactose Monohydrate / Croscarmellose Sodium / Magnesium Stearate / Premium Polymeric Film Diluent / Binder / Superdisintegrant / Lubricant / Heavy Film-Coating (Engineered utilizing elite rapid-burst disintegrants to ensure the tablet dissolves instantly in gastric fluid, expediting the hepatic conversion to Carebastine for the fastest possible onset of clinical relief) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict, 30-day chronic allergy and dermatology dispensing regimens).
Elevat
FreeThe term Elevat most commonly refers to Elevat 20 Capsule, a prescription antidepressant. However, depending on the specific product name (such as “Elevat D”), it may contain entirely different active ingredients like vitamins
Enalapril Tablets
FreeEnalapril Maleate Tablets IP 2.5 mg
Each Uncoated Tablet Contains:
Enalapril Maleate IP 2.5 mgENALAPRIL MALEATE TABLETS IP 5 MG
Each uncoated Tablet Contains:
Enalapril Maleate IP 5 mg Excipients q.s.Enalapril Maleate tablets IP 10 mg
Each Uncoated Tablet contains :
Enalapril maleate IP 10 mgOELPRIL -5 ENALAPRIL TABLETS BP 5 MG EACH UNCOATED TABLET CONTAINS : ENALAPRIL MALEATE BP 5 MG
Oepril – 10
Enalapril Tablets USP
Each Uncoated tablet contains :
Enalapril Maleate USP 10 mgOepril – 20
Enalapril Maleate Tablets USP
Each Uncoated tablet contains :
Enalapril Maleate USP 20 mg.Usage: – Reduce high blood pressure and to prevent or treat heart failure
Category: – Anti Hypertensive / Cardiac Drugs
Therapeutic category: – Antihypertensive
Enoxaparin Injection
FreeEnoxaparin Sodium injection (brand name Lovenox) is a sterile, clear, colourless to pale yellow aqueous solution of a low molecular weight heparin (LMWH). It is primarily used to prevent and treat deep vein thrombosis (DVT) and pulmonary embolism.
Active IngredientStrengthTherapeutic RoleEnoxaparin Sodium USP/BP40 mg / 0.4 mlProphylaxis (DVT Prevention)Enoxaparin Sodium USP/BP60 mg / 0.6 mlTreatment (Weight Adjusted)Enoxaparin Sodium USP/BP80 mg / 0.8 mlTreatment (DVT/PE/ACS)OriginPorcine MucosaViral Safety Certified