In the pharmaceutical and healthcare sectors, Therapy is a broad term that refers to any process or treatment used to remediate a health problem, usually following a medical diagnosis.
While many people associate the word “therapy” exclusively with mental health (psychotherapy), it actually encompasses a wide range of medical and physical interventions designed to restore health or manage chronic conditions.
1. The Three Main Categories of Therapy
In a professional pharmaceutical context, therapy is generally divided into three major branches:
| Category | Definition | Examples |
| Pharmacotherapy | The use of pharmaceutical drugs to treat a disease or condition. | Antibiotics for infection, Insulin for diabetes, Chemotherapy for cancer. |
| Physical Therapy | Rehabilitative treatment focused on movement, strength, and physical function. | Physiotherapy for injury, Occupational therapy (OT), Speech therapy. |
| Psychotherapy | “Talk therapy” aimed at treating emotional, behavioral, or mental health disorders. | Cognitive Behavioral Therapy (CBT), Dialectical Behavior Therapy (DBT). |
2. Pharmacotherapy: The Pharmaceutical Focus
As a pharmacist and manufacturer, your primary focus is Pharmacotherapy. This is the treatment of disease through the administration of drugs.
Curative Therapy: Intended to cure a disease (e.g., an injectable antibiotic to clear a bacterial infection).
Palliative Therapy: Designed to relieve symptoms rather than cure the underlying disease (e.g., an ointment to reduce itching or a chronic infusion for pain management).
Supportive Therapy: Used to help other treatments work better (e.g., IV infusions to maintain hydration while a patient undergoes surgery).
3. Psychotherapy vs. Counseling
In the mental health space, “therapy” is often used interchangeably with “psychotherapy,” but there is a technical difference:
Counseling: Usually short-term and focused on a specific issue (e.g., grief, stress, or career changes).
Psychotherapy: Often long-term and focused on deep-seated psychological patterns, trauma, or chronic mental health conditions (e.g., Depression, Bipolar Disorder).
4. Why Healthy Inc. is Your Strategic Therapy Partner
In 2026, the success of any medical therapy depends on the quality of the pharmaceutical tools used to deliver it. Healthy Inc. serves as your strategic bridge to these tools.
Specialized Drug Delivery: Whether a patient’s therapy requires Lyophilized Injections for stability or PFS (Pre-filled Syringes) for ease of use, we source the exact delivery systems that make therapy more effective.
Pharmacist-Led Expertise: We provide technical vetting for Pharmacotherapy supplies. We understand the chemistry behind the therapy, ensuring that the drugs you source meet the highest purity and potency standards.
Compliance & Reliability: We ensure that the products you use for your patients’ therapies are WHO-GMP certified and supported by full CTD/ACTD dossiers, ensuring no interruption in their treatment cycle.
Showing 1–500 of 833 results
Acarbose Tablet
FreeAcarbose Tablet 25 mg
Each Film coated tablet contains :
Acarbose BP 25 mgAcarbose Tablets50 mg
Each Filmcoated tablet contains :
Acarbose 50 mgUsage :- control high blood sugar in people with type 2 diabetes
Category :- Anti Diabetic Drugs
Therapeutic category :- Anti Diabetic Drugs
Aceclofenac ,Paracetamol & Trypsin-Chymotrypsin 150000AU Tablets
FreeProduct Composition & Strength
We supply this product as a Film-Coated, Enteric-Shielded Tablet to ensure the enzymes are protected from stomach acid and absorbed in the intestine.
Active Ingredient Strength Primary Clinical Function Aceclofenac USP/BP 100 mg The Anti-Inflammatory: Reduces joint pain and physical swelling of the tissues. Paracetamol USP/BP 325 mg The Analgesic: Provides rapid relief from acute pain and manages fever. Trypsin-Chymotrypsin 150,000 AU The Enzyme Payload: Resolves inflammation and clears edema to accelerate tissue repair. Aceclofenac , Paracetamol and Serratiopeptidase Tablets
FreeProduct Composition & Strength
We supply this product as a Film-Coated, Enteric-Shielded Tablet to ensure the enzyme remains active through the stomach acid.
Active Ingredient Strength Primary Clinical Function Aceclofenac USP/BP 100 mg The Anti-Inflammatory: Reduces joint and tissue inflammation and stiffness. Paracetamol USP/BP 325 mg The Analgesic: Provides rapid relief from acute pain and manages fever. Serratiopeptidase 15 mg The Enzyme Payload: Resolves inflammation and clears edema to accelerate tissue repair. Aceclofenac & Paracetamol Tablets
FreeProduct Composition & Strength
We supply this product as a Film-Coated Tablet, engineered for rapid disintegration and synchronized absorption.
Active Ingredient Strength Primary Clinical Function Aceclofenac USP/BP 100 mg The Anti-Inflammatory: Targets tissue inflammation and reduces joint stiffness. Paracetamol USP/BP 325 mg / 500 mg The Analgesic: Provides rapid relief from acute pain and manages fever. Excipients Proprietary Matrix Rapid-Release System: Engineered to ensure both APIs reach therapeutic levels within 30–60 minutes. Aceclofenac & Thiocolchicoside Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet, packed in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute stability of the semi-synthetic Thiocolchicoside component.
Active Ingredient Strength Primary Clinical Use Aceclofenac BP/Ph.Eur.
Thiocolchicoside INN/Ph.Eur.100 mg
4 mgStandard Orthopedic Dose: Twice-daily therapy for moderate-to-severe acute back pain and torticollis. Aceclofenac BP/Ph.Eur.
Thiocolchicoside INN/Ph.Eur.100 mg
8 mgSevere Trauma Dose: Maximum therapy for severe, immobilizing orthopedic spasms (strictly twice daily). Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Magnesium Stearate Diluent / Superdisintegrant (Engineered for immediate gastric dissolution and rapid onset of relief) Aceclofenac Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Amber Glass Ampoules. We utilize advanced solubilizing agents to ensure the drug remains stable in liquid form.
Active Ingredient Strength (Standard) Therapeutic Role Aceclofenac BP/IP 150 mg / ml Analgesic / Anti-inflammatory Excipients Q.S. Benzyl Alcohol / Propylene Glycol Preservative / Co-solvent *Pack Sizes: Tray of 5 Ampoules (1ml each) or 10 Ampoules.
Aceclofenac Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets (Standard) or Sustained Release (SR) Tablets.
Active Ingredient Strength Release Type Role Aceclofenac IP/BP/USP 100 mg Immediate Release Standard Adult Dose Aceclofenac IP/BP/USP 200 mg Sustained Release (SR) Once Daily Dosing Excipients Q.S. — Disintegrant / Binder Acetazolamide Tablet
FreeAcetazolamide Tablets IP 250 mg
Each uncoated tablet Contains : Acetazolamide IP 250 mg
Excipients q.sAcetazolamide Tablets BP 250 mg
Each uncoated tablet Contains : Acetazolamide BP 250 mg
Excipients q.sUsage :- Prevent and reduce the symptoms of altitude sickness
Category :- Diuretic drugs
Therapeutic category :- Diuretic drugs
Adrenaline Injections
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Amber Glass Ampoules. The formulation includes stabilizers to prevent rapid oxidation.
Active Ingredient Strength (Standard 1:1000) Therapeutic Role Adrenaline Bitartrate BP/USP Eq. to Adrenaline 1 mg / ml Vasopressor / Bronchodilator Excipients Q.S. Sodium Metabisulfite / Sodium Chloride Antioxidant / Isotonicity *Pack Sizes: Tray of 10 Ampoules (1ml each) or Box of 50/100 Ampoules (Hospital Pack).
Adrenochrome Monosemicarbazone Tablet
FreeAdrenochrome Monosemicarbazone compound Tablets
each uncoated tablet contains:
Adronochrome Monosemi Carbazone (In House) (0.33 mg)
Menadione Sodium Bisulphite IP (10 mg)
Sodium Ascorbate IP eq. To Ascorbic Acid IP (100 mg)
Rutin NF (50 mg)
Dibasic Calium Phosphate IP (125 mg)
excipient (In House) (- QS)Albendazole Tablet
FreeADZOLE 200
Albendazole Tablets
Each Uncoated tablet Contains:
Albendazole USP 200 mg
Excipients q.sAlbendazole Tablet
s IP 400 mg
Each Uncoated tablet Contains:
Albendazole IP 400 mg
Colour : Sunset YellowUltiworm
Albendazole Tablets 400 mg
Each Uncoated chewable tablet contains:
Albendazole BP 400 mgZENBEST – 400 / ALBENDAZOLE TABLETS 400 MG Each Uncoated Tablets Contains: Albendazole BP 400 mg Excipients – QS
Albendazole Tablets IP 400 mg
Each Uncoated Chewable Tablet Contains: Albendazole IP 400 mg
Sunset yellow –
Excipients – QS
orange –
Usage :- certain infections caused by worms such as pork tapeworm and dog tapewormCategory :- Anthelmintic drugs
Therapeutic category :- Antiparasitic Anthelmintic,Antiprotozoal,
Alendronate Tablet
FreeAlenfos
Alendronate Tablets 70 mg
Each tablet contains :
Alendronate Sodium
Eq. To Alendronate Sodium anhydrous 70 mgAlfacalcidol Softgel Capsules
FreeProduct Composition & Strength
We supply this product as a Liquid-Filled Soft Gelatin Capsule, packed in highly protective, opaque blister strips to prevent the extreme light-degradation associated with vitamin D analogues.
Active Ingredient Strength Primary Clinical Use Alfacalcidol BP/EP 0.25 mcg (Micrograms) Initial Titration / Pediatric / Maintenance Dosing Alfacalcidol BP/EP 0.5 mcg (Micrograms) Standard Adult Dialysis / Osteoporosis Dosing Alfacalcidol BP/EP 1.0 mcg (Micrograms) Severe Hypocalcemia / High-PTH Suppression Excipients Refined Arachis (Peanut) Oil / BHA Lipid Carrier System / Antioxidant Stabilizer *Pack Sizes: 10×10 Opaque Alu-PVC Blisters or Alu-Alu Blisters. (Opaque packaging is an absolute necessity to maintain microgram API stability).
Allopurinol Tablet
FreeAlloric – 100 Tablets
Allopurinol Tablets IP 100 mg
Each Uncoated tablet contains :
Allopurinol IP 100 mgAllopurinol Tablets BP 300 mg
Each uncoated tablet contains :
Allopurinol BP 300 mgAllylestrenol Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Bio-Stable Tablet, packed in high-barrier Alu-Alu blister strips to ensure hormonal stability.
Active Ingredient Strength Primary Clinical Function Allylestrenol USP/BP/Ph.Eur. 5 mg The Standard OB-GYN Anchor: The globally validated dose for maintaining the corpus luteum and supporting the early placenta. Excipients Proprietary Matrix Bio-Synchronized System: Engineered to ensure rapid oral absorption and high plasma concentration to provide immediate uterine stability. Altretamine Capsules
FreeAltretamine capsules are antineoplastic (chemotherapy) medications used primarily for the palliative treatment of advanced ovarian cancer. They are most commonly available in a 50 mg strength.
Aluminium hydroxide Magnesium Trisilicate Tablet
FreeCompound Magnesium Trisilicate Tablets BP
Aluminium Hydroxide & magnesium Trisilicate tablets
Each Uncoated tablet contains :
Magnesium Trisilicate BP 250 mg
Dried Aluminium Hydroxide BP 120 mg
Peppermint FlavourDRIED ALUMINIUM HYDROXIDE GE+MAGNESIUM TRISILICATE TABLETS
Each uncoated chewable tablet contains: Magnesium Trisilicate IP 500 mg
Dried Aluminium Hydroxide Gel IP 250 mg
Colour: Sunset Yellow –
Excipients – QSAluminium Hydroxide & Magnesium Trisilicate Tablet NFI
Each uncoated Chewable Tablet Contains:
Dried Aluminium Hydroxide Gel IP 120 mg
Magnesium Trisilicate IP 250 mg
Peppermint Oil IP 0.003 mlALUMINUM HYDROXIDE GEL + MAGNESIUM TRISILICATE
Each Uncoated chewable tablet contains:
Magnesium Trisilicate IP 500 mg
Dried Aluminium Hydroxide gel IP 250 mgDRIED ALUMINIUM HYDROXIDE GE + MAGNESIUM TRISILICATE TABLETS Each uncoated chewable tablet contains: Magnesium Trisilicate IP 500 mg
Dried Aluminium Hydroxide Gel IP 250 mg
Colour: Sunset Yellow –
Excipients – QS
Usage: – Relieve the symptoms of indigestion, heartburn, or gastro esophageal reflux disorderCategory: – Antacid – Anti Ulcer drugs
Therapeutic category: – Antacid
Amikacin Sulphate Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Liquid in Glass Vials or Ampoules. The solution is clear, colorless to pale yellow, and requires no reconstitution (ready-to-use).
ant Active Ingredient Strength (Standard) Therapeutic Role Amikacin Sulphate USP/BP 100 mg / 2 ml Pediatric Dose Amikacin Sulphate USP/BP 250 mg / 2 ml Intermediate Dose Amikacin Sulphate USP/BP 500 mg / 2 ml Adult Standard Dose Excipients Q.S. Sodium Citrate / Sodium Metabisulfite Buffer / Antioxid *Pack Sizes: Tray of 10 Ampoules or Box of 1/10 Vials.
Amiodarone Tablet
FreeAmiodarone Tablets IP 100 mg
Each Uncoated Tablet contains:
Amiodarone Hydrochloride IP 100 mg
AMODAR – 200
Amiodarone Tablets IP 200 mg
Each Uncoated Tablet contains:
Amiodaron Hydrochloride IP 200 mg
Amodar – 200
Amiodarone Tablets BP 200 mg
Each Uncoated tablet contains:
Amiodarone Hydrochloride BP 200 mg
Usage: – Restore normal heart rhythm and maintain a regular, steady heartbeat.
Category: – Antiarrhytmics drugs
Therapeutic category: – Antiarrhythmics
Amitriptyline & Chlordiazepoxide Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Film-Coated Tablet, packed in high-barrier Alu-Alu blister strips to ensure the stability of the benzodiazepine component.
Active Ingredient Strength Primary Clinical Function Amitriptyline (as HCl) USP/BP 12.5 mg / 25 mg The Antidepressant Anchor: Elevates mood and energy levels by modulating neurotransmitter reuptake. Chlordiazepoxide USP/BP 5 mg / 10 mg The Anxiolytic Shield: Provides rapid relief from nervousness, agitation, and muscle tension. Excipients Proprietary Matrix Bio-Synchronized System: Engineered to ensure the rapid onset of the anxiolytic effect while the antidepressant effect builds over time. Amitriptyline Hydrochloride Tablet
FreeAmitriptyline Hydrochloride Tablets IP 10 mg
Each suger coated tablets contains: Amitriptyline Hydrochloride IP 10 mg
– Excipients –Amitriptyline Hydrochloride Tablets USP 10 mg
Each Sugarcoated tablet contains :
Amitriptyline Hydrochloride USP 10 mgAmitriptyline Hydrochloride Tablets IP 25 mg
Each Sugarcoated tablet contains :
Amitriptyline Hydrochloride IP 25 mgAmitriptyline Tablets BP 25 mg
Each Sugarcoated tablet contains:
Amitriptyline Hydrochloride BP 25 mgAmitriptyline Hydrochloride Tablets IP 75 mg
Each Sugarcoated tablet contains :
Amitriptyline Hydrochloride IP 75 mgUsage: – treat mental/mood problems such as depression
Category: – Anti psychotic + CNS Drugs
Therapeutic category: – Anti psychotic,Antidepressant,CNS Drugs,Anti Anxiety
Amlodipine & Enalapril Maleate Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets to protect the Enalapril Maleate from moisture-induced degradation and to ensure the physical integrity of the combination.
Active Ingredient Strength (Standard) Therapeutic Role Amlodipine (as Besylate) BP/USP 5 mg / 10 mg Calcium Channel Blocker (CCB) Enalapril Maleate BP/USP 5 mg / 10 mg / 20 mg ACE Inhibitor Excipients Q.S. Sodium Stearyl Fumarate / Lactose Lubricant & Stabilizer Amlodipine & Hydrochlorothiazide Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Film-Coated Tablet, engineered for synchronized 24-hour hemodynamic control.
Active Ingredient Strength Primary Clinical Function Amlodipine (as Besylate) USP/BP 5 mg / 10 mg The Vasodilator: Reduces the resistance of blood vessels to lower systemic pressure. Hydrochlorothiazide USP/BP 12.5 mg / 25 mg The Volume Reducer: Excretes excess sodium and water to reduce circulatory load. Excipients Proprietary Matrix Steady-State System: Engineered to ensure consistent plasma concentrations of both drugs to prevent “dips” in BP control. Amlodipine Besylate & Losartan Potassium Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets. The formulation is optimized for 24-hour efficacy, allowing for convenient once-daily dosing.
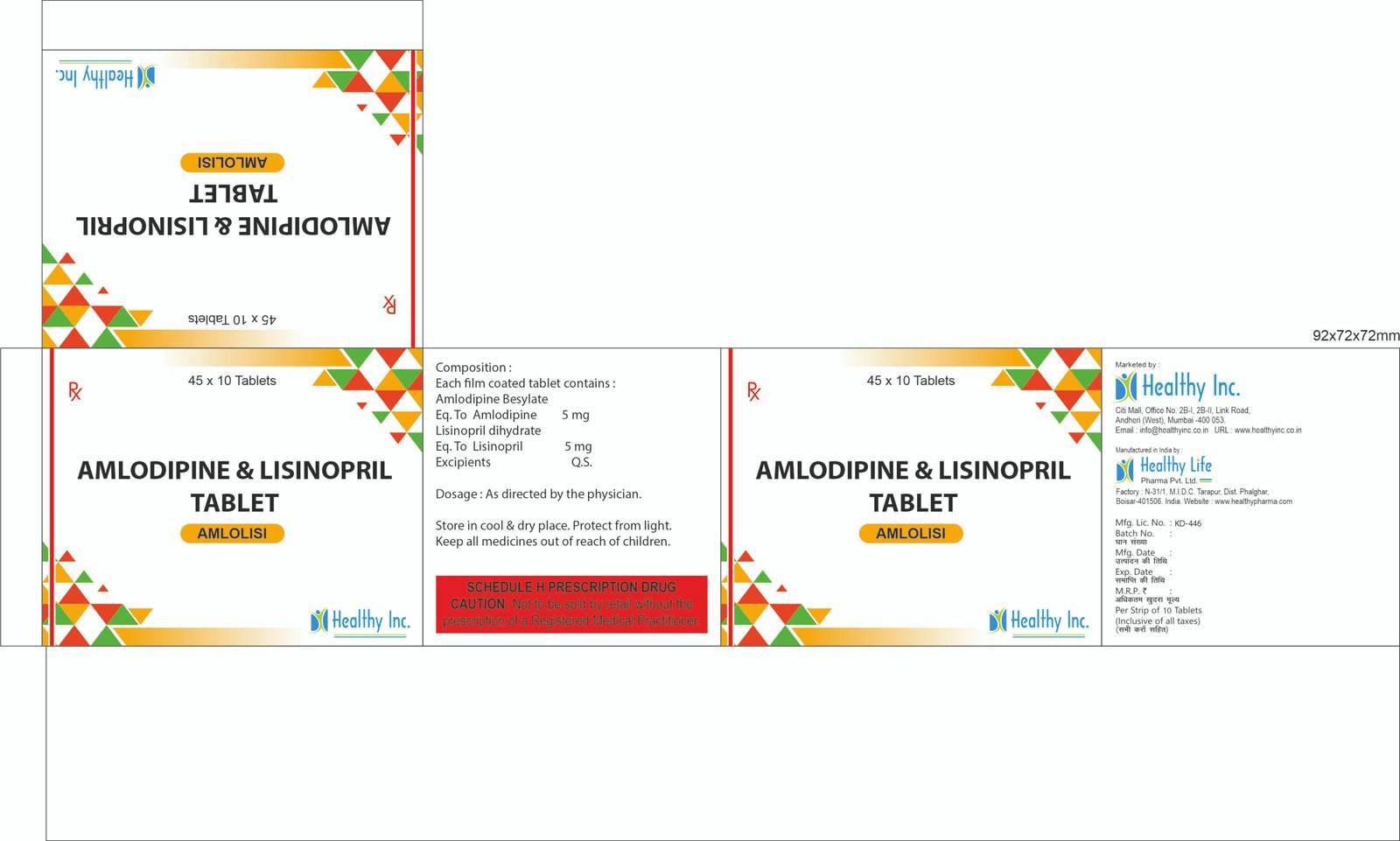
Active Ingredient Strength (Standard) Therapeutic Role Amlodipine Besylate BP/USP 5 mg Calcium Channel Blocker (CCB) Losartan Potassium BP/USP 50 mg Angiotensin Receptor Blocker (ARB) Excipients Q.S. Microcrystalline Cellulose / Starch Stabilizing Core Matrix Amlodipine With Lisinopril Dehydrate Tablets
FreeProduct Composition & Strength
We supply this product as Uncoated or Film Coated Tablets. The use of Lisinopril in its Dihydrate form ensures superior stability and predictable dissolution profiles.
Active Ingredient Strength (Standard) Therapeutic Role Amlodipine (as Besylate) BP/USP 5 mg / 10 mg Calcium Channel Blocker Lisinopril (as Dihydrate) BP/USP 5 mg / 10 mg / 20 mg ACE Inhibitor Excipients Q.S. Calcium Phosphate / Mannitol Stabilizing Agents Amodiaquine Tablets
FreeAmodiaquine Hydrochloride Tablets USP 100 mg
Each Uncoated Tablet Contains:
Amodiaquine Hydrochloride USP
eq. To Amodiaquine 100 mgAmodiaquine Hydrochloride Tablets USP 200 mg
Each Uncoated Tablet contains
Amodiaquine Hydrochloride USP
Eq. To Amodiaquine 200 mgAmodiaquine Hydrochloride Tablets USP 600 mg
Each Uncoated Tablet Contains:
Amodiaquine Hydrochloride USP
eq. To Amodiaquine 600 mgUsage: – Treat to malaria
Category: – Antimalarial drugs
Therapeutic category: – Antimalarial,
Amoxapine Tablets
FreeProduct Composition & Strength
We supply this product in a full range of strengths to allow for precise clinical titration, packed in high-barrier Alu-Alu blister strips.
Active Ingredient Strength Primary Clinical Function Amoxapine USP/BP 25 mg / 50 mg The Titration Unit: Used for gradual dose escalation to minimize initial sedative effects. Amoxapine USP/BP 100 mg The Standard Anchor: The primary maintenance dose for outpatients with moderate depression. Amoxapine USP/BP 150 mg The High-Potency Payload: For hospitalized patients with severe psychotic or endogenous depression. Amoxicillin & Potassium Clavulanate Dispersible Tablets
FreeProduct Composition & Strength
We supply this product in the globally standardized 7:1 ratio, packed in high-barrier Alu-Alu blister strips to ensure the stability of the moisture-sensitive Clavulanate.
Active Ingredient Strength Primary Clinical Function Amoxicillin (as Trihydrate) USP/BP 200 mg / 400 mg The Growth Inhibitor: Destroys the bacterial cell wall across a broad range of pathogens. Potassium Clavulanate USP/BP 28.5 mg / 57 mg The Resistance Shield: Neutralizes bacterial enzymes, allowing Amoxicillin to function. Excipients Sweetened Matrix Pediatric-Friendly System: Engineered with pleasant fruit flavors to ensure 100% child compliance. Amoxicillin & Potassium Clavulanate Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet, packed exclusively in highly secure, ultra-low moisture Alu-Alu blister strips (often containing integrated desiccants) to ensure the absolute stability of the incredibly hygroscopic Clavulanate salt.
Active Ingredient Strength Primary Clinical Function Amoxicillin Trihydrate USP/Ph.Eur.
Potassium Clavulanate USP/Ph.Eur.500 mg
125 mg
(Total: 625 mg)Standard Adult Dose: Therapy for moderate RTIs, dental infections, and skin infections. Amoxicillin Trihydrate USP/Ph.Eur.
Potassium Clavulanate USP/Ph.Eur.875 mg
125 mg
(Total: 1000 mg)High-Dose Adult Therapy: Aggressive intervention for severe pneumonia, severe sinus infections, and bite wounds. Excipients Microcrystalline Cellulose / Sodium Starch Glycolate / Colloidal Silicon Dioxide / Magnesium Stearate Diluent / Superdisintegrant / Moisture Scavenger (Engineered for immediate gastric dissolution) *Pack Sizes: 1×6 or 1×10 Alu-Alu Blisters (Optimized specifically for strict 5-to-7 day acute antibiotic regimens).
Amoxicillin Capsules
FreeAmoxicillin capsules are common penicillin-type antibiotics typically available in 250 mg and 500 mg strengths.
Amoxicillin Sodium Injection 250mg
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. It must be reconstituted with Sterile Water for Injection prior to use.
Active Ingredient Strength (Pediatric Dose) Therapeutic Role Amoxicillin Sodium USP/BP Equivalent to 250 mg Amoxicillin Broad Spectrum Antibiotic Excipients None (Pure Sterile Powder) – – *Pack Sizes: Tray of 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Amoxicillin Sodium Injection 500mg
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. It must be reconstituted with Sterile Water for Injection prior to use.
Active Ingredient Strength (Standard Adult Dose) Therapeutic Role Amoxicillin Sodium USP/BP Equivalent to 500 mg Amoxicillin Broad Spectrum Antibiotic Excipients None (Pure Sterile Powder) – – *Pack Sizes: Tray of 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Amoxicillin Trihydrate Dispersible Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Fruit-Flavored Tablet, packed in high-barrier Alu-Alu blister strips to ensure the stability of the moisture-sensitive API.
Active Ingredient Strength Primary Clinical Function Amoxicillin (as Trihydrate) USP/BP 125 mg The Pediatric Starter: Ideal for infants and toddlers requiring low-dose respiratory or ear infection therapy. Amoxicillin (as Trihydrate) USP/BP 250 mg The Junior Anchor: The standard dose for older children and adolescents for broad-spectrum anti-infective coverage. Excipients Sweetened Matrix Taste-Masking System: Engineered with pleasant flavors (Strawberry/Pineapple/Orange) to overcome the natural bitterness of the antibiotic. Amoxycillin , Cloxacillin And Lactic Acid Bacillus Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Film-Coated Tablet or Capsule, packed in high-barrier Alu-Alu blister strips to ensure the stability of the live probiotic spores.
Active Ingredient Strength Primary Clinical Function Amoxycillin (as Trihydrate) USP/BP 250 mg The Growth Inhibitor: Destroys the bacterial cell wall across a broad range of pathogens. Cloxacillin (as Sodium) USP/BP 250 mg The Defense Breaker: Specifically neutralizes bacterial enzymes, making the treatment effective against “Staph” infections. Lactic Acid Bacillus (LAB) 1.5 Billion Spores The Biological Shield: Replenishes healthy gut bacteria to prevent diarrhea and boost immunity. Amoxycillin & Cloxacillin Capsules
FreeAmoxycillin and cloxacillin capsules (commonly known as co-amoxiclox) are combination antibiotics used to treat infections where bacteria might produce enzymes that resist standard penicillin. They are typically available in a 500 mg total strength (250 mg / 250 mg)
Amoxycillin & Dicloxacillin Capsules
FreeAmoxycillin and dicloxacillin capsules are combination antibiotics designed to broaden the spectrum of treatment while protecting the medicine from bacterial resistance. They are typically available in a 500 mg total strength (often 250 mg / 250 mg)
Amoxycillin & Potassium Clavulanate Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The ratio is critically balanced (usually 5:1) for optimal pharmacokinetics in intravenous use.
Strength Variant Amoxicillin Content (as Sodium) Clavulanic Acid Content (as Potassium) Target Patient 1.2 g Vial 1000 mg (1 g) 200 mg Adults (Severe) 600 mg Vial 500 mg 100 mg Adults / Children 300 mg Vial 250 mg 50 mg Pediatric 150 mg Vial 125 mg 25 mg Infants *Pack Sizes: Tray of 1 Vial or Box of 1/10 Vials with Water for Injection (WFI).
Amoxycillin Capsules
FreeAmoxycillin capsules are widely used penicillin-type antibiotics. They are most frequently prescribed in 250 mg and 500 mg strengths.Amoxycillin Dispersible Kid Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Fruit-Flavored Tablet, packed in high-barrier Alu-Alu blister strips to ensure the stability of the moisture-sensitive API.
Active Ingredient Strength Primary Clinical Function Amoxycillin (as Trihydrate) USP/BP 125 mg The Pediatric Starter: Ideal for infants and toddlers requiring low-dose respiratory or ear infection therapy. Amoxycillin (as Trihydrate) USP/BP 250 mg The Junior Anchor: The standard dose for older children and adolescents for broad-spectrum anti-infective coverage. Excipients Sweetened Matrix Taste-Masking System: Engineered with pleasant flavors (Strawberry/Pineapple/Orange) to overcome the natural bitterness of the antibiotic. Ampicillin & Cloxacillin Capsules
FreeAmpicillin and cloxacillin capsules (often called co-ampiclox) are combination antibiotics used to treat infections where the bacteria may produce enzymes that destroy standard penicillin. They are most commonly available in a 500 mg total strength (250 mg / 250 mg)
Ampicillin & Cloxacillin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. It must be reconstituted with Sterile Water for Injection prior to use.
Active Ingredient Strength (Standard 1:1 Ratio) Therapeutic Role Ampicillin Sodium USP/BP 250 mg Broad Spectrum Coverage Cloxacillin Sodium USP/BP 250 mg Anti-Staphylococcal Shield Total Vial Strength 500 mg Pediatric / Mild Infection (Adult Variant) 500 mg Ampicillin + 500 mg Cloxacillin (1 g Total) Standard Adult Dose Excipients None (Pure Sterile Powder) – – *Pack Sizes: Tray of 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Ampicillin & Dicloxacillin Capsules
FreeAmpicillin and dicloxacillin capsules are combination antibiotics designed to provide broad-spectrum coverage while resisting degradation by certain bacterial enzymes. They are commonly available in a 500 mg total strength (typically 250 mg / 250 mg).
Ampicillin and Flucloxacillin Capsules
FreeAmpicillin and flucloxacillin capsules (often referred to by the co-drug name co-fluampicil) are combination antibiotics used to treat infections where the specific bacteria is unknown or suspected to be a penicillin-resistant strain. They are typically available in a 250 mg/250 mg strength.
Ampicillin Capsules
FreeAmpicillin capsules are a penicillin-type antibiotic used to treat various bacterial infections. They are most commonly available in 250 mg and 500 mg strengths.
Ampicillin Injection 500mg
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. It must be reconstituted with Sterile Water for Injection prior to use.
Active Ingredient Strength (Standard Pediatric/Mild Dose) Therapeutic Role Ampicillin Sodium USP/BP Equivalent to 500 mg Ampicillin Broad Spectrum Antibiotic Excipients None (Pure Sterile Powder) – – *Pack Sizes: Tray of 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Ampicillin Sulbactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The 2:1 ratio is clinically optimized for pharmacokinetics.
Active Ingredient Strength (Standard 1.5g Vial) Strength (High Dose 3g Vial) Ampicillin Sodium USP/BP 1000 mg (1 g) 2000 mg (2 g) Sulbactam Sodium USP/BP 500 mg (0.5 g) 1000 mg (1 g) Total Vial Content 1.5 g 3.0 g Excipients None (Pure Sterile Powder) None (Pure Sterile Powder) *Pack Sizes: Tray of 10 Vials, or Box of 1 Vial with Water for Injection (WFI).
Arteether Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Oily Solution in Amber Glass Ampoules. The oily base (refined Arachis Oil or Ethyl Oleate) ensures depot formation in the muscle.
Active Ingredient Strength (Standard) Therapeutic Role α-β Arteether 150 mg / 2 ml Adult Dose (Standard) α-β Arteether 75 mg / 1 ml Pediatric Dose Vehicle Q.S. Refined Arachis Oil / Ethyl Oleate Oily Base (Sustained Release) *Pack Sizes: Tray of 3 Ampoules (Full Course) or Box of 10 Ampoules.
Artemether & Lumefantrine Tablets
FreeArtmefarin – ADULT
Artemether & Lumefantrine tablets
Each Uncoated tablet contains:
Artemether 80mg
Lumefantrine 480 mg
Excipients q.sArtmefarin – Disp
Artemether & Lumefantrine tablets
Each Uncoated Dispersible tablet contains:
Artemether 20 mg
Lumefantrine 120 mg
Excipients q.sArtmefarin – DS
Artemether & Lumefantrine Tablets
Each Film Tablet contains:
Artemether 40mg
Lumefantrine 240 mg
Excipients q.s
Approved Colour UsedARTMEFARIN / ARETEMETHER & LUMEFANTRINE TABLETS
Each uncoated tablet: – ARETEMETHER 20 mg
– LUMEFANTRINE 120 mgArtemether & Lumefantrine Tablets
Each Uncoated tablet contains:
Artemether 80mg
Lumefantrine 480 mg
Excipients q.sartemether & lumefantrine tablets 20 / 120 mg
Each Uncoated Dispersible tablet Contains: –
artemether In House 20 mg
Lumefantrine In House 120 mg
titanium dioxide –
Excipients – QSUsage: – Treat to malaria
Category: – Antimalarial drugs
Therapeutic category: – Antimalarial,
Artemether 40mg Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Oily Solution in Amber Glass Ampoules. The oily base ensures a depot effect for sustained release.
Active Ingredient Strength (Standard) Therapeutic Role Artemether IP/Ph.Int 40 mg / 1 ml Pediatric / Low Body Weight Dose Artemether IP/Ph.Int 80 mg / 1 ml Adult Standard Dose Vehicle Q.S. Refined Arachis Oil / Ethyl Oleate Oily Base (Depot) *Pack Sizes: Tray of 6 Ampoules or 10 Ampoules (Standard Course).
Artemether-80 Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Oily Solution in Amber Glass Ampoules. The 80mg strength is specifically formulated to minimize the injection volume for adult patients while delivering a potent therapeutic dose.
Active Ingredient Strength (Adult Dose) Therapeutic Role Artemether IP/Ph.Int 80 mg Potent Antimalarial Vehicle Q.S. to 1 ml Refined Arachis Oil / Ethyl Oleate *Pack Sizes: Tray of 6 Ampoules or 10 Ampoules (Standard Adult Course).
Artesunate 120mg Injection
FreeProduct Composition & Strength
We supply this product as a Combi-Pack containing the Lyophilized Powder and special solvents required for activation.
Component Content Function Vial (Powder) Artesunate 120 mg (Sterile Lyophilized) Active Antimalarial Ampoule 1 Sodium Bicarbonate Injection (5%) – 1ml Activator / Solvent (Solubilizes the acid) Ampoule 2 Sodium Chloride Injection (0.9%) – 5ml Diluent (Adjusts volume/tonicity) *Pack Sizes: Single Combi-Pack or Tray of 5 Combi-Packs.
Artesunate 30mg Injection
FreeProduct Composition & Strength
We supply this product as a Combi-Pack containing the Lyophilized Powder and special solvents required for activation.
Component Content Function Vial (Powder) Artesunate 30 mg (Sterile Lyophilized) Active Antimalarial Ampoule 1 Sodium Bicarbonate Injection (5%) – 0.5ml Activator (Solubilizes the acid) Ampoule 2 Sodium Chloride Injection (0.9%) – 2.5ml Diluent (Adjusts volume/tonicity) *Pack Sizes: Single Combi-Pack or Tray of 5/10 Combi-Packs.
Artesunate 60mg Injection
FreeProduct Composition & Strength
We supply this product as a Combi-Pack containing the Lyophilized Powder and special solvents required for activation.
Component Content Function Vial (Powder) Artesunate 60 mg (Sterile Lyophilized) Active Antimalarial Ampoule 1 Sodium Bicarbonate Injection (5%) – 0.5ml/1ml Activator (Solubilizes the acid) Ampoule 2 Sodium Chloride Injection (0.9%) – 2.5ml/5ml Diluent (Adjusts volume/tonicity) *Pack Sizes: Single Combi-Pack or Tray of 5 Combi-Packs.
Artesunate Amodiaquine Tablets
FreeGlamosunate Tablets Combipak
Artesunate Tablets + Amodiaquine Hydrochloride Tablets USP
Each Filmcoated tablet contains:
Artesunate IP 50 mg
Each Tablet contains :
Amodiaquine Hydrochloride USP
Eq. To Amodiaquine 200 mg
Usage: – Treat severe MalariaCategory: – Antimalarial drugs
Therapeutic category: – Antimalarial,
Artesunate Mefloquine Tablets
FreeArsugin M Combipack
Artesunate Tablets + Mefloquine Tablets
a) Each Film Coated Tablet Contains:
Artesunate 200 mg
b) Each Film Coated Tablet Contains:
Mefloquine Hydrochloride BP
eq. To Mefloquine 250 mgUsage: – Treat severe Malaria
Category: – Antimalarial drugs
Therapeutic category: – Antimalarial,
Artesunate Tablets
FreeARTESUNATE TABLETS 25 MG
each uncoated tablet contains:
artesunate IP 25 mg
excipients q.s.Armax – 50
Artesunate Tablets 50 mg
Each Filmcoated tablet contains:
Artesunate 50 mgArtesunate Tablets 100 mg
Each uncoated tablet contains:
Artesunate IP 100 mg
Excipients – QSArtesunate Tablets 150 mg
Each Uncoated tablet contains:
Artesunate 150 mg
Excipients – q.sArtesunate Tablets 200 mg
Each Filmcoated tablet contains:
Artesunate IP 200 mgARTESUNATE TABLETS INT. PH.+PYRIMETHAMINE AND
SULPHADOXINE- AThree Tablets Of
– Each uncoated tablet contains:
– SULFADOXINE IP 500 MG
– Pyrimethamine IP 25 MG
– BThree Tablet Of
– Each Film coated tablet contains:
– Artesunate IP 200 MGARTESUNNATE + SULPHADOXINE AND
PYRIMETHAMINE IP COB
Each kit contains:Artisunate tab+sulphadoxine and Pyrimethamine tab
three tab of artesunate 150 mg and two tablets of sulphadoxine and pyrimethamine ip 525 mg
CMB
ARTESUNATE + SULPHADOXINE & PYRIMETHAMINE IP-
5 TO 8 YEARS 3 TABLETS OF ARTESUNATE 100MG AND
1 TABLET OF PYRIMETHAMINE AND SULPHADOXINE
TABLETS IP 750 & 37.5MGCMB
EACH KIT CONTAINSARTESUNATE + SULPHADOXINE & PYRIMETHAMINE IP – ADULT 15 YEARS & ABOVE
[ 3 TABLETS OF ARTESUNATE 200 MG AND 3 TABLET OF PYRIMETHAMINE AND SULPHADOXINE TABLETS IP 500 & 25 MG]
CMB
ARTESUNATE + SULPHADOXINE & PYRIMETHAMINE IP – INFANT BELOW 1 YEAR
[ 3 TABLETS OF ARTESUNATE 25 MG AND 2 TABLETS OF SULPHADOXINE & PYRIMETHAMINE TABLETS IP 125 & 6.25 MG] CMB Artesunate Tablets + Pyrimethamine & Sulphadoxine Tablets IP Age 1 to 4 Years CMB
3 TABLETS OF ARTESUNATE AND 1 TABLET OF PYRIMETHAMINE & SULPHADOXINE Each Combipack Contains:
ARTESUNATE TABLETS 50 MG
Each film coated tablet contains:
ARTESUNATE IP 50 mgPYRIMETHAMINE AND SULPHADOXINE TABLETS IP
ARTESUNATE TABLETS + PYRIMETHAMINE & SULPHADOXINE TABLETS IP Age 9 to 14 years CMB
3 TABLETS OF ARTESUNATE AND 2 TABLETS OF PYRIMETHAMINE & SULPH
ARTESUNATE TABLETS 150 MG
Each Uncoated tablet contains:
Artesunate 150 mg
Excipients – q.sPYRIMETHAMINE AND SULPHADOXINE TABLETS IP
Supp Each uncoated tablet contains: Sulphadoxine IP 500 mg
Pyrimethamine IP 25 mg
Excipients – QSArsugin M Combipack
Artesunate Tablets + Mefloquine Tablets
a Each Film Coated Tablet Contains:
Artesunate 200 mg
b Each Film Coated Tablet Contains:
Mefloquine Hydrochloride BP
eq. To Mefloquine 250 mgArsugin SP Combipack
Artesunate Tablets +
Sulfadoxine and Pyrimethamine Tablets USP
a Each Film Coated Tablet Contains:
Artesunate 200 mg
b Each Uncoated Tablet Contains:
Sulfadoxine USP 500 mg
Pyrimethamine USP 25 mgUsage: – Treat severe Malaria
Category: – Antimalarial drugs
Therapeutic category: – Antimalarial,
Ascorbic Acid Tablets
FreeAscorbic Acid Tablets IP 100 mg
Each Uncoated tablet contains :
Ascorbic Acid IP 100 mgAscorbic Acid Tablet IP 300 mg
Each Uncoated Tablet Contains:
Sodium Ascorbate IP
eq. To Ascorbic Acid 300 mgHealthy Vita C
(Ascorbic Acid tablets BP 500 mg)
Each Chewable uncoated tablet contains:
Ascorbic Acid BP 500 mg
Usage: – Treat or prevent low levels of vitamin C and to treat scurvyCategory: – Vitamin supplement
Therapeutic category: – Vitamin supplement
Aspirin Tablets
FreeProduct Composition & Strength
We supply this product in various formats, including Gastro-resistant (Enteric-coated), Dispersible, and standard Film-coated tablets.
Active Ingredient Strength Primary Clinical Function Aspirin (Acetylsalicylic Acid) 75 mg / 81 mg The Cardiac Shield: Low-dose daily therapy for the prevention of heart attack and stroke. Aspirin (Acetylsalicylic Acid) 150 mg Post-Surgical Maintenance: Used for patients with stents or high-risk vascular profiles. Aspirin (Acetylsalicylic Acid) 300 mg / 500 mg The Analgesic Anchor: High-dose relief for acute pain, migraine, and rheumatic fever. Atenolol & Chlorthalidone Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets. The combination is engineered to maintain therapeutic plasma levels over a 24-hour period, supporting once-daily dosing.
Active Ingredient Strength (Standard) Therapeutic Role Atenolol BP/USP 50 mg / 100 mg Cardioselective Beta-Blocker Chlorthalidone BP/USP 12.5 mg / 25 mg Long-acting Diuretic Excipients Q.S. Magnesium Stearate / Povidone Stabilizing Core Matrix Atenolol & S-Amlodipine Besylate Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Film-Coated Tablet, utilizing high-purity S-Amlodipine Besylate to ensure stability.
Active Ingredient Strength Primary Clinical Function Atenolol USP/BP 25 mg / 50 mg The Heart Rate Regulator: Controls the “Pump” by reducing heart rate and myocardial oxygen demand. S-Amlodipine (as Besylate) USP/BP 2.5 mg / 5 mg The Vessel Relaxant: Pure chiral isomer for maximum vasodilation with minimum side effects (edema). Excipients Proprietary Matrix Chiral-Stability System: Engineered to ensure the S-enantiomer remains stable and does not racemize during the shelf-life. Atenolol Tablets
FreeAtenolol Tablets IP 25 mg Each Uncoated Tablet
contains :- Atenolol IP 25 mg
Excipients – QSAtenolol Tablets IP 50 mg
Each Uncoated Tablet contains :
Atenolol IP 50 mg
Excipients q.sOtenol 50
Atenolol Tablets BP
Each Uncoated tablet contains :
Atenolol BP 50 mgOtenol
Atenolol Tablets BP
Each Uncoated tablet contains :
Atenolol BP 100 mgUsage: – Treat high blood pressure and irregular heartbeat
Category: – Hypertensive Cardiac drugs
Therapeutic category: – Cardiovascular Agent,Anti hypertensive
Atorvastatin Clopidogrel and Aspirin Capsules
FreeAtorvastatin, clopidogrel, and aspirin capsules are fixed-dose combination (FDC) medications used for the secondary prevention of cardiovascular events like heart attacks and strokes.
Atorvastatin Tablets
FreeAtorvastatin Tablet IP 10 mg
Each Film Coated Tablet Contains :
Atorvastatin Calcium IP
Eq. to Atorvastatin 10 mgAtorvastatin Tablets IP 20 mg
Each Filmcoated tablet contains :
Atorvastatin Calcium IP
Eq. To Atorvastatin 20 mgUsage: – Lower cholesterol
Category: – Antibiotic drugs
Therapeutic category: – Antilipidemic,Cardiovascular Agent
Atropine Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Clear Glass Ampoules.
Active Ingredient Strength (Standard) Therapeutic Role Atropine Sulphate USP/BP 0.6 mg / ml Pre-operative / Cardiac Dose Atropine Sulphate USP/BP 1 mg / ml Emergency / Antidote Dose Excipients Q.S. Sodium Chloride / Sulfuric Acid (pH adjustment) Isotonicity / Stabilizer *Pack Sizes: Tray of 10 Ampoules, 50 Ampoules, or 100 Ampoules (Hospital/Military Pack).
Azathioprine Tablets
FreeATHEPRIN TABLETS
Azathioprine Tablets IP 50 mg
Each Uncoated Tablet Contains :
Azathioprine IP 50 mgUsage: – Prevent organ rejection in people who have received a kidney transplant
Category: – Arthritis drugs
Therapeutic category: – Arthritis
Azithromycin Dispersible Tablet
FreeProduct Composition & Strength
We supply this product as Dispersible Tablets (DT) with advanced taste-masking technology.
Active Ingredient Strength Flavor Profile Role Azithromycin IP/BP/USP 100 mg Orange / Peppermint Pediatric Low Dose Azithromycin IP/BP/USP 250 mg Orange / Peppermint Pediatric Standard / Adult Excipients Q.S. Sweeteners Disintegrant / Flavor Azithromycin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Infusion in glass vials. It must be reconstituted and further diluted before administration.
Active Ingredient Strength (Standard Adult Dose) Therapeutic Role Azithromycin (as Dihydrate/Citrate) Equivalent to 500 mg Azithromycin Broad Spectrum Macrolide Excipients Q.S. Citric Acid / Sodium Hydroxide pH Adjuster / Solubilizer *Pack Sizes: Tray of 1 Vial, 5 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Azithromycin Tablets
FreeAzithromycin Dispersible Tablet 100mg
Each Disersible tablet cotain:
Azithromycin Dihydrate
Eq.To Anhydrous Azithromycin(100mg)
Std:IP(ACT)
Excipients(QS)(EXC)Azithromycin Tablets IP 250 mg
Each Filmcoated tablet contains:
Azithromycin Dihydrate IP
Eq. To Anhydrous Azithromycin 250 mg
Excipients q.sAzithromycin Tablets IP 500 mg
Each Filmcoated Tablet contains:
Azithromycin Dihydrate IP
Eq. To Anhydrous Azithromycin 500 mg
Excipients q.s
Colour : Ponceau 4RAzithromycin Tablets IP 500 mg
Each Filmcoated Tablet contains:
Azithromycin Dihydrate IP
Eq. To Anhydrous Azithromycin 500 mg
Excipients q.s
Colour : Ponceau 4RAzithromycin Tablets IP 1 gm
Each Film coated tablet contains:
Azithromycin Anhydrous IP 1 gm
As Azithromycin Dihydrate IPLAMITHROCIN / Azithromycin Tablets USP 500 Mg
Each film coated tablet contains:
– Azithromycin Dihydrate
eq. to Azithromycin USP (500 mg)
– Excipients (- QS)Usage: – chest infections
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic
Benzathine Penicillin Injection 1.2 MIU
FreeProduct Composition & Strength
We supply this product as a Sterile Powder for Suspension in glass vials. It requires reconstitution with Sterile Water for Injection to form a milky white suspension.
Active Ingredient Strength (International Units) Therapeutic Role Benzathine Penicillin G USP/BP 1,200,000 IU (1.2 MIU) Standard Adult Prophylaxis Excipients Q.S. Lecithin / Sodium Citrate Suspending / Buffering Agents *Pack Sizes: Tray of 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Benzathine Penicillin Injection 2.4 MIU
FreeProduct Composition & Strength
We supply this product as a Sterile Powder for Suspension in glass vials. It requires reconstitution with Sterile Water for Injection to form a milky white suspension.
Active Ingredient Strength (International Units) Therapeutic Role Benzathine Penicillin G 1,200,000 IU (1.2 MIU) Pediatric / Standard Dose Benzathine Penicillin G 2,400,000 IU (2.4 MIU) Adult Syphilis / High Dose Benzathine Penicillin G 600,000 IU (0.6 MIU) Pediatric Prophylaxis Excipients Q.S. Lecithin / Sodium Citrate Suspending / Buffering Agents *Pack Sizes: Tray of 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Benzhexol Tablet
FreeProduct Composition & Strength
We supply this product as Uncoated Tablets. The chemical name Benzhexol and Trihexyphenidyl refer to the exact same molecule.
Active Ingredient Strength (Standard) Role Trihexyphenidyl HCl (Benzhexol) 2 mg Initial Dose / Mild EPS Trihexyphenidyl HCl (Benzhexol) 5 mg Forte / Maintenance Dose Excipients Q.S. Binder / Filler Benzyl Penicillin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Buffered Powder for Injection in glass vials. The potency is measured in International Units (IU).
Active Ingredient Strength (IU) Mass Equivalent Therapeutic Role Benzyl Penicillin Sodium 1,000,000 IU (1 MU) ~600 mg Standard Dose Benzyl Penicillin Sodium 5,000,000 IU (5 MU) ~3000 mg High Dose (Meningitis) Benzyl Penicillin Sodium 10,000,000 IU (10 MU) ~6000 mg Severe Sepsis Buffering Agent Sodium Citrate – pH Stabilizer *Pack Sizes: Tray of 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Betahistine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Disintegration Tablet, packed in high-barrier Alu-Alu blister strips to ensure the stability of the active dihydrochloride salt.
Active Ingredient Strength Primary Clinical Function Betahistine Dihydrochloride USP/Ph.Eur. 8 mg / 16 mg The Standard Maintenance Anchor: Ideal for long-term management of vertigo and tinnitus. Betahistine Dihydrochloride USP/Ph.Eur. 24 mg / 48 mg The Therapeutic Payload: High-potency dose for acute phases of Meniere’s Disease and severe vestibular dysfunction. Excipients Proprietary Matrix Gastric-Gentle System: Engineered to ensure rapid absorption while minimizing the common “histamine-like” gastric irritation. Biperiden Tablets
FreeBiperiden Hydrochloride Tablets 2 mg
Each Filmcoated Tablet Contains:
Biperiden Hydrochloride BP 2 mgUsage: – stiffness, tremors, spasms, poor muscle control
Category: – Antiparkinson drugs
Therapeutic category: –
Bisacodyl Tablets
FreeBisacodyl Tablets IP 5 mg
Each Enteric tablet contains :
Bisacodyl IP 5 mgBISLAX
Bisacodyl Tablets BP 5 mg
Each Enteric tablet contains :
Bisacodyl BP 5 mgNovolax
Bisacodyl Tablets BP
Each Enteric Coated tablet contains :
Bisacodyl BP 5 mgU-LAX
Bisacodyl Tablets BP
Each Enteric coated tablet contains:
Bisacodyl BP 5 mgUsage: – Treat to constipation
Category: – Laxative drugs
Therapeutic category: –
Bisoprolol Tablets
FreeSolol
Bisoprolol Fumarate Tablets USP
Each Film Coated Tablet Contains:
Bisoprolol Fumarate USP 5 mgUsage: – Treat high blood pressure and heart failure
Category: – Anti Hypertensive / Cardiac Drugs
Therapeutic category: – Antihypertensive
Bleomycin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder in glass vials. Potency is measured in USP Units, not milligrams.
Active Ingredient Strength Therapeutic Role Bleomycin Sulphate USP/IP 15 Units (approx. 15 mg) Antineoplastic Antibiotic Excipients Nitrogen (Inert Atmosphere) None (Pure Powder) *Pack Sizes: Single Vial Box or Tray of 10 Vials.
Bortenat
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder in glass vials. It must be reconstituted with Normal Saline (0.9% NaCl).
Active Ingredient Strength Reference Brand Bortezomib (as Mannitol Ester) 2 mg / Vial Bortenat 2mg Bortezomib (as Mannitol Ester) 3.5 mg / Vial Bortenat 3.5mg Excipients Mannitol USP/BP Stabilizer *Pack Sizes: Single Vial Box with protective Cytotoxic shrink-wrapping.
Bortezomib Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder in glass vials. It contains Mannitol, which forms a stable boronic ester with Bortezomib for shelf stability.
Active Ingredient Vial Strength Primary Use Bortezomib (as Mannitol Boronic Ester) 2 mg / Vial Single Dose / Low BSA Bortezomib (as Mannitol Boronic Ester) 3.5 mg / Vial Standard Adult Dose Excipients Mannitol (Sterile) Stabilizer / Bulking Agent *Pack Sizes: Single Vial Box with protective Cytotoxic shrink-wrapping.
ortezomib injection is an antineoplastic (cancer) medication supplied as a sterile lyophilised powder (freeze-dried cake) for reconstitution. It is most commonly known by the brand name Velcade.
Bromocriptin Tablets
FreeBromocriptin Mesylate Tablets IP 2.5 MG
Each uncoated tablet contains
Bromocriptin Mesylate IP
eq.to Bromocriptine 2.5 mgUsage: – high levels of a natural substance prolactin in the body
Category: – Anti Parkinson drugs
Therapeutic category: – Anti Parkinson
Calcium Carbonate with Vitamin D3 Tablets
FreeCalcium Carbonate With Vitamin D3
Each Uncoated Tablet contains :
Calcium Carbonate 1250 Eq Elemental Calcium IP (500 mg )
Cholecalciferol IP (250 IU)
Excipient (Inhouse ) (-QS)Calcium Carbonate With Vitamin D3Tablets
Each Uncoated Chewable Tablet Contains:
Calcium Carbonate IP 625 mg
Vitamin D3 IP 200 IUCalcium Gluconate Tablets IP 500 mg
Each Uncoated Tablet Contains:
Calcium Gluconate IP 500 mgCAL4U TABLETS
Calcium carbonate with Vitamin D3 Tablets
Each Film Coated Tablet Contains:
Calcium Carbonate IP 625 mg
eq. To Calcium 250 mg
Vitamin D3 125 IU
Shelf life: 1 yearSHREYCAL TABLETS
(Calcium, Minerals with Vitamin D3 Tablets )
Each Filmcoated Tablets Contains:
Calcium Citrate USP 1000 mg
Eq To Elemental Calcium 211 mg
Vitamin D3 IP 200 IU
Magnesium Hydroxide IP Eq To Elemental Magnesium 100 mg
Zinc Sulphate USP Eq To Elemental Zinc 4 mg
Colour : Titanium Dioxide IP
Shelf Life: 2 years
Dosage: As Directed by PhysicianCalcium Gluconate Tablets IP 500 mg
Each Uncoated Tablet Contains:
Calcium Gluconate IP 500 mgUsage: – Treat by low calcium levels
Category: – Generic Drugs
Therapeutic category: – Generic Drugs
Calcium Dobesilate Capsules
FreeCalcium dobesilate capsules are vasoprotective medications primarily used to treat chronic venous insufficiency and diabetic retinopathy. They are most commonly available in a 500 mg strength
Calcium Gluconate Injection
FreeProduct Composition & Strength
We supply this product as a Supersaturated Sterile Solution in 10ml ampoules.
Active Ingredient Concentration Elemental Calcium Content Calcium Gluconate Monohydrate USP/BP 10% w/v (100 mg / ml) 0.465 mEq/ml (Approx 9 mg Ca++ / ml) Stabilizer Calcium D-Saccharate Prevents Crystallization *Pack Sizes: Tray of 10 Ampoules (10ml) or 50 Ampoules.
Calcium Gluconate Tablets
FreeProduct Composition & Strength
We supply this product as Uncoated Tablets or Chewable Tablets in bulk jars or blisters.
Active Ingredient Strength Elemental Calcium Role Calcium Gluconate IP/BP/USP 500 mg ~ 45 mg Pediatric / Maintenance Calcium Gluconate IP/BP/USP 1000 mg (1g) ~ 90 mg Adult Therapeutic Dose Excipients Q.S. — Binder / Sweetener Candesartan Tablets
FreeCandesartan Tablets
Each Film Coated Tablet Contains:
Candesartan Cilexitil 16 mgUsage: – High blood pressure and heart failure
Category: – Anti Hypertensive / Cardiac drugs
Therapeutic category: – Hypertensive
Carbamazepine Tablets
FreeCarbamazepine Tablets IP 200 mg
Each Uncoated Tablet Contains :
Carbamazepine IP 200 mgCarbamezapine Tablets IP 200 mg
Each film Tablet Contains :
Carbamezapine IP 200 mg
excipients q.s.Cirocar – 200 Tablets
Carbamazepine Tablets BP
Each Uncoated Tablet contains :
Carbamazepine BP 200 mgUsage: – Treat to epilepsy
Category: – Hypertensive / Cardiac drugs
Therapeutic category: – Antiepileptic, Anticonvulsant
Carbidopa and Levodopa Tablets
FreeCarbidopa and Levodopa Tablets IP
Each uncoated tablet contains :
Levodopa IP 100 mg
Carbidopa IP
As Anhydrous Carbidopa 10 mgCombimet Tablets
Carbidopa and Levodopa Tablets IP
Each Uncoated tablet contains :
Carbidopa IP
Eq. To anhydrous Carbidopa 25 mg
Levodopa IP 250 mgLEVODOPA AND CARBIDOPA TABLETS 100 +10 MG
Each filmcoated tablet contains: Levodopa BP 100 mg
– Carbidopa BP –
– As Anhydrous Carbidopa BP 10 mg
– Titanium dioxide –
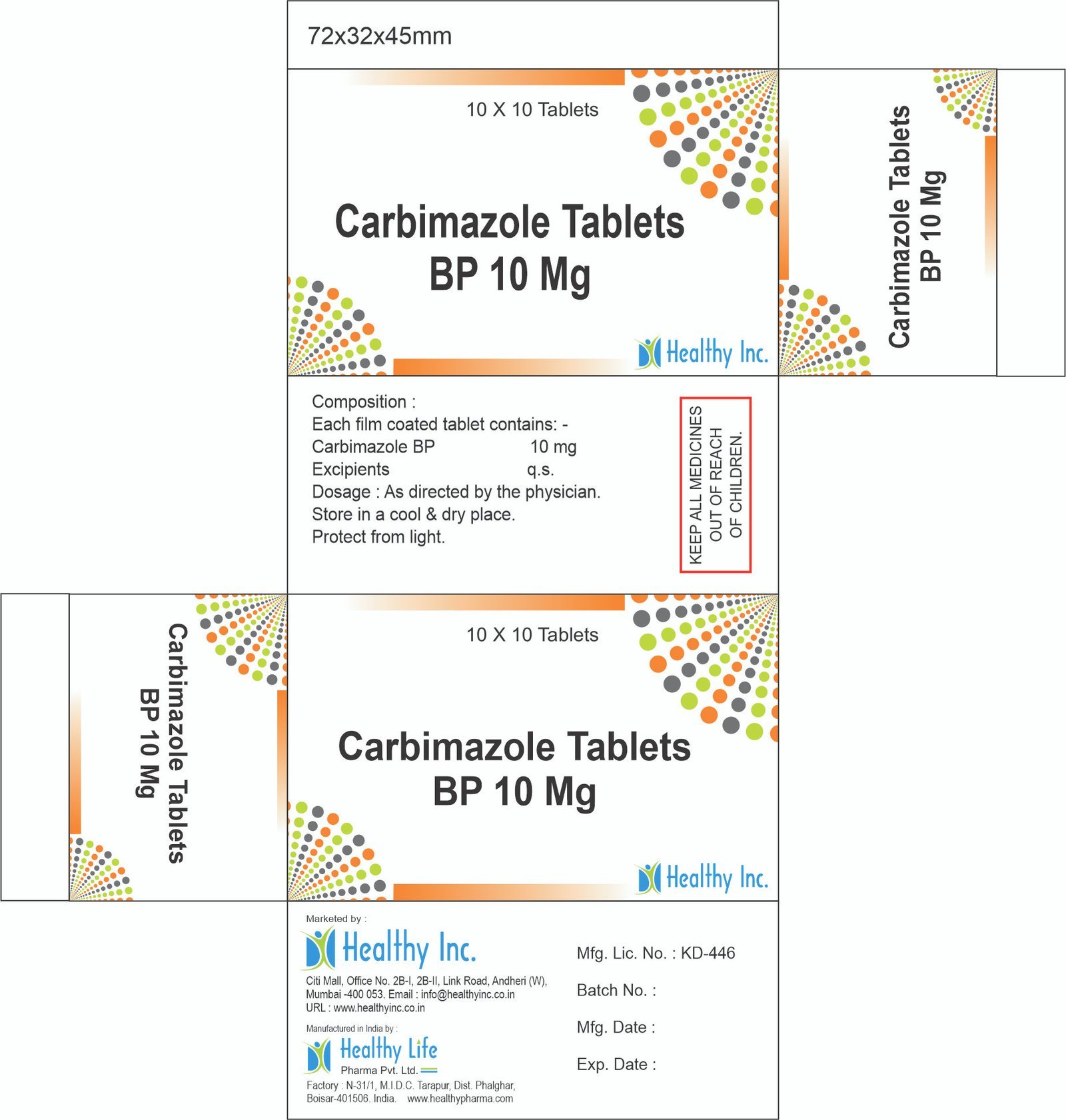
– Excipients – QSCarbimazole Tablets
FreeCarbimazole Tablets BP 10 Mg
Each film coated tablet contains: Carbimazole BP 10 mg Excipient – QS
Usage: – Treat an overactive thyroidCategory: – Thyroid Hormones drugs
Therapeutic category: – Thyroid Hormones
Carboplatin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Liquid Concentrate in glass vials. It is ready to dilute.
Active Ingredient Concentration Vial Size (Total Drug) Carboplatin USP/BP 10 mg / ml 150 mg (15 ml vial) Carboplatin USP/BP 10 mg / ml 450 mg (45 ml vial) Excipients Water for Injection / Mannitol – *Pack Sizes: Single Vial Box with protective Cytotoxic shrink-wrapping.
Carvedilol Tablets
FreeCarvedilol Tablets 3.125 mg
Each Uncoated tablet contains :
Carvedilol BP 3.125 mgCarvedilol Tablets 6.25 mg
Each Uncoated Tablet Contains:
Carvedilol BP 6.25 mgCarvy
Carvedilol Tablets 12.5 mg
Each Film Coated Tablet Contains:
Carvedilol BP 12.5 mgUsage: – Treat high blood pressure and heart failure
Category: – Anthelmintic drugs
Therapeutic category: –
Cefadroxil 500 mg Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet, packed in highly secure, moisture-resistant Alu-Alu or High-Density Alu-PVC blister strips to ensure the absolute stability of the beta-lactam ring.
Active Ingredient Strength Primary Clinical Function Cefadroxil Monohydrate USP/Ph.Eur. 500 mg (Equivalent to anhydrous Cefadroxil) Global Standard: Twice-daily adult therapy for pharyngitis, skin infections, and UTIs. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Macrogol / Magnesium Stearate Diluent / Superdisintegrant / Film-Coating (Engineered for rapid gastric dissolution and high bioavailability) Cefepime & Sulbactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 2:1.
Strength Variant Cefepime Content Sulbactam Content Ratio 1.5 g Vial 1000 mg (1 g) 500 mg (Sodium) 2:1 (Standard) Excipients L-Arginine Buffer / Stabilizer – *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefepime & Tazobactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 8:1.
Strength Variant Cefepime Content Tazobactam Content Ratio 1.125 g Vial 1000 mg (1 g) 125 mg (Sodium) 8:1 (Standard) Excipients L-Arginine Buffer / Stabilizer – *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefepime Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. It contains L-Arginine as a buffering agent (instead of sodium salts).
Active Ingredient Strength Therapeutic Role Cefepime Hydrochloride USP/BP Equivalent to 1000 mg (1 g) Cefepime Severe Infection / Sepsis Cefepime Hydrochloride USP/BP Equivalent to 500 mg Cefepime Pediatric / Moderate Infection Buffer L-Arginine pH Stabilizer (Approx 725 mg per g) *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefixime Capsules 400mg
FreeCefixime 400mg capsules are third-generation cephalosporin antibiotics used for a variety of bacterial infections. While 200mg is a common dose, the 400mg strength is often used for once-daily dosing.
Cefoperazone & Sulbactam 2Gm Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in 20ml glass vials.
Active Ingredient Strength Function Cefoperazone Sodium USP/BP 1000 mg (1 g) Anti-Pseudomonal Cephalosporin Sulbactam Sodium USP/BP 1000 mg (1 g) Beta-Lactamase Inhibitor Ratio 1 : 1 High Sulbactam Load *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with 10ml Water for Injection (WFI).
Cefoperazone & Sulbactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 1:1.
Strength Variant Cefoperazone Content Sulbactam Content Ratio 1.5 g Vial 500 mg (Sodium) 500 mg (Sodium) 1:1 (Standard) 3.0 g Vial 1000 mg (1 g) 1000 mg (1 g) 1:1 (High Dose) Excipients None (Sterile Blend) – – *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefoperazone & Tazobactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 8:1.
Strength Variant Cefoperazone Content Tazobactam Content Ratio 1.125 g Vial 1000 mg (1 g) 125 mg (Sodium) 8:1 (Standard) Excipients None (Sterile Blend) – – *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
1.125 g Vial 1000 mg (1 g) 125 mg (Sodium) 8:1 (Standard) Excipients None (Sterile Blend) – – Cefoperazone Sodium Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. It contains approximately 34 mg (1.5 mEq) of sodium per gram.
Active Ingredient Strength (Standard) Therapeutic Role Cefoperazone Sodium USP/BP Equivalent to 1000 mg (1 g) Cefoperazone Adult Surgical / Biliary Dose Excipients None (Pure Sterile Powder) – *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefotaxime Sodium Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The sodium content is lower than many other cephalosporins (approx 2.2 mmol/g).
Active Ingredient Strength Primary Patient Cefotaxime Sodium USP/BP Equivalent to 1000 mg (1 g) Adults (Meningitis/Sepsis) Cefotaxime Sodium USP/BP Equivalent to 500 mg Pediatrics Cefotaxime Sodium USP/BP Equivalent to 250 mg Neonates / Infants Excipients None (Pure Sterile Powder) – *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefpirome & Sulbactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 2:1.
Strength Variant Cefpirome Content (as Sulfate) Sulbactam Content (as Sodium) Ratio 1.5 g Vial 1000 mg (1 g) 500 mg 2:1 (Standard) Excipients Sodium Carbonate Buffer – *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefpirome Sulfate Injection
FreeCefpirome sulfate injection is a fourth-generation cephalosporin antibiotic typically supplied as a sterile dry powder for intravenous (IV) or intramuscular (IM) administration
Ceftazidime & Sulbactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is either 2:1 or 1:1 depending on the market requirement.
Strength Variant Ceftazidime Content Sulbactam Content Ratio 1.5 g Vial 1000 mg (1 g) 500 mg (Sodium) 2:1 (Standard) 2.25 g Vial 1125 mg 1125 mg 1:1 (High Potency) Excipients Sodium Carbonate Buffer – *Pack Sizes: Tray of 1 Vial or Box of 1/10 Vials with Water for Injection (WFI).
Ceftazidime & Tazobactam Injection
FreeCeftazidime and tazobactam injection is a combination antibiotic used primarily in hospital settings for serious, multi-drug resistant bacterial infections. It is typically available as a sterile dry powder for reconstitution.
Product Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 8:1.
Strength Variant Ceftazidime Content Tazobactam Content Target Patient 1.125 g Vial 1000 mg (1 g) 125 mg (Sodium) Standard Adult Dose 281.25 mg Vial 250 mg 31.25 mg Pediatric Dose Excipients Sodium Carbonate Buffer Solubilizer Ceftazidime Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. It contains Sodium Carbonate as a solubilizer.
Active Ingredient Strength Primary Patient Ceftazidime Pentahydrate USP/BP Equivalent to 1000 mg (1 g) Adults (Standard Dose) Ceftazidime Pentahydrate USP/BP Equivalent to 250 mg Pediatrics / Neonates Excipients Sodium Carbonate Gas-Releasing Buffer *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Ceftriaxone & Sulbactam injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 2:1.
Strength Variant Ceftriaxone Content Sulbactam Content Target Patient 1.5 g Vial 1000 mg (1 g) 500 mg (Sodium) Adult Standard Dose 750 mg Vial 500 mg 250 mg Pediatric / Moderate Dose 375 mg Vial 250 mg 125 mg Infant / Neonate Dose Ratio 2 : 1 – Optimal Synergy *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Ceftriaxone & Tazobactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 8:1.
Strength Variant Ceftriaxone Content Tazobactam Content Ratio 1.125 g Vial 1000 mg (1 g) 125 mg (Sodium) 8:1 (Standard) 562.5 mg Vial 500 mg 62.5 mg Pediatric Excipients None (Sterile Blend) – – *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Ceftriaxone 1gm Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials.
Active Ingredient Strength Standard Use Ceftriaxone Sodium USP/BP Equivalent to 1000 mg (1 g) Ceftriaxone Adult Standard Dose Sodium Content Approx 83 mg (3.6 mEq) Per 1g Vial *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Ceftriaxone 500mg Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials.
Active Ingredient Strength Target Use Ceftriaxone Sodium USP/BP Equivalent to 500 mg Ceftriaxone Pediatrics / IM Gonorrhea Sodium Content Approx 41.5 mg (1.8 mEq) Low Sodium Load *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefuroxime & Sulbactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 2:1.
Strength Variant Cefuroxime Content Sulbactam Content Primary Use 1.5 g Vial 1000 mg (1 g) 500 mg (Sodium) Adult Therapy / Surgery 1.125 g Vial 750 mg 375 mg Moderate Infection 375 mg Vial 250 mg 125 mg Pediatrics *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefuroxime Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials.
Active Ingredient Strength Primary Use Cefuroxime Sodium USP/BP Equivalent to 750 mg Cefuroxime Standard Adult Dose Cefuroxime Sodium USP/BP Equivalent to 1500 mg (1.5 g) Severe Infections / Surgery Excipients None (Pure Sterile Powder) – *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Celecoxib Capsules
FreeCelecoxib capsules are selective non-steroidal anti-inflammatory drugs (NSAIDs) used to manage pain and inflammation. They are commonly available in strengths of 50 mg, 100 mg, 200 mg, and 400 mg.
Cephalexin Capsules
FreeCephalexin capsules are a first-generation cephalosporin antibiotic used to treat various bacterial infections. They are typically available in strengths of 250 mg, 500 mg, and sometimes 333 mg or 750 mg.
Cephalothin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. It contains approximately 2.8 mEq (64 mg) of Sodium per gram.
Active Ingredient Strength Therapeutic Role Cephalothin Sodium USP/BP Equivalent to 1000 mg (1 g) Cephalothin Surgical Prophylaxis / Skin Infection Excipients Sodium Bicarbonate Buffer / pH Adjuster *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cetrizine Tablets
FreeLevocetirizine Tablets IP 5 mg
Each Uncoated Tablet Contains:
Levocetirizine IP 5 mg
Excipients q.sCitrin – 10 Cetrizine Tablets 10 mg
Each Filmcoated tablet contains:
Cetrizine Dihydrochloride BP 10 mgUsage: – Relieve runny nose
Category: – Anti Allergic Drugs
Therapeutic category: – Antihistamine, Anti Allergic
Chloramphenicol Sodium Succinate Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in glass vials. It must be reconstituted before use.
Active Ingredient Strength Therapeutic Role Chloramphenicol Sodium Succinate USP/BP Equivalent to 1000 mg (1 g) Chloramphenicol Broad Spectrum Antibiotic Excipients None (Pure Sterile Powder) – *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
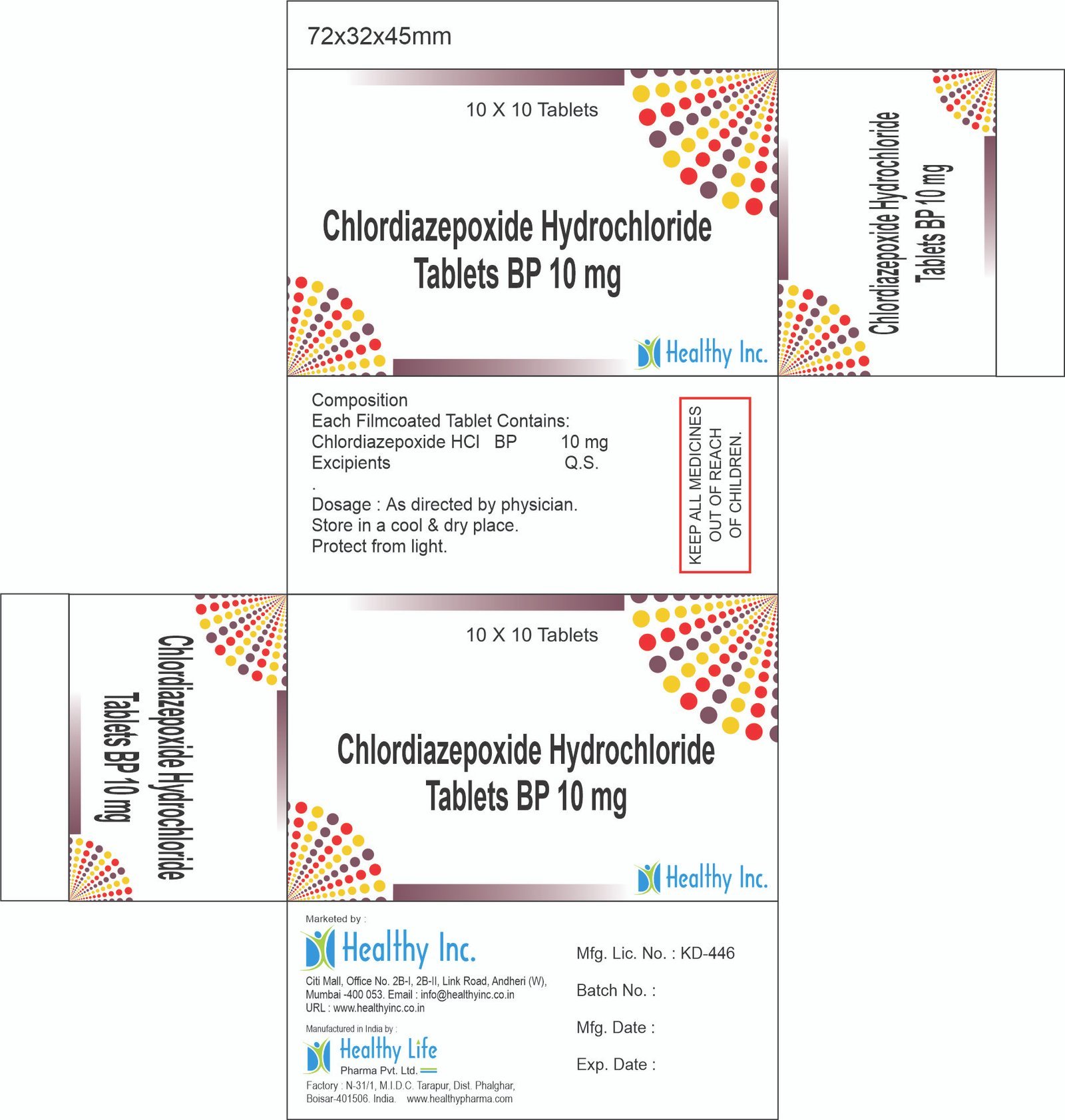
Chlordiazepoxide Hydrochloride Tablets
FreeProduct Composition & Strength
We supply this product as Sugar Coated Tablets (often Green or Yellow). The robust coating is essential to mask the intensely bitter taste of the API and protect the light-sensitive drug.
Active Ingredient Strength (Standard) Therapeutic Role Chlordiazepoxide HCl USP/BP 10 mg Mild Anxiety / Elderly / Taper End Chlordiazepoxide HCl USP/BP 25 mg Severe Anxiety / Alcohol Detox Loading Excipients Q.S. Sucrose / Talc / Calcium Carbonate Sugar Coating (Light Barrier) Chloroquine Phosphate Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Amber Glass Vials/Ampoules.
Active Ingredient Salt Strength Base Equivalent Chloroquine Phosphate USP/BP 64.5 mg / ml Equivalent to 40 mg Chloroquine Base Total Vial Content 30 ml Multi-dose Vial 1200 mg Base Total Excipients Chlorocresol (Preservative) Water for Injection *Pack Sizes: Tray of 10 Ampoules (5ml) or Multi-dose Vials (30ml).
Chloroquine Phosphate Tablets
FreeCHLOROQUINE PHOSPHATE TABLETS IP 100 MG
Each film coated tablet contains: – CHLOROQUINE PHOSPHATE IP 160 MG Eq. to Chloroquine base 100 MGM QUEEN – 250
Chloroquine Phosphate Tablets IP 250 mg
Each Sugarcoated Tablet Contains
Chloroquine Phosphate IP 250 mgMosquin
Chloroquine phosphate Tablets BP
Each Uncoated tablet contains :
Chloroquine Phosphate BP 160 mg
Eq. To Chloroquine Base 100 mgMqueen -250
Chloroquine Phosphate Tablets BP
Each Sugarcoated tablet contains :
Chloroquine Phosphate BP 250 mgChloroquine Tablets
FreeCHLOROQUINE PHOSPHATE TABLETS IP 100 MG Each film coated tablet contains: CHLOROQUINE PHOSPHATE IP 160 MG Eq. to Chloroquine base 100 MG
Mqueen -250
Chloroquine Phosphate Tablets BP
Each Sugarcoated tablet contains :
Chloroquine Phosphate BP 250 mgMosquin
Chloroquine phosphate Tablets BP
Each Uncoated tablet contains :
Chloroquine Phosphate BP 160 mg
Eq. To Chloroquine Base 100 mgM QUEEN – 250
Chloroquine Phosphate Tablets IP 250 mg
Each Sugarcoated Tablet ContainsChloroquine Phosphate IP 250 mg
Usage: – Prevent and treat malaria
Category: – Antimalarial drugs
Therapeutic category: – Antimalarial
Chlorpheniramine Tablets
FreeChlorpheniramine Maleate Tablets IP 4 mg
Each Uncoated tablet contains :
Chlorpheniramine Maleate IP 4 mgUsage: – Uses Hay fever, and the common cold
Category: – Anti Allergic drugs
Therapeutic category: – Antiasthmatic
Chlorpromazine Hydrochloride Tablets
FreeChlorpromazine Hydrochloride Tablets IP 25 mg
Each Sugarcoated tablets contains :
Chlorpromazine Hydrochloride IP 25 mgCHLORPROMAZINE HYDROCHLORIDE TABLETS IP 50 MG Each sugar coated Tablet Contains:
chlorpromazine hydrochloride IP 50 mg Excipients q.s.Chlorpromazine Tablets IP 100 mg
Each Sugar Coated Tablet Contains:
Chlorpromazine Hydrochloride IP 100 mgChlorpromazine Tablets IP 200 mg
Each Sugarcoated tablet contains :
Chlorpromazine Hydrochloride IP 200 mg Excipients -QSUsage: – treat the symptoms of schizophrenia
Category: – Anti Psychotic Drugs + CNS
Therapeutic category: –Cholecalciferol (Vitamin D3) Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Oily Solution in glass ampoules.
Active Ingredient Strength Vehicle Cholecalciferol USP/BP (Vitamin D3) 300,000 IU / 1 ml (7.5 mg) Oily Base (Arachis or Sesame Oil) Cholecalciferol USP/BP (Vitamin D3) 600,000 IU / 1 ml (15 mg) Oily Base (Arachis or Sesame Oil) Excipients BHT / Benzyl Alcohol Antioxidant / Preservative *Pack Sizes: Tray of 5 Ampoules, 10 Ampoules, or 50 Ampoules.
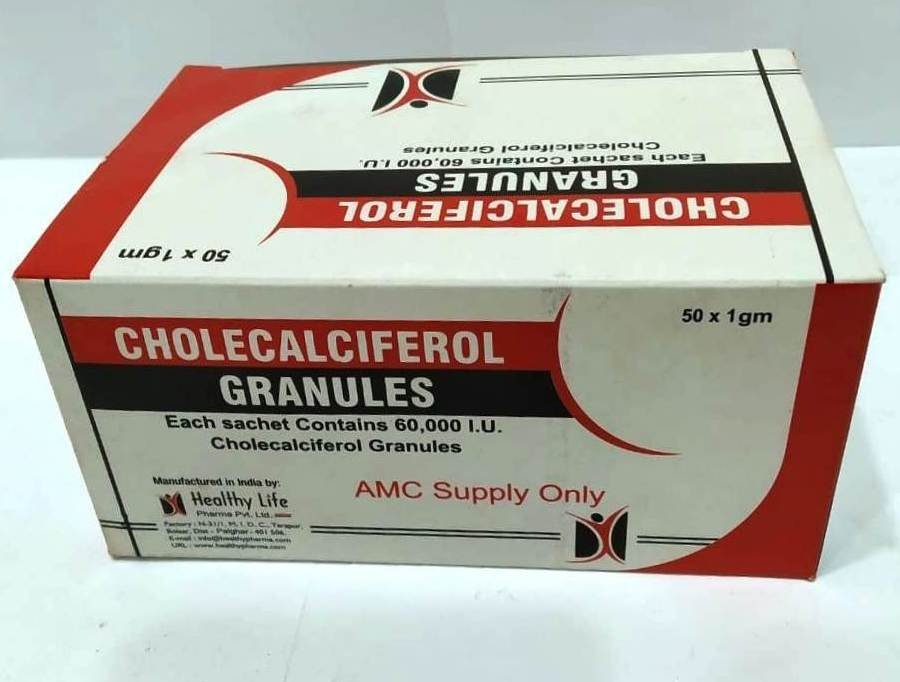
Cholecalciferol Granules Sachet
FreeCHOLECALCIFEROL GRANULES each sachet of 1 gm contains: cholecalciferol IP 60000 IU Excipients
Usage: – Dietary supplement
Category: – Vitamin Supplement
Therapeutic category: – Vitamin Supplement
Chymotrypsin Tablets
FreeCirozyme – 1 Tablets
Trypsin – Chymotrypsin Tablets
Each Enteric coated tablet contains :100000 Armour units of enzymatic activity supplied by a purified concentrate
which has specific trypsin & chymotrypsin activity in a ratio of approximately six to one
Usage: – Redness and Swelling with pockets of infection (abscesses),ulcers, surgery, or critical illness
Category: – Anti Allergic drugs
Therapeutic category: – Allergic drugs
Cilostazol Tablets
FreeCilostazol Tablets 50 mg
Each Uncoated Tablet Contains:
Cilostazol 50 mgCilostazol Tablets 100 mg
Each Uncoated Tablet contains :
Cilostazol 100 mgUsage: – improve the symptoms of a certain blood flow problem in the legs
Category: – Anti Hypertensive / Cardiac Drugs
Therapeutic category: – Anti Hypertensive
Cimetidine Tablets
FreeGlatidine 200
Cimetidine Tablets USP 200 mg
Each Uncoated tablet contains :
Cimetidine USP 200 mgGlatidine 400
Cimetidine Tablets USP 400 mg
Each Uncoated tablet contains :
Cimetidine USP 400 mgUsage: – Treat and prevent certain types of stomach ulcer
Category: – Antacid & Anti ulcer drugs
Therapeutic category: – Antiulcer,Antacid
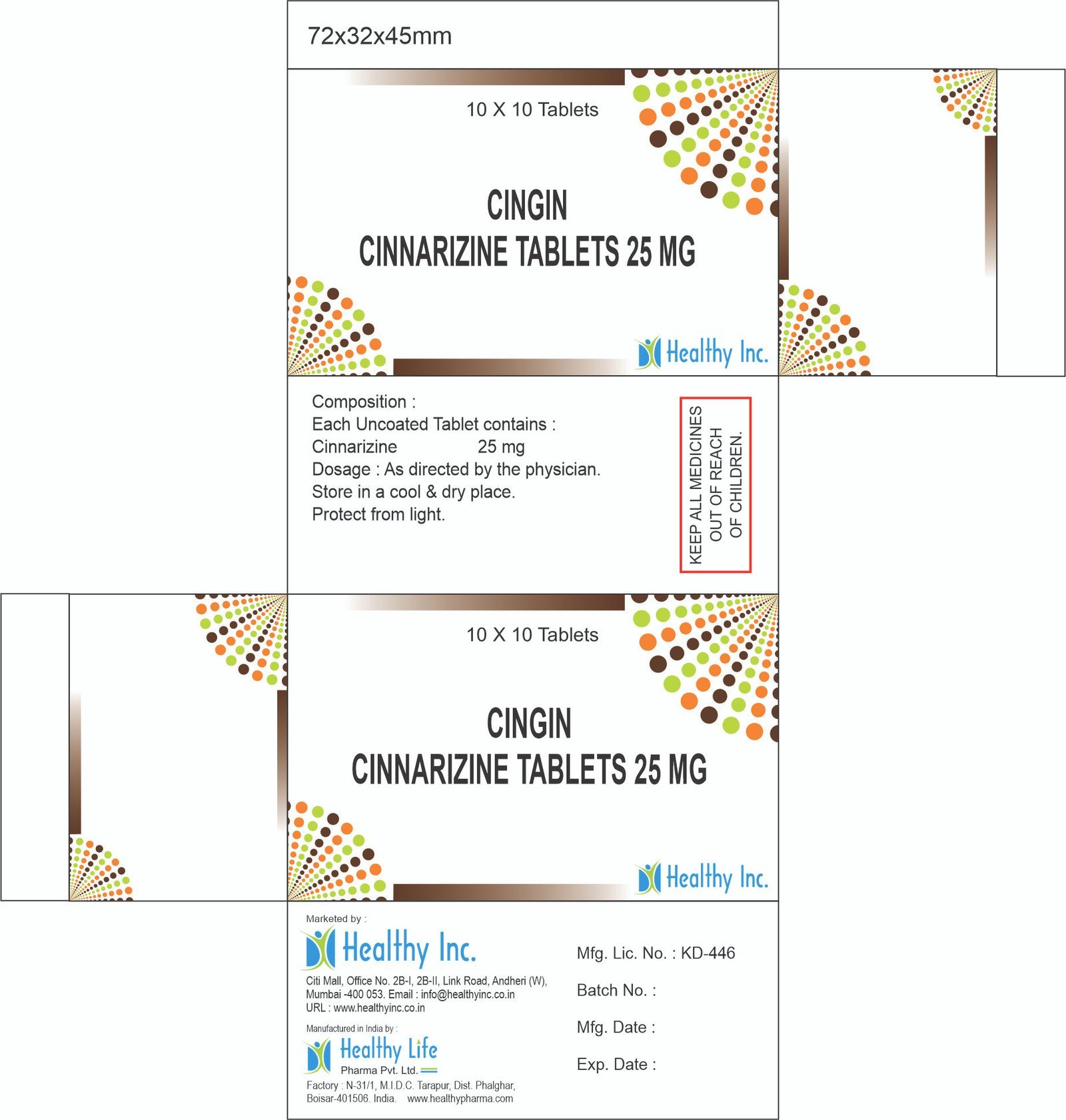
Cinnarizine Tablets
FreeCingin
Cinnarizine Tablets 25 mg
Each Uncoated Tablet contains :
Cinnarizine 25 mgUsage: – Travel sickness
Category: – Anti Allergic Drugs
Therapeutic category: – Anti Allergic
Ciprofloxacin Tablets
FreeCiprofloxacin Dispersible Tablets
Each Uncoated Dispersible tablet contains:
Ciprofloxacin Hydrochloride IP
Eq. Ciprofloxacin 100 mg
Approved colour & FlavourCiprofloxacin Tablets IP 250 mg
Each Film coated tablet contains:
Ciprofloxacin Hydrochloride IP
Eq.To Ciprofloxacin Anhydrous 250 mg
Color : Titanium Dioxide IPCiprofloxacin Tablets IP 500 mg
Each Filmcoated tablet contains :
Ciprofloxacin Hydrochloride IP
Eq.To Ciprofloxacin Anhydrous 500 mg
Excipients q.sCiproctan – 250 Tablets
Ciprofloxacin Tablets BP 250 mg
Each Filmcoated tablet contains :
Ciprofloxacin Hydrochloride BP
Eq.To Ciprofloxacin Anhydrous 250 mgCiproctan
Ciprofloxacin Tablets BP 500 mg
Each Filmcoated tablet contains :
Ciprofloxacin Hydrochloride BP
Eq.To Ciprofloxacin Anhydrous 500 mgUsage: – Treat or prevent certain infections caused by bacteria such as pneumonia
Category: – Antibiotic Drugs
Therapeutic category: – Antibiotic
Ciprofloxacin Tinidazole Tablets
FreeOctum – TZ
Each Filmcoated tablet contains :
Ciprofloxacin HCl BP
Eq.To Ciprofloxacin Anhydrous 500 mg
Tinidazole BP 600 mgUsage: – Bacterial & Parasitic infections
Category: – Antibiotic Drugs
Therapeutic category: – Antibiotic
Citicoline Sodium Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Glass Ampoules or Vials.
Active Ingredient Strength Volume Citicoline Sodium USP/BP 500 mg 2 ml Ampoule (250 mg/ml) Citicoline Sodium USP/BP 1000 mg (1 g) 4 ml Ampoule (250 mg/ml) Excipients Water for Injection / pH Adjusters Preservative Free (Single Use) *Pack Sizes: Tray of 5 Ampoules, 10 Ampoules, or 50 Ampoules.
Citicoline Tablets
FreeCITICOLINE -1000</p>
CITICOLINE CONTROLLED RELEASE TABLETS 1000 MG
CITICOLINE SODIUMEQ TO CITICOLINE 1000 MG
ERYTHROCYINE & BRILLIANT BLUE FCF
EXCIPIENTS QSUsage: – Help memory loss due to aging
<strong>Category: – Antiparkinson drugs
Therapeutic category: – Nootropic, CNS Drugs
Clarithromycin Tablets
FreeClarithromycin Tablets USP 500 mg
Each Filmcoated tablet contains :
Clarithromycin USP 500 mgUsage: – Chest infections
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic
Clindamycin Capsules
FreeClindamycin capsules are lincosamide antibiotics primarily used to treat serious bacterial infections, including those of the skin, lungs, and internal organs. They are most commonly formulated as Clindamycin Hydrochloride.
Clindamycin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Glass Ampoules or Vials.
Active Ingredient Strength Volume Clindamycin Phosphate USP/BP 300 mg 2 ml Ampoule (150 mg/ml) Clindamycin Phosphate USP/BP 600 mg 4 ml Ampoule (150 mg/ml) Excipients Benzyl Alcohol / EDTA Preservative / Stabilizer *Pack Sizes: Tray of 5 Ampoules, 10 Ampoules, or 50 Ampoules.
Clobazam Tablets
FreeClobazam Tablet 5 mg
Each Uncoated Tablet Contains :
Clobazam IP 5 mgClobazam Tablet 10 mg
Each Uncoated Tablet Contains :
Clobazam IP 10 mgUsage: – Control seizures
Category: – Hypertensive / Cardiac drugs
Therapeutic category: – Anti convulsant , Antiepileptic
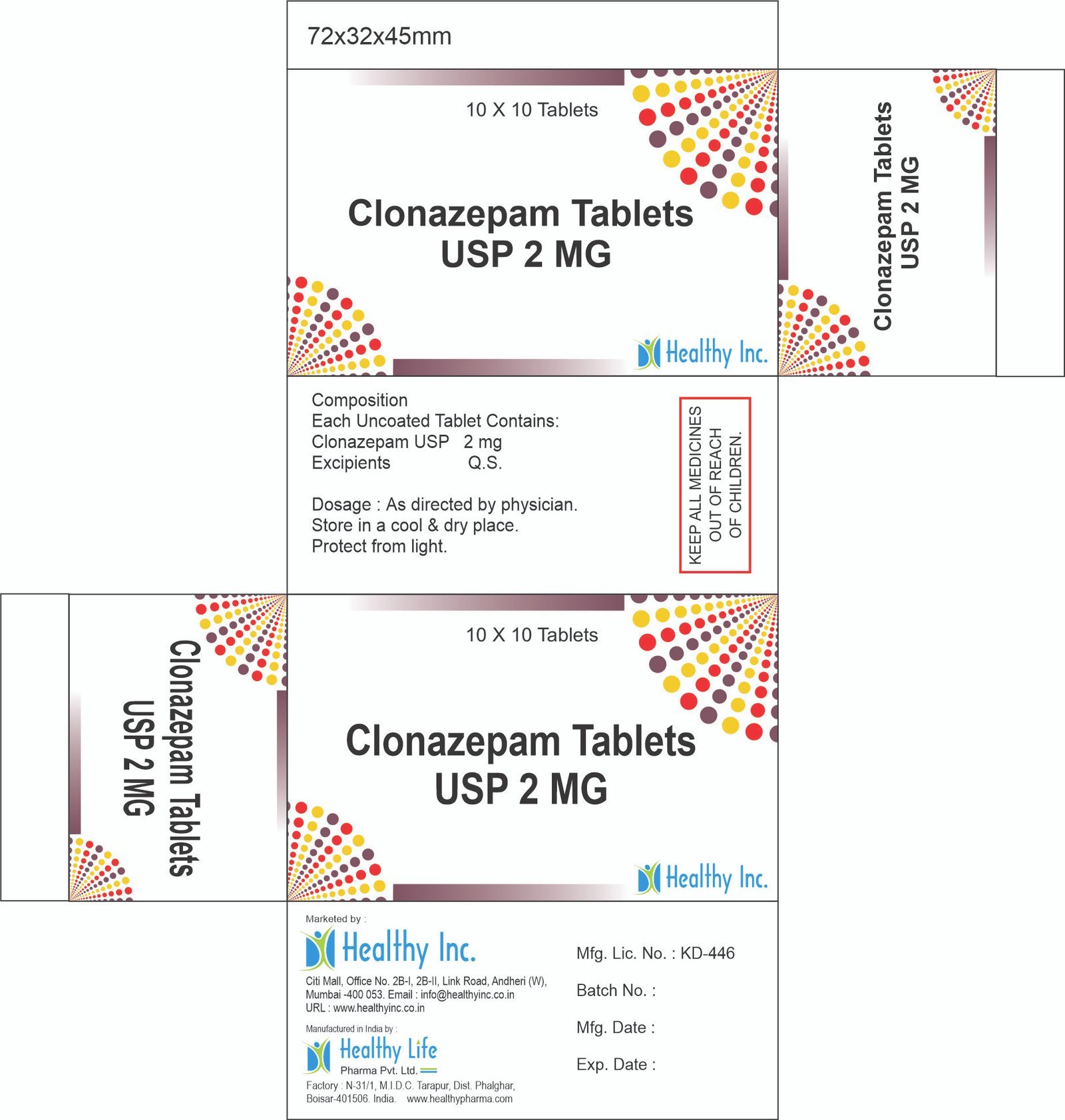
Clonazepam Tablets
FreeClonazepam Tablets IP 0.5 mg
Each Uncoated Tablet contains :
Clonazepam IP 0.5 mgClonazepam Tablets USP 2 MG
Each Uncoated Tablet Contains:
Clonazepam USP 2 mgUsage: – Control seizures or fits due to epilepsy, involuntary muscle spasms, panic disorder and sometimes restless legs syndrome
Category: – Anti Hypertensive / Cardiac drugs
Therapeutic category: – Anti convulsant , Antiepileptic
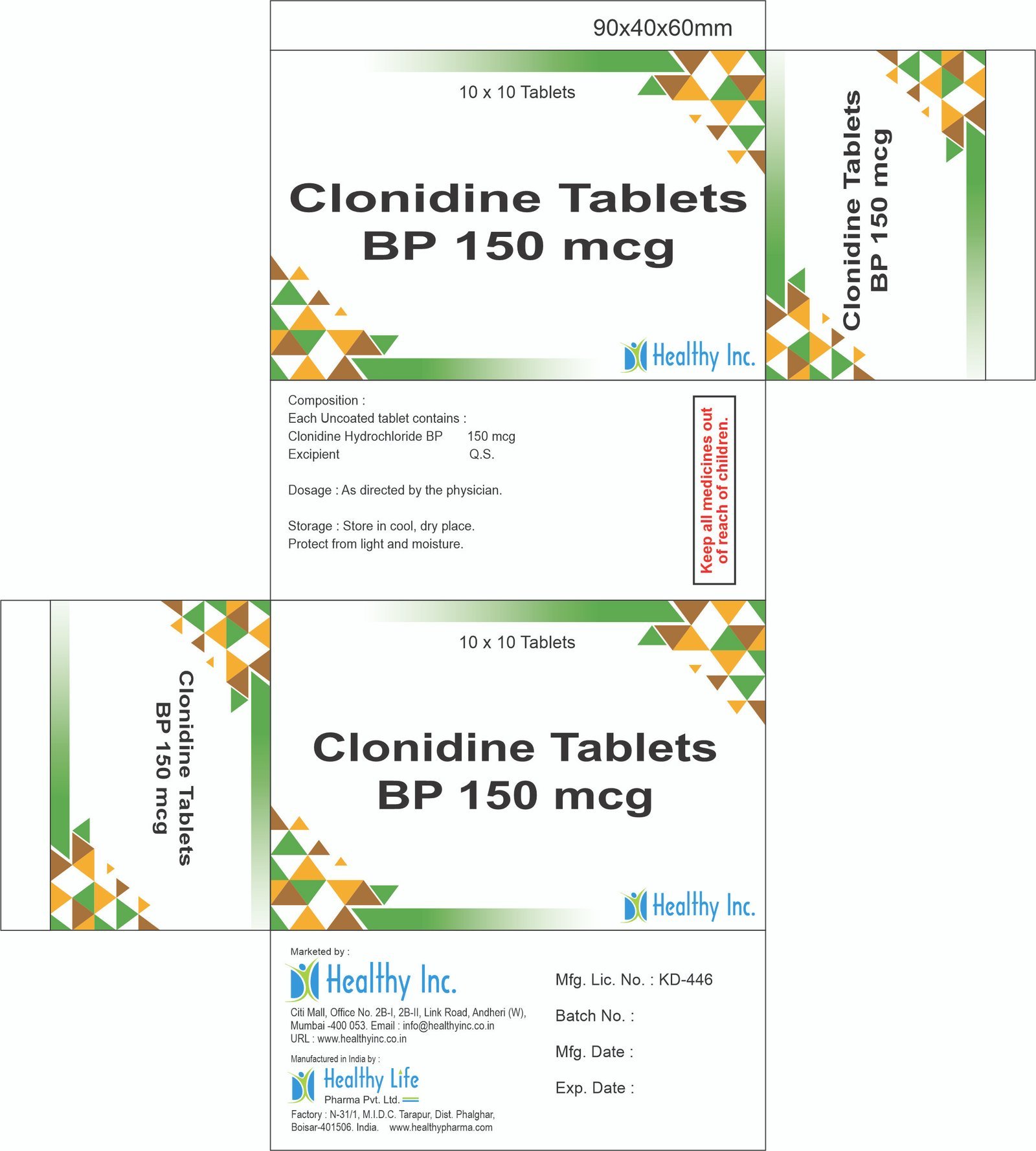
Clonidine Tablets
FreeClonidine Tablets BP 75 mcg
Each Uncoated tablet contains :
Clonidine Hydrochloride BP 75 mcgClonidine Tablets BP 150 mcg
Each Uncoated tablet contains :
Clonidine Hydrochloride BP 150 mcgUsage: – Treat high blood pressure (hypertension)
Category: – Anti Hypertensive / Cardiac drugs
Therapeutic category: – Antiplatelets
Clopidogrel & Aspirin Capsules
FreeClopidogrel and Aspirin capsules are a combination of two antiplatelet medications (blood thinners) used primarily to prevent heart attacks and strokes in patients with high cardiovascular risk.
Clopidogrel Tablets
FreeClopidogrel Tablets IP
Each Film Coated Tablet Contains:
Clopidogrel Bisulphate IP
eq. To Clopidogrel 75 mgClopidogrel Tablets USP 75 mg
Each Filmcoated tablet contains :
Clopidogrel Bisulphate USP
Eq. To Clopidogrel 75 mgPlaxina
Clopidogrel With Aspirin Tablets
Each Film Coated Tablet Contains:
Clopidogrel Bisulphate USP
E.Q To Clopidogrel 75 mg
Aspirin BP 75 mgUsage: – Prevents Platelets
Category: – Anti Hypertensive / Cardiac drugs
Therapeutic category: – Antiplatelets
Clopidogrel, Aspirin Tablets Clopidogrel, Aspirin Tablets
FreeA Clopidogrel + Aspirin tablet is a dual antiplatelet therapy (DAPT) used primarily to prevent serious cardiovascular events like heart attacks and strokes
Clotrimazole Vaginal Tablets
FreeClotry Clotrimazole Vaginal Tablets IP 100 mg
Each Uncoated tablet contains:
Clotrimazole IP 100 mgClotry
Clotrimazole Vaginal Tablets USP
Each Uncoated tablet contains:
Clotrimazole USP 100 mgClotry – 500
Clotrimazole Vaginal Tablets USP
Each Uncoated tablet contains :
Clotrimazole USP 500 mgUsage: – Treat yeast infections of the vagina.
Category: – AntiFungal drugs
Therapeutic category: – Anti fungal
Cloxacillin Capsules
FreeCloxacillin capsules are penicillin-type antibiotics used specifically to treat infections caused by staphylococci (staph infections) that produce an enzyme called penicillinase.
Cloxacillin 1gm Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials.
Active Ingredient Strength Primary Use Cloxacillin Sodium USP/BP Equivalent to 1000 mg (1 g) Cloxacillin Severe Infection / Surgery Sodium Content Approx 2.3 mEq (53 mg) per gram Monitor in Heart Failure *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Clozapine Tablets
FreeCirocloz Tablets
Clozapine Tablets USP 100 mg
Each Uncoated tablet contains :
Clozapine USP 100 mgUsage: – Treat certain mental/mood disorders
Category: – Antipsychotic / CNS Drugs
Therapeutic category: –
Co -Trimoxazole Tablets Tablet
FreeCo- Trimoxazole Tablets IP
Trimethoprime & Sulphamethoxazole Tablets IP
Each Uncoated tablet contains :
Trimethoprime IP 20 mg
Sulphamethoxazole IP 100 mgTrimethoprim & sulphamethoxazole tablets IP Co -trimoxazole Tablets IP
Each film coated tablets : Excipients q.s
Trimethoprim IP 80 mg
Sulphamethoxazole IP 400 mgTrimethoprim & sulphamethoxazole tablets IP
Each film coated tablets :
Trimethoprim IP 160 mg
Sulphamethoxazole IP 800 mgCo – Trimoxazole Tablets BP
Trimethoprim & Sulphamethoxazole Tablets BP
Each Uncoated Tablet contains :
Trimethoprim BP 80 mg
Sulfamethoxazole BP 400 mgTACOT – DS
Trimethoprim & Sulphamethoxazole Tablets BP
Each Uncoated tablet contains :
Trimethoprim BP 160 mg
Sulphamethoxazole BP 800 mgUsage: – Traveler’s diarrhea
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic
Corion
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in glass vials with a specific solvent.
Active Ingredient Strength (IU) Primary Use Chorionic Gonadotropin USP/BP 5000 IU Standard Ovulation Trigger Chorionic Gonadotropin USP/BP 10000 IU High Dose / IVF Trigger Chorionic Gonadotropin USP/BP 2000 IU Male Hypogonadism / Maintenance Solvent Sodium Chloride (Isotonic) 1 ml or 2 ml Ampoule *Pack Sizes: Tray of 1 Vial + Solvent, 5 Vials + Solvents.
Cyclophosphamide Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in glass vials.
Active Ingredient Strength Primary Use Cyclophosphamide USP/BP 200 mg Vial Low Dose / Autoimmune Cyclophosphamide USP/BP 500 mg Vial Standard Chemotherapy Cyclophosphamide USP/BP 1000 mg (1 g) Vial High Dose / Hematology Excipients Mannitol / Sodium Chloride Bulking Agent (For Cake Structure) *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials.
Cyproheptadine Tablets
FreeCYPROHEPTADINE TABLETS IP 4 MG
Each uncoated tablets contains: Cyproheptadine Hydrochloride (- )
Eq to Anhydrous Cyproheptadine hclIP (4 mg)
Excipients (- QS)CYPROHEPTADINE TABLETS BP 4 MG
Each Uncoated Tablet Contains::
Cyproheptadine Hydrochloride Equivalent to Anhydrous Cyproheptadine Hydrochloride BP 4 mg
Excipients – QSCYPROHEPTADINE TABLETS BP 4 MG
Each Sugar Coated Tablet Contains:
Cyproheptadine Hydrochloride Equivalent to Anhydrous Cyproheptadine Hydrochloride BP 4 mg
Colour : Sunset yellow, Titanium Dioxide. –
Excipients – QSUsage: – Relieve the itching of allergic skin conditions, and to treat hives, including hives caused by exposure to cold temperatures and by rubbing the skin
Category: – Anti Allergic Drugs
Therapeutic category: – Antihistamine,Anti Allergic
Cytarabine Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution (Liquid) or Lyophilized Powder.
Active Ingredient Strength Format Cytarabine USP/BP 100 mg / 5 ml (20 mg/ml) Ready-to-use Liquid Cytarabine USP/BP 500 mg / 10 ml (50 mg/ml) Ready-to-use Liquid Cytarabine USP/BP 1000 mg (1 g) Vial Lyophilized Powder or Conc. Liquid Excipients Water for Injection Preservative-Free (Available for IT use) *Pack Sizes: Tray of 1 Vial, 5 Vials, or Box of 1/10 Vials.
Dapsone Tablets
FreeDapsone Tablets IP 50 mg
Each Uncoated tablet contains :
Dapsone IP 50 mgUsage: – Type of skin disorder (dermatitis herpetiformis)
Category: – Anti Fungal Drugs
Therapeutic category: – Anti bacterial
Daunorubicin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in red-capped glass vials.
Active Ingredient Strength Appearance Daunorubicin Hydrochloride USP/BP Equivalent to 20 mg Daunorubicin Red-Orange Lyophilized Cake Excipients Mannitol Bulking Agent *Pack Sizes: Tray of 1 Vial. (Cytotoxic Packaging).
Dexlansoprazole & Domperidone Capsules
FreeDexlansoprazole and Domperidone capsules are a combination medication primarily used for erosive esophagitis and Gastroesophageal Reflux Disease (GERD). This combination reduces stomach acid while also speeding up gastric emptying to prevent reflux.
Diazepam Tablets
FreeDiazepam Tablets IP 5 mg
Each Uncoated tablet contains :
Diazepam IP 5 mg
Excipients q.sDiazepam Tablets BP 5 mg
Each Uncoated tablet contains :
Diazepam BP 5 mgUsage: – Anxiety, alcohol withdrawal, and seizures
Category: – Anti Anxiety Drugs
Therapeutic category: – Anti Anxiety
Diclofenac Potassium Tablets
FreeDiclofenacPotassium Tablets 50 mg
Each Sugarcoated tablet contains:
Diclofenac Potassium BP 50 mgDiclofenac Potassium and Paracetamol Tablets
Each uncoated Tablet contains: Diclofenac Potassium BP 50 mg
Paracetamol BP 650 mg
colour: Tartrazine –
Excipient – QSUsage: – Muscle aches, backaches, dental pain, menstrual cramps, and sports injuries
Category: – Anti Analgesic Painkiller Drugs
Therapeutic category: – NSAID, Arthritis, Painkiller
Diclofenac Sodium & Serratiopeptidase Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Multi-Particulate or Bilayer Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute stability of the sensitive biological enzyme.
Active Ingredient Strength Primary Clinical Function Diclofenac Sodium USP/Ph.Eur. 50 mg Potent peripheral NSAID for sustained localized pain and inflammation blockade. Serratiopeptidase IP/Ph.Eur. 10 mg or 15 mg (Enteric-Coated) Proteolytic enzyme engineered to drain localized post-surgical/traumatic edema. Excipients Methacrylic Acid Copolymer / Microcrystalline Cellulose / Magnesium Stearate Enteric Coating Polymer for Serratiopeptidase / Diluent / Lubricant *Pack Sizes: 10×10 Alu-Alu Blisters (Optimized specifically for 7-to-14 day post-operative or orthopedic recovery regimens).
Diclofenac Sodium Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Amber Glass Ampoules. We offer both the standard 3ml volume and the advanced 1ml (AQ) technology.
Active Ingredient Strength Technology Variant Diclofenac Sodium USP/BP 75 mg / 3 ml Standard (Propylene Glycol Base) Diclofenac Sodium USP/BP 75 mg / 1 ml AQ / Dynapar Generic (Painless, Water Base) Excipients Benzyl Alcohol / Mannitol Preservative / Stabilizer *Pack Sizes: Tray of 5 Ampoules, 10 Ampoules, or 50 Ampoules.
Diclofenac Sodium Paracetamol Serratiopeptidase Tablets
FreeA tablet containing Diclofenac Sodium + Paracetamol + Serratiopeptidase is a triple-action medication formulated to provide comprehensive relief from pain, fever, and inflammation. It is widely used for musculoskeletal disorders, post-surgical recovery, and dental pain.
Diclofenac Sodium Tablets
FreeDiclofenac Sodium Tablets 25 MG Each Filmcoated Tablet contains :
Diclofenac Sodium BP 25 mg Excipient -QSDiclofenac Sodium Tablets BP 50 mg
Each Enteric Coated tablet contains :
Diclofenac Sodium BP 50 mgDiclofenac Sodium Tablets IP 50 mg
Each Enteric coated tablet contains :
Diclofenac Sodium IP 50 mgEach Uncoated tablet contains :
Diclofenac Sodium BP 50 mg
Enteric Coated
Paracetamol BP 500 mgDiclofenac sodium tablets 50 mg
each uncoated tablet contains :
Diclofenac Sodium BP 50 mg
colour -tartrazine In House –
Excipient – In House – QSDiclofenac sodium tablets 50 mg
each uncoated tablet contains :
Diclofenac Sodium BP 50 mg
colour -tartrazine In House –
Excipient – In House – QSDiclofenac sodium tablets 50 mg
each uncoated tablet contains :
Diclofenac Sodium BP 50 mg
colour -tartrazine In House –
Excipient – In House – QSDiclofenac sodium tablets 50 mg
each uncoated tablet contains :
Diclofenac Sodium BP 50 mg
colour -tartrazine In House –
Excipient – In House – QSDiclofenac Sodium Tablets 100 mg
each film coated tablet contains: Diclofenac Sodium BP (100 mg)
colour Titanium Dioxide (In House) (- )
Excipient (In House) (- QS)Usage: – Relieve pain, reduce swelling and ease inflammation
Category: – Anti Analgesic Painkiller Drugs
Therapeutic category: – NSAID, Arthritis, Painkiller
Dicyclomine HCL Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Amber Glass Ampoules.
Active Ingredient Strength Volume Dicyclomine Hydrochloride USP/BP 20 mg 2 ml Ampoule (10 mg/ml) Excipients Sodium Chloride / Water for Injection Isotonic Vehicle *Pack Sizes: Tray of 5 Ampoules, 10 Ampoules, or 50 Ampoules.
Diethyl Carbamazine Tablets
FreeDiethylcarbamazine Citrate Tablets IP 50 mg
Each Uncoated tablet contains :
Diethylcarbamazine Citrate IP 50 mgDiethyl Carbamazine Citrate Tablets IP 100 mg
Each Uncoated Tablet Contains: Diethylcarbamazine Citrate Tablets 100 mg
Usage: – Certain parasitic diseases caused by infection with roundworms
Category: – Anti Hypertensive/ Cardiac drugs
Therapeutic category: – Antiepileptic, Anticonvulsant
Digoxin Tablets
FreeDigoxin Tablets IP 0.25 mg
Each Uncoated tablet contains:
Digoxin IP 0.25 mgDIGOXIN TABLETS IP 62.5 MCG Each uncoated tabelt contiains: digoxin tablets IP 62. 5 mcg excipients q.s.
Usage: – treat heart failure
Category: – Generic drugs and Medicine
Therapeutic category: – Generic Drugs
Diltiazem Tablets
FreeDIL- 60
Diltiazem Hydrochloride Tablets IP 60 mg
Each Filmcoated tablet contains :
Diltiazem Hydrochloride IP 60 mgUsage: – Treat high blood pressure
Category: – Hypertensive/ Cardiac drugs
Therapeutic category: –
Disulfiram Tablets
FreeDisulfiram Tablet IP 500 MG Each uncoated tablets cotains: Diulfiram IP 500 mg Excipient -QS
Usage: – Side effects and safety, interactions
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Doripenem Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in glass vials.
Active Ingredient Strength Primary Use Doripenem Monohydrate USP/BP Equivalent to 500 mg Doripenem Standard Adult Dose Doripenem Monohydrate USP/BP Equivalent to 250 mg Renal Impairment Excipients None (Pure Sterile Powder) – *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials.
Doxepin Hydrochloride Capsules
FreeDoxepin Hydrochloride capsules are tricyclic antidepressants (TCAs) used to treat depression, anxiety, and sleep disorders (insomnia).
Doxycycline 100mg Capsules
FreeDoxycycline 100mg capsules are broad-spectrum tetracycline antibiotics used for bacterial infections, malaria prophylaxis, and severe acne.
Doxycycline Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in amber glass vials.
Active Ingredient Strength Primary Use Doxycycline Hyclate USP/BP Equivalent to 100 mg Doxycycline Standard Adult Dose Excipients Ascorbic Acid / Mannitol Stabilizer / Bulking Agent *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials.
Doxycycline Tablets
FreeDoxycycline Tablets 100 mg
Each Film Coated Tablet Contains:
Doxycycline Hydrochloride IP
eq. to Doxycycline 100 mgDispersible Doxycycline Tablets BP 100 mg
Each Uncoated Tablet Contains:
Doxycycline Monohydrate BP
eq. To Anhydrous Doxycycline 100 mgUsage: – Infections
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic
Drotaverine Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Amber Glass Ampoules.
Active Ingredient Strength Volume Drotaverine Hydrochloride USP/BP 40 mg 2 ml Ampoule (20 mg/ml) Excipients Sodium Metabisulfite Antioxidant *Pack Sizes: Tray of 5 Ampoules, 10 Ampoules, or 50 Ampoules.
Duloxetine Capsules
FreeDuloxetine capsules are delayed-release (gastro-resistant) medications used to treat depression, anxiety, and chronic pain. Because the medicine can be destroyed by stomach acid, it is formulated as acid-resistant pellets inside a hard gelatin capsule.
Duloxetine Tablets
FreeDuloxetine Tablets 20 mg
Each enteric coated tablet contains:
Duloxetine Hydrochloride
eq. To Duloxetine 20 mgUsage: – Depression and anxiety
Category: – Antidepressant drugs
Therapeutic category: – Antidepressant
Dutasteride Capsules
FreeDutasteride capsules are most commonly available as 0.5 mg soft gelatin capsules. They contain the active drug dissolved in a liquid medium to ensure proper absorption
Elevat
FreeThe term Elevat most commonly refers to Elevat 20 Capsule, a prescription antidepressant. However, depending on the specific product name (such as “Elevat D”), it may contain entirely different active ingredients like vitamins
Enalapril Tablets
FreeEnalapril Maleate Tablets IP 2.5 mg
Each Uncoated Tablet Contains:
Enalapril Maleate IP 2.5 mgENALAPRIL MALEATE TABLETS IP 5 MG
Each uncoated Tablet Contains:
Enalapril Maleate IP 5 mg Excipients q.s.Enalapril Maleate tablets IP 10 mg
Each Uncoated Tablet contains :
Enalapril maleate IP 10 mgOELPRIL -5 ENALAPRIL TABLETS BP 5 MG EACH UNCOATED TABLET CONTAINS : ENALAPRIL MALEATE BP 5 MG
Oepril – 10
Enalapril Tablets USP
Each Uncoated tablet contains :
Enalapril Maleate USP 10 mgOepril – 20
Enalapril Maleate Tablets USP
Each Uncoated tablet contains :
Enalapril Maleate USP 20 mg.Usage: – Reduce high blood pressure and to prevent or treat heart failure
Category: – Anti Hypertensive / Cardiac Drugs
Therapeutic category: – Antihypertensive
Enoxaparin Injection
FreeEnoxaparin Sodium injection (brand name Lovenox) is a sterile, clear, colourless to pale yellow aqueous solution of a low molecular weight heparin (LMWH). It is primarily used to prevent and treat deep vein thrombosis (DVT) and pulmonary embolism.
Active IngredientStrengthTherapeutic RoleEnoxaparin Sodium USP/BP40 mg / 0.4 mlProphylaxis (DVT Prevention)Enoxaparin Sodium USP/BP60 mg / 0.6 mlTreatment (Weight Adjusted)Enoxaparin Sodium USP/BP80 mg / 0.8 mlTreatment (DVT/PE/ACS)OriginPorcine MucosaViral Safety Certified
Ergometrine Tablets
FreeErgometrine Tablets IP 250 mcg
Each Sugarcoated Tablet Contains:
Ergometrine Maleate IP 250 mcgUsage: – Postpartum haemorrhage and post abortion hemorrhage in patients with uterine atony
Category: – Generic Drugs and Medicine
Therapeutic category: –
Erythromycin Capsules
FreeErythromycin capsules are macrolide antibiotics used to treat various bacterial infections. Because erythromycin base is easily destroyed by stomach acid, these capsules are typically formulated as delayed-release systems containing enteric-coated pellets
Erythromycin Tablets
FreeErythromycin Stearate Tablets IP 250 mg
Each Film coated tablet contains:
Erythromycin Stearate IP
Eq. To Erythromycin 250 mg
Color: ErythrosineErythromycin Stearate Tablets IP 500 mg
Each Film coated tablet contains:
Erythromycin Stearate IP
Eq. To Erythromycin 500 mg
Excipients q.sElthro – 250
Erythromycin Stearate Tablets IP 250 mg
Each Filmcoated tablet contains:
Erythromycin Stearate IP
Eq. To Erythromycin 250 mgERYTHROMYCIN STEARATE TABLETS IP 500 MG
Each Film Coated Tablet Contains:
Erythromycin Stearate IP (- )
Eq. to Erythromycin (500 mg)
Excipients (- QS)Eritro C
(Erythromycin Stearate Tablets BP)
Each Filmcoated tablet contains :
Erythromycin Stearate BP
Eq. To Erythromycin 500 mgYALLETHRO BP 500 (ERYTHROMYCIN STEAATE TABLETS BP 500 MG) Each film coated tablets contains: Erythromycin stearate eq. To Erythromycin BP 500 mg.
Usage: – Chest infections
Category: – Antibiotic Drugs
Therapeutic category: – Antibiotic
Erythropoietin Prefilled Syringe Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Liquid in a Prefilled Syringe (PFS) with a fine needle.
Active Ingredient Strength (IU) Patient Profile Recombinant Human Erythropoietin 2000 IU / 0.5 ml Maintenance / Low Weight Recombinant Human Erythropoietin 4000 IU / 0.4 ml Standard Dialysis Dose Recombinant Human Erythropoietin 10000 IU / 1.0 ml Chemotherapy / High Dose Stabilizer Human Albumin / Polysorbate Prevents Protein Aggregation *Pack Sizes: Blister Pack of 1 PFS or Box of 6 PFS.
Esomeprazole & Levosulpiride Capsules
FreeEsomeprazole and Levosulpiride capsules are a combination medication primarily used for gastroesophageal reflux disease (GERD) and peptic ulcers in patients who do not respond to a proton pump inhibitor (PPI) alone.
Esomeprazole Capsule
FreeEsomeprazole capsules primarily contain Esomeprazole Magnesium (as a dihydrate or trihydrate) as the active ingredient. Because esomeprazole is “acid-labile” (broken down by stomach acid), these capsules are formulated as delayed-release or gastro-resistant systems containing enteric-coated pellets.
Esomeprazole Domperidone Capsules
FreeEsomeprazole and Domperidone capsules are combination medications containing two active pharmaceutical ingredients, typically formulated as pellets within a hard gelatin capsule.
Esomeprazole Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in glass vials.
Active Ingredient Strength Form Esomeprazole Sodium USP/BP Equivalent to 40 mg Esomeprazole White to Off-White Cake Excipients Disodium Edetate / Sodium Hydroxide Chelating Agent / pH Adjuster *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials.
Esomeprazole Tablets
FreeEsomeprazole Tablets 20 mg
Each Enteric Coated Tablet Contains:
Esomeprazole Magnesium Trihydrate USP
eq. To Esomeprazole 20 mgEsomeprazole Tablets 40 mg
Each Enteric Coated Tablet Contains:
Esomeprazole Magnesium Trihydrate USP
eq. To Esomeprazole 40 mgUsage: – Heartburn, acid reflux and gastro-oesophageal reflux disease
Category: – Antacid & Antiulcer Drugs
Therapeutic category: – Antiulcer, Antacid
Ethambutol Tablets
FreeEthambutol Hydrochloride Tablets IP 200 mg
Each Film Coated Tablet contains:
Ethambutol Hydrochloride IP 200 mgEthambutol tablets IP 400 mg
Each film coated tablet contains :
Ethambutol hydrochloride IP 400 mgEthambutol Hydrochloride Tablets IP 800 mg
Each Film Coated Tablet contains:
Ethambutol Hydrochloride IP 800 mgEthambutol Tablets BP 200 mg
Each Film Coated Tablet Contains :
Ethambutol Hydrochloride BP 200 mgEthambutol Tablets BP 400 mg
Each Film Coated Tablet Contains :
Ethambutol Hydrochloride BP 400 mgEthambutol Tablets BP 800 mg
Each Film Coated Tablet Contains :
Ethambutol Hydrochloride BP 800 mgUsage: – Treat Tuberculosis
Category: – Antibiotic Drugs
Therapeutic category: – Antibiotic, Anti tuberculosis
Ethambutol Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets. The coating is essential as Ethambutol HCl is highly hygroscopic (absorbs moisture) and has a bitter taste.
Active Ingredient Strength (Standard) Role in DOTS Therapy Ethambutol HCl USP/BP 200 mg Pediatric / Renal Adjustment Ethambutol HCl USP/BP 400 mg Standard Adult Unit Ethambutol HCl USP/BP 600 mg / 800 mg High Weight Band / Forte Ethambutol HCl USP/BP 1000 mg High Dose (Special Order) Excipients Q.S. Sorbitol / Magnesium Stearate Ethamsylate tablets
FreeAn Ethamsylate tablet is a hemostatic (antihemorrhagic) medication used to prevent and control excessive bleeding, particularly from small blood vessels (capillaries).
Ethionamide Tablets
FreeEthionamide Tablets IP 125 mg
Each Film Coated Tablet contains:
Ethionamide IP 125 mgEthionamide Tablets IP 250 mg
Each Film Coated Tablet contains:
Ethionamide IP 250 mgETHIONAMIDE TABLETS 250 MG
Each filmcoated tablet contains:
Ethionamide BP 250 mgUsage: – Treat Tuberculosis
Category: – Antibiotic Drugs
Therapeutic category: – Antibiotic, Ant tuberculosis
Etmsylate Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Glass Ampoules.
Active Ingredient Strength Volume Etamsylate USP/BP 250 mg 2 ml Ampoule (125 mg/ml) – Standard Etamsylate USP/BP 125 mg 2 ml Ampoule – Pediatric Excipients Sodium Metabisulfite Antioxidant *Pack Sizes: Tray of 10 Ampoules, 50 Ampoules, or 100 Ampoules.
Etofylline Theophylline Tablets
FreeEt Phylline Retard Plus
Etofylline & Theophylline Tablets
Each Sugar Coated Sustained Release Tablet Contains:
Etofylline BP 231 mg
Theophylline BP 69 mgET Phyllin Retard
Etofylline & Theophylline Tablets
Each Sustained release sugarcoated tablet contains:
Etofylline IP 115 mg
Theophylline IP 35 mgET Phyllin
Etofylline & Theophylline Tablets
Each sugarcoated tablet contains:
Etofylline IP 77 mg
Theophylline IP 23 mgUsage: – Treatment and prevention of asthma and chronic obstructive pulmonary disease
Category: – Anti Allergic Drugs
Therapeutic category: – Antiasthmatic
Etoricoxib Tablets
FreeEtoricoxib tablets 90 mg
Each film coated tablet contains:
Etoricoxib 90 mgUsage: – Pain relief
Category: – Anti Analgesic Painkiller Drugs
Therapeutic category: – NSAID, Analgesic, Generic Drugs
Famotidine Tablets
FreeFamotidine Tablets USP 20 mg
Each Filmcoated tablet contains :
Famotidine USP 20 mgUsage: – Prevent and treat heartburn due to acid indigestion and sour stomach
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Ferrous Folic Tablets
FreeFerrous Sulfate With Folic Acid Tablets
Each Enteric-Coated Tablet Contains:
Dried Ferrous Sulfate IP 150 mg
eq to elemental Iron 45 mg
Folic Acid IP 0.4 mg
Excipent ….Q.s.
Colour : Indigo carmineFerrous Sulfate With Folic Acid Tablets
(IFA large)
Each Enteric-Coated Tablet Contains:
Dried Ferrous Sulfate IP 335 mg
eq to elemental Iron 100 mg
Folic Acid IP 0.5 mg
Excipent q.s
Colour: Red Oxide of IronIron-Folate Tablets
(Ferrous Sulphate With Folic Acid Tablets)
Each film coated tablet contains:
Dried Ferrous Sulphate IP 335 mg
eq. To Elemental Iron 100 mg
Folic Acid IP 0.5 mgFerrous Sulfate With Folic Acid Tablets
(large)
Each Enteric-Coated Tablet Contains:
Dried Ferrous Sulfate IP 335 mg
eq to elemental Iron 100 mg
Folic Acid IP 0.5 mg Colour: Blue Coloured (Indigo Carmine)FERROUS SULPHATE WITH FOLIC ACID ( LARGE) Each enteric coated tablet contains: Dried ferrous sulphate IP 335 mg Equivalent to elemental iron IP 100 mg Folic acid IP 0.5 mg Blue coloured (indigo Carmine) excipients q.s.
Ferrous Sulphate with Folic Acid Tablets
(Pediatric)
Each Film Coated tablet contains:
Dried Ferrous Sulphate IP 67 mg
Eq.to elemental iron 20 mg
Folic Acid IP 100 mcgFerro Force
(Folic Acid & Ferrous Sulphate Tablets)
Each Sugarcoated tablet contains:
Dried Ferrous Sulphate BP 200 mg
Eq.to elemental iron 60 mg
Folic Acid BP 0.25 mgFERROUS SULFATE WITH FOLIC ACID TABLETS
Each Enteric-Coated Tablet Contains:
Dried Ferrous Sulfate IP 335 mg
eq to elemental Iron 100 mg
Folic Acid IP 0.5 mgFESFO- E
(Ferrous Sulphate With Folic Acid Tablets)
Each Enteric Coated Tablet Contains:
Dried Ferrous Sulphate IP 100 mg
eq. To Elemental Iron 30 mg
Folic Acid IP 0.1 mgColour: Indigo Carmine lake
Ferrous Sulfate With Folic Acid Tablets
Each Enteric-Coated Tablet Contains:
Dried Ferrous Sulfate IP 150 mg
eq to elemental Iron 45 mg
Folic Acid IP 0.4 mg
Excipent ….Q.s.
Colour : Indigo carmineFerrous Sulphate with Folic Acid Tablets
(Pediatric)
Each Film Coated tablet contains:
Dried Ferrous Sulphate IP 67 mg
Eq.to elemental iron 20 mg
Folic Acid IP 100 mcgUsage: – Treat or prevent low blood levels of iron
Category: – Generic Drugs and Medicine
Therapeutic category: – Vitamin supplement
Flucloxacillin and Amoxicillin Capsules
FreeFlucloxacillin and amoxicillin combination capsules (often referred to by trade names like Flamox (cosmos-pharm.com) or Amoxam) contain two distinct penicillin-type antibiotics (hra.nhs.uk) that work together to treat a broader range of bacterial infections.
Flucloxacillin Capsules
FreeFlucloxacillin capsules contain flucloxacillin sodium as the active ingredient, along with specific excipients that vary slightly by manufacturer but typically include a lubricant, a glidant, and sodium.
Flucloxacillin Injection
FreeFlucloxacillin injection is a narrow-spectrum penicillin antibiotic used to treat infections caused by staphylococci. It is typically supplied as a white to off-white sterile powder for reconstitution.
Fluconazole Capsules
FreeFluconazole capsules (commonly known by the brand name Diflucan (pfizermedical.com)) contain the active antifungal ingredient fluoxetine hydrochloride along with a powder fill of inactive excipients and a hard gelatin shell.
Fluconazole Tablets
FreeFLUCONAZOLE TABLETS IP 150 MG
Each uncoated tablet contains:
Fluconazole IP 150 mg Excipients q.s.Nuflucon – 200
Fluconazole Tablets IP 200 mg
Each Uncoated Tablet contains :
Fluconazole IP 200 mgUsage: – Prevent and treat a variety of fungal and yeast infections
Category: – Antifungal Drugs
Therapeutic category: – Antifungal
Fludac
FreeFludac is a brand name for capsules manufactured by Cadila Pharmaceuticals Ltd (amazon.in), used primarily as an antidepressant. It belongs to the class of Selective Serotonin Reuptake Inhibitors (SSRIs) (1mg.com).
Fluoxetine Capsules
FreeFluoxetine capsules, commonly known by the brand name Prozac (accessdata.fda.gov), contain the active antidepressant fluoxetine hydrochloride along with several inactive ingredients that vary by manufacturer and dosage strength.
Fluphenazine Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Oily Solution in Amber Glass Ampoules or Vials.
Active Ingredient Strength Type Fluphenazine Decanoate USP/BP 25 mg 1 ml Ampoule / Vial Fluphenazine Decanoate USP/BP 12.5 mg 0.5 ml Ampoule (Starter Dose) Vehicle Sesame Oil Depot Base Preservative Benzyl Alcohol 1.5% v/v (In Vials) *Pack Sizes: Tray of 5 Ampoules, 10 Ampoules, or 10 ml Multi-Dose Vial.
Folic Acid, Niacinamide Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated or Sugar Coated Tablets (typically Yellow or White). The coating protects the photosensitive vitamins from degradation.
Active Ingredient Strength (Standard) Therapeutic Role Folic Acid USP/BP 1.5 mg / 5 mg DNA Repair / Anemia Prevention Niacinamide USP/BP 20 mg / 50 mg / 100 mg Metabolic Support / Skin Barrier Excipients Q.S. Dibasic Calcium Phosphate / Starch Filler / Disintegrant Fortified Procaine Peniciilin Injection with Streptomycin
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials.
Active Ingredient Strength Role Procaine Penicillin G 300,000 IU Sustained Release (24 Hours) Benzyl Penicillin Sodium 100,000 IU Rapid Onset (Peak Levels) Streptomycin Sulfate Equivalent to 0.5 g (500 mg) Base Gram-Negative Coverage Total Formulation 400,000 IU + 0.5 g Standard Adult Dose *Pack Sizes: Tray of 50 Vials or 100 Vials.
Fortified Procaine Penicillin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials.
Active Ingredient Proportion Role Procaine Penicillin G 300,000 IU (75%) Long Acting (Depot) Benzyl Penicillin Sodium 100,000 IU (25%) Rapid Acting (Peak) Total Strength 400,000 IU (4 Lac) Standard Adult Dose Total Strength 4,000,000 IU (40 Lac) High Dose / Veterinary *Pack Sizes: Tray of 50 Vials or 100 Vials.
Frusemide Tablets
FreeFrusemide Tablets IP 40 mg
Each Uncoated tablet contains:
Frusemide IP 40 mgFurosemide Tablets
Frusemide Tablets BP 40 mg
Each Uncoated tablet contains:
Frusemide BP 40 mgUsage: – High blood pressure, heart failure and edema
Category: – Diuretic drugs
Therapeutic category: – Diuretic
Furosemide Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Amber Glass Ampoules.
Active Ingredient Strength Volume Furosemide USP/BP 20 mg 2 ml Ampoule (10 mg/ml) – Standard Furosemide USP/BP 40 mg / 4 ml 4 ml Ampoule – High Dose Furosemide USP/BP 250 mg / 25 ml 25 ml Vial – Renal Failure Excipients Sodium Hydroxide / NaCl pH Adjuster / Isotonicity *Pack Sizes: Tray of 10 Ampoules, 50 Ampoules, or 100 Ampoules.
Gabapentin Capsules
FreeGabapentin capsules (commonly 100 mg, 300 mg, and 400 mg) contain the active pharmaceutical ingredient gabapentin USP, along with a variety of inactive excipients that aid in the drug’s delivery and stability.
Gentamicin Injection
FreeGentamicin injection (brand name Garamycin) is a sterile, clear, colourless to slightly yellow aqueous solution of an aminoglycoside antibiotic. It is used to treat serious gram-negative bacterial infections.
Glibenclamide & Metformin Hydrochloride Tablets
FreeProduct Composition & Standard Strengths
We supply this product in standardized Fixed-Dose Combinations (FDCs) designed for flexible clinical titration.
Active Ingredient Standard Strengths (per Tablet) Primary Clinical Function Metformin HCl 400 mg / 500 mg The Foundation: Reduces glucose production. Glibenclamide (Glyburide) 2.5 mg / 5 mg The Stimulator: Enhances insulin secretion. Excipients Pharma Grade The Matrix: Ensures stability of the FDC. Glibenclamide Tablets
FreeGlibenclamide Tablets IP 5 mg Each Uncoated tablet contains: Glibenclamide IP 5 mg
Excipients – QSGLIBENCLAMIDE & METFORMIN HYDROCHLORIDE SR TABLETS
Each sustained release tablet contains:: – Glibenclamide 5 mg
– Metformin Hydrochloride 500 mg
– Excipients – QSGilben
Glibenclamide Tablets BP 5 mg
Each Uncoated tablet contains :
Glibenclamide BP 5 mgTrippleace Glibenclamide – 5
Glibenclamide Tablets BP 5 mg
Each Uncoated tablet contains :
Glibenclamide BP 5 mgUsage: – Along with diet and exercise to improve blood sugar control
Category: – Anti Diabetic drugs
Therapeutic category: – Anti Diabetic
Gliclazide Tablets
FreeGLICAZIDE TABLETS BP 80 mg Each Uncoated tablet contains : GLICLAZIDE BP 80 mg
Usage: – Type 2 diabetes is an illness
Category: – Anti Diabetic drugs
Therapeutic category: – Anti Diabetic
Glimepiride Tablets
FreeGlimepiride Tablets IP 1 mg
Each Filmcoated tablet contains :
Glimepiride IP 1 mgGlimepiride Tablets IP 2 mg
Each Filmcoated tablet contains :
Glimepiride IP 2 mgGlimepiride Tablets 1 mg
Each Filmcoated tablet contains :
Glimepiride BP 1 mgGlimepiride Tablets 2 mg
Each Filmcoated tablet contains :
Glimepiride BP 2 mgGlimepiride Tablets 4 mg
Each Film Coated Tablet Contains:
Glimepiride BP 4 mgSMEGLIP -1 / GLIMEPIRIDE TABLETS BP 1 MG Each Filmcoated Tablet Contains: Glimepiride BP 1 mg
Excipients – QSSMEGLIP -2 / GLIMEPIRIDE TABLETS BP 2 MG Each Filmcoated Tablets Contains: Glimepiride BP 2 mg
Excipients – QSGLIMEPIRIDE,PIOGLITAZONE & METMIN HYDROCHLORIDE SR TABLET
Each Sustained Release Tablet Contains :
Glimepiride 2 mg
Pioglitazone 15 mg
Metmin Hydrochloride 500 mg
Excipients q.sUsage: – Type 2 diabetes is a condition where the body doesn’t make enough insulin, or the insulin that it makes doesn’t work properly
Category: – Anti Diabetic drugs
Therapeutic category: – Anti Diabetic
Glimepiride, Pioglitazone & Metformin Hydrochloride Tablets
FreeProduct Composition & Standard Strengths
We supply this product in standardized Fixed-Dose Combinations (FDCs) designed for flexible clinical titration.
Active Ingredient Common Strength Primary Clinical Function Metformin HCl (SR/IR) 500 mg / 1000 mg The Foundation: Reduces glucose production. Glimepiride 1 mg / 2 mg The Stimulator: Enhances insulin secretion. Pioglitazone 15 mg The Sensitizer: Fixes insulin resistance. Glipizide Tablets
FreeGlipizide Tablet IP
Each uncoated Tablet contains :
Glipizide IP 5 mgGynoglid Tablets
Glipizide Tablets BP 5 mg
Each Uncoated tablet contains :
Glipizide BP 5 mgUsage: – Lowers blood sugar by causing the pancreas to produce insulin
Category: – Anti Diabetic drugs
Therapeutic category: – Anti Diabetic
Glucosamine Capsule
FreeGlucosamine capsules typically contain one or more forms of glucosamine, an amino sugar essential for building cartilage and joint tissues. These active ingredients are often derived from the shells of shellfish (crabs, shrimp, and lobsters) or produced from vegetarian sources like fermented corn or fungi.
Glucosamine Sulphate Chondroitin Sulphate Tablets
FreeGlucosamine Sulphate And Chondroitin Sulphate Tablets
Each Film Coated Tablet Contains:
Glucosamine Sulphate 500 mg
added as Glucosamine Sulphate Sodium Chloride
Chondroitin Sulphate 400 mg
added as Chondroitin Sulphate SodiumUsage: – slow or prevent the degeneration of joint cartilage
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Glyceryl Trinitrate Tablets
FreeGLYCERYL TRINITRATE TABLETS IP 0.5 MG
Each sublingual tablets contains: Dilute Nitroglycerin IP Equivalent to Nitroglycerin 0.5 mg
Excipients q.s.Usage: – Treat angina (chest pain)
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic
Griseofulvin Tablets
FreeGriseofulvin Tablets IP 125 mg
Each Uncoated tablet contains :
Griseofulvin IP 125 mg
Excipients q.sGRIDORN – 125
(Griseofulvin Tablets IP 125 mg)
Each Uncoated tablet contains :
Griseofulvin IP 125 mgGRISEOFULVIN TABLETS IP 250 MG Each uncoated tablet contains : Griseofulvin IP 250 mg Excipients q.s.
Griseofulvin Tablets IP 500 mg
Each Uncoated tablet contains :
Griseofulvin IP 500 mgGRIDORN-125
(Griseofulvin tablets BP 125 mg)
Each uncoated tablet contains :
Griseofulvin BP 125 mgUsage: – Treat skin infections such as jock itch, athlete’s foot, and ringworm
Category: – Antifungal Drugs
Therapeutic category: – Antifungal
Haematinic Capsules
FreeHaematinic capsules are combination supplements containing essential nutrients required for the production of healthy red blood cells and haemoglobin. While specific formulations vary by brand, they typically contain the following categories of ingredients:
Haloperidol Tablets
FreeHaloperidol Tablets BP 1.5 mg
Each Uncoated tablet contains :
Haloperidol BP 1.5 mgH PERIDOL-2
Haloperidol Tablets BP 2 mg
Each Uncoated tablet contains :
Haloperidol BP 2 mgHaloperidol Tablets BP 5 mg
Each Uncoated tablet contains :
Haloperidol BP 5 mgHaloperidol Tab BP 5 mg Haloperidol BP 5 mg Pantone Excipent QS
Haloperidol Tablets BP 5 mg
Each uncoated tablet contains:
Haloperidol B.P. 5 mg.Usage: – Treat certain mental/mood disorders
Category: – Antipsychotic / CNS Drugs
Therapeutic category: –
Heparin Sodium Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Glass Vials.
Active Ingredient Strength Volume Heparin Sodium USP/BP 5,000 IU (1,000 IU/ml) 5 ml Multi-Dose Vial Heparin Sodium USP/BP 25,000 IU (5,000 IU/ml) 5 ml Multi-Dose Vial Preservative Benzyl Alcohol 0.95% v/v *Available in preservative-free versions for neonatal use upon request.
Hucog
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection accompanied by a solvent (Sodium Chloride Injection).
Brand Reference Strength Primary Use Hucog / Corion 2000 IU Pediatric Cryptorchidism / Male Fertility Hucog / Corion 5000 IU Standard Ovulation Trigger Hucog / Corion 10000 IU IVF / Assisted Reproductive Technology (ART) Solvent 0.9% NaCl 1 ml or 2 ml Ampoule *Pack Sizes: Single Vial with Ampoule or Tray of 10.
Hydrochlorothiazide Tablets
FreeHydrochlorothiazide Tablets IP 50 mg
Each Uncoated Tablet contains :
Hydrochlorothiazide IP 50 mgHydrochlorothiazide Tabtes BP 25 mg Each uncoated tablet contains : Hydrochlorothiazide BP 25 mg , Excipients q.s.
Hydrochlorothiazide tablets USP
Each Uncoated tablet contains :
Hydrochlorothiazide USP 25 mgHydrochlorothiazide Tablets USP 50 mg
Each Uncoated Tablet contains :
Hydrochlorothiazide USP 50 mgUsage: – Treat edema
Category: – Diuretic Drugs
Therapeutic category: – Diuretic
Hydrocortisone Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in glass vials.
Active Ingredient Strength Primary Use Hydrocortisone Sodium Succinate USP/BP Equivalent to 100 mg Hydrocortisone Standard Adult / Pediatric Emergency Hydrocortisone Sodium Succinate USP/BP Equivalent to 250 mg Severe Asthma / Sepsis Pulse Hydrocortisone Sodium Succinate USP/BP Equivalent to 500 mg High-Dose ICU Protocol Excipients Sodium Phosphate / Sodium Hydroxide Buffer / pH Adjuster *Pack Sizes: Tray of 1 Vial + 1 Ampoule (WFI), 10 Vials, or 50 Vials.
Product Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in glass vials.
Active Ingredient Strength Primary Use Hydrocortisone Sodium Succinate USP/BP Equivalent to 100 mg Hydrocortisone Standard Adult / Pediatric Emergency Hydrocortisone Sodium Succinate USP/BP Equivalent to 250 mg Severe Asthma / Sepsis Pulse Hydrocortisone Sodium Succinate USP/BP Equivalent to 500 mg High-Dose ICU Protocol Excipients Sodium Phosphate / Sodium Hydroxide Buffer / pH Adjuster *Pack Sizes: Tray of 1 Vial + 1 Ampoule (WFI), 10 Vials, or 50 Vials.
Hyoscine Butylbromide Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Glass Ampoules.
Active Ingredient Strength Volume Hyoscine Butylbromide USP/BP 20 mg 1 ml Ampoule (Standard Dose) Excipients Sodium Chloride / Water for Injection Isotonic Solution *Pack Sizes: Tray of 5 Ampoules, 10 Ampoules, or 100 Ampoules.
Hyoscine Butylbromide Tablets
FreeProduct Composition & Strength
We supply this product as Sugar Coated Tablets or Film Coated Tablets. The coating is essential to mask the naturally bitter taste of the active ingredient and ensure patient compliance.
Active Ingredient Strength (Standard) Therapeutic Role Hyoscine Butylbromide BP/USP 10 mg Muscarinic Antagonist (Antispasmodic) Excipients Q.S. Calcium Hydrogen Phosphate / Maize Starch Stabilizing Core Matrix Coating Agent Q.S. Sucrose / Talc / Acacia Traditional Sugar Coating Ibuprofen & Paracetamol Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets. The coating protects the gastric mucosa from direct contact with Ibuprofen and masks the bitter taste of the active ingredients.
Active Ingredient Strength (Standard) Therapeutic Role Ibuprofen BP/USP 200 mg NSAID (Anti-inflammatory/Analgesic) Paracetamol BP/USP 325 mg Analgesic & Antipyretic Excipients Q.S. Starch / Croscarmellose Sodium Disintegrant & Binder Ibuprofen Solution For Infusion
FreeProduct Composition & Strength
We supply this product in two formats: Ready-to-Use Infusion and Concentrate for Dilution.
Active Ingredient Strength Format Ibuprofen (with Arginine) 400 mg / 100 ml Ready-to-Use (IV Bag/Bottle) Ibuprofen (with Arginine) 400 mg / 4 ml Concentrate (Vial – Must Dilute) Solubilizer L-Arginine Ensures Water Solubility *Pack Sizes: 100 ml Bottle (Single) or Tray of 10 Vials.
Ibuprofen Tablets
FreeIbuprofen Tablets IP 200 mg
Each Sugar Coated tablet contains :
Ibuprofen IP 200 mgIbuprofen Tablets IP 400 mg
Each Sugar Coated tablet contains :
Ibuprofen IP 400 mgIbupramol
Ibuprofen & Paracetamol Tablets
Each Uncoated tablet contins :
Ibuprofen IP 200 mg
Paracetamol IP 325 mgIBUPROFEN TABLETS IP 400 MG Each Filmcoated Tablet contains: – Ibuprofen IP 400 mg
– Excipients – QSMakupar – 200
Ibuprofen Tablets BP
Each Sugarcoated Tablet contains :
Ibuprofen BP 200 mgMakupar – 400
Ibuprofen Tablets BP
Each Sugarcoated Tablet contains :
Ibuprofen BP 400 mgIbu – 600
Ibuprofen Tablets BP
Each Filmcoated tablet contains :
Ibuprofen BP 600 mgIBUPROFEN TABLETS BP 400 MG
Each Filmcoated Tablets Contains:
Ibuprofen BP 400 mg
– Erythrosine –
– Excipients – QSUsage: – Painkiller
Category: – Anti Analgesic Painkiller Drugs
Therapeutic category: – NSAID, Analgesic, Antipyretic, Anti Inflammatory
Ibuprofen with Paracetamol Tablets
FreeProduct Composition & Standard Strengths
We supply this product in standardized Fixed-Dose Combinations (FDCs) designed for maximum therapeutic synergy and patient safety.
Active Ingredient Standard Strengths (per Tablet) Primary Clinical Function Ibuprofen IP/BP/USP 200 mg / 400 mg The Anti-Inflammatory: Reduces physical swelling. Paracetamol IP/BP/USP 325 mg / 500 mg The Analgesic: Raises the chemical pain threshold. Excipients Pharma Grade The Matrix: Ensures stability and rapid release. Imatinib Capsules
FreeImatinib capsules contain the active oncology drug and a specific set of inactive ingredients designed to stabilize the compound for oral delivery.
Imidapril Hydrochloride Tablets
FreeIMAPRIL TABLETS 5 MG
IMIDAPRIL HYDROCHLORIDE 5 MG
EACH FILM COATED TABLET CONTAINS :
IMIDAPRIL HYDROCHLORIDE 5 MGIMAPRIL TABLETS 10 MG
IMIDAPRIL HYDROCHLORIDE 10 MG
EACH FILM COATED TABLET CONTAINS :
IMIDAPRIL HYDROCHLORIDE 10 MGUsage: – reduce high blood pressure
Category: – Hypertensive / Cardiac Drugs
Therapeutic category: – Hypertensive
Imipenem & Cilastatin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Buffered Lyophilized Powder for Injection in glass vials.
Active Ingredient Strength Role Imipenem USP/BP 500 mg Antibiotic (Carbapenem) Cilastatin Sodium USP/BP Equivalent to 500 mg Cilastatin DHP-I Inhibitor Buffer Sodium Bicarbonate pH Stabilizer *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials.
Imipramine Tablets
FreeFraneel 10 Tablets
Imipramine tablets BP 10 mg
Each Sugar Coated tablet contains :
Imipramine Hydrochloride BP 10 mgFRANEEL 25 MG IMIPRAMINE TABLET 25 MG
EACH SUGAR COATED TABLET CONTAINS: IMIPRAMINE HYDROCHLORIDE BP 25 MG EXCIPIENTS- QSUsage: – Treat depression
Category: – Antidepressant drugs
Iron Folic Acid Tablets
FreeIRON WITH FOLIC ACID TABLETS IP
Each Enteric Coated Tablet Contains: –
Dried Ferrous Sulphate 335 mg
Eq. to Elemental iron IP (100 mg)
Folic Acid IP (0.5 mg)
Indigo Caramine (- )
Excipients (- )Iron with Folic acid Tablets IP
Each sugarcoated tablet contains:Ferrous Sulphate IP (200 mg)
Eq. to Elemental Iron IP (60 mg)
Folic Acid IP (0.5 mg)
Ponceau 4R (In House) (- )
Excipients (- QS)Usage: – Treat or prevent low iron in the body
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs / Medicine
Isoniazid & Pyridoxine Hydrochloride Tablets
FreeProduct Composition & Strength
We supply this product as Uncoated or Film Coated Tablets. The ratio of Isoniazid to Pyridoxine is carefully calibrated to meet international clinical guidelines for neuroprotection.
Active Ingredient Strength (Standard) Therapeutic Role Isoniazid BP/USP 300 mg Primary Antitubercular (Bactericidal) Pyridoxine Hydrochloride BP/USP 10 mg / 25 mg / 50 mg Neuroprotective Vitamin (B6) Excipients Q.S. Starch / Lactose / Magnesium Stearate Tablet Core Matrix Isoniazid Tablets
FreeIsoniazide Tablets IP
Each Coated Tablet Contains :
Isoniazide IP 50 mgIsoniazid Tablet IP 100 mg
Each Coated Tablets Contains :
Isoniazid IP 100 mgIsoniazid Tablet IP 300 mg
Each Coated Tablets Contains :
Isoniazid IP 300 mgPYRINH ISONIAZIDE WITH PYRIDOXINE TABLETS Each Uncoated tablet contain: – Isoniazide IP 100 MG
– Pyridoxin Hyrochloride IP 5 MGISONIAZID TABLETS BP 100 MG
Each Film Coated Tablet contains:
Isoniazid BP 100 mgISONIAZID TABLETS BP 300 MG
Each filmcoated tablet contains:
Isoniazid BP 300 mgUsage: – Treat active tuberculosis (TB) infections
Category: – Anti tuberculosis drugs
Therapeutic category: –
Isosorbide Mononitrate Tablets
FreeProduct Composition & Strength
We supply this product as Immediate Release Tablets (20 mg). We also offer Extended Release (ER/SR) variants for once-daily dosing through our contract manufacturing division.
Active Ingredient Strength (Standard) Therapeutic Role Isosorbide Mononitrate BP/USP 20 mg Organic Nitrate / Vasodilator Excipients Q.S. Microcrystalline Cellulose / Lactose Stabilizing Matrix Isosorbide Tablets
FreeIsosorbide Dinitrate Tablets IP 5 mg
Each Uncoated tablet contains :
Diluted Isosorbide Dinitrate IP
Eq. To Isosorbide Dinitrate 5 mgIsosorbide Dinitrate Tablets IP 10 mg
Each Uncoated tablet contains :
Diluted Isosorbide Dinitrate IP
Eq.To Isosobide Dinitrate 10 mgIsosorbide Dinitrate Tablets BP 5 mg
Each uncoated tablet contains:
Diluted Isosorbide Dinitrate BP
Eq. to Isosorbide Dinitrate 5 mgIsosorbide Mononitrate Tablets 10 mg
Each Uncoated tablet contains :
Diluted Isosorbide Mononitrate
Eq.To Isosobide mononitrate 10 mgM- Trate tablets
Isosorbide 5 -Mononitrate Tablet BP
Each Uncoated tablet contains :
Diluted Isosorbide 5- Mononitate BP
eq to Isosorbide 5- Mononitate 20 mgUsage: – Prevent chest pain caused by angina
Category: – Anti Cardiovascular drugs
Therapeutic category: – Cardiovascular Agent
Isosorbide Tablets
FreeProduct Composition & Variants
We supply both Mononitrate and Dinitrate forms in immediate and sustained-release formats.
Variant Type Standard Strengths Primary Clinical Use Isosorbide Mononitrate 10mg, 20mg, 40mg Prophylaxis: Long-term prevention of angina. Isosorbide Dinitrate 5mg, 10mg, 20mg Acute & Chronic: Short and long-term heart failure. Sustained Release (SR) 30mg, 60mg Once-Daily: For improved patient compliance. Isotretinoin Capsules
FreeProduct Composition & Strengths
We supply this product as Liquid-Filled Soft Gelatin Capsules, optimized for maximum systemic absorption.
Active Ingredient Strength (per Softgel) Primary Clinical Target Isotretinoin IP/BP/USP 10 mg Standard Starting Dose: For mild-to-moderate systemic treatment. Isotretinoin IP/BP/USP 20 mg Therapeutic Dose: For severe, cystic, or conglobate acne. Excipients Soybean Oil / Beeswax The Vehicle: Ensures lipid-solubility for better absorption. Itopride Tablets
FreeItopride Tablets
Each Film Coated Tablet Contains:
Itopride Hydrochloride 50 mgUsage: – treatment of heartburn and nausea
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Itraconazole Capsules
FreeItraconazole capsules contain a combination of the active antifungal drug and specialized inactive ingredients designed to overcome the medication’s poor solubility in water.
Ivermectin Tablets
FreeIvermectin Tablets 3 mg
Each tablet contains :
Ivermectin BP 3 mgIvermectin Tablets 6 mg
Each Uncoated tablet contains :
Ivermectin IP 6 mgUsage: – Parasitic infections
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic, Antiparasitic
Ketoconazole Tablets
FreeKetoconazole Tablets IP
Each Uncoated Tablet contains :
Ketoconazole IP 200 mgKetoconazole Tablets USP
Each Uncoated Tablet contains :
Ketoconazole USP 200 mgUsage: – Some kinds of fungal infections
Category: – Antifungal drugs
Therapeutic category: – Antifungal
Ketorolac Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Amber Glass Ampoules.
Active Ingredient Strength Volume Ketorolac Tromethamine USP/BP 30 mg 1 ml Ampoule (Standard Dose) Ketorolac Tromethamine USP/BP 60 mg 2 ml Ampoule (High Dose) Excipients Alcohol USP / Sodium Chloride Solubilizer / Isotonicity *Pack Sizes: Tray of 5 Ampoules, 10 Ampoules, or 50 Ampoules.
L-Ornithine-L-Aspartate Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Hypertonic Solution in Glass Ampoules. (Must be diluted).
Active Ingredient Strength Concentration L-Ornithine-L-Aspartate 5 g (5000 mg) 500 mg/ml (10 ml Ampoule) Excipients Water for Injection – *Pack Sizes: Tray of 5 Ampoules or 10 Ampoules.
Lamotrigine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet or Rapid-Dispersible (DT) Tablet, packed exclusively in highly secure Alu-PVC or Alu-Alu blister strips, frequently customized into highly regulated Titration Starter Kits.
Active Ingredient Strength Primary Clinical Function Lamotrigine USP/Ph.Eur. 25 mg (Starter Kit) The SJS-Prevention Standard: Mandatory base unit for the strict, slow titration schedule required to prevent fatal cutaneous reactions. Often formulated as a Dispersible Tablet (DT) for pediatric epilepsy. Lamotrigine USP/Ph.Eur. 50 mg / 100 mg / 200 mg Global Maintenance Standard: High-efficacy adult maintenance doses for lifelong seizure control and bipolar stabilization. Excipients Microcrystalline Cellulose / Sodium Starch Glycolate / Povidone / Sucralose & Flavoring (If DT) / Magnesium Stearate Diluent / Superdisintegrant / Binder / Sweetener (Engineered specifically to either swallow intact or disperse rapidly in a teaspoon of water for children or elderly psychiatric patients suffering from dysphagia) *Pack Sizes: 10×10 Blisters, Bottles of 100, or Dedicated 28-Day Titration Calendar Packs (Optimized specifically to eliminate caregiver dosing errors during the critical initiation phase).
Lansoprazole Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in glass vials.
Active Ingredient Strength Form Lansoprazole Sodium USP/BP Equivalent to 30 mg Lansoprazole White Lyophilized Cake Excipients Mannitol / Meglumine / Sodium Hydroxide Bulking Agent / pH Stabilizer *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials.
Leflunomide Tablets
FreeLeflunomide Tablets
Each Film Coated Tablet Contains:
Leflunomide BP 20 mgUsage: – Rheumatoid arthritis
Category: – Antiarrhytmics drugs
Therapeutic category: –
Lenalidomide Capsules
FreeLenalidomide capsules (brand name Revlimid) are a potent immunomodulatory medication used to treat certain types of cancers, such as multiple myeloma and myelodysplastic syndromes.
Levamisole Tablets
FreeJantrax
Levamisole Hydrochloride Tablets USP
Each Filmcoated tablet contains :
Levamisole Hydrochloride USP
Eq. To Levamisole 40 mgUsage: – Treat infections of the worm
Category: – Anthelmintic drugs
Therapeutic category: –
Levetiracetam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile, Clear, Colorless Solution in glass vials.
Active Ingredient Strength Concentration Levetiracetam USP/BP 500 mg 100 mg/ml (in 5 ml Vial) Excipients Sodium Acetate / Glacial Acetic Acid Buffer / pH Adjuster (5.5) *Pack Sizes: Tray of 5 Vials, 10 Vials, or Single Vial packs.
Levocetirizine Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets. The small, easy-to-swallow tablets are engineered for rapid disintegration to facilitate quick relief from acute allergic flares.
Active Ingredient Strength (Standard) Therapeutic Role Levocetirizine Dihydrochloride BP/USP 5 mg Potent H1-Receptor Antagonist Excipients Q.S. Microcrystalline Cellulose / Lactose Diluent & Disintegrant Coating Agent Q.S. Opadry® White Protective Film Coating Levofloxacin Tablets
FreeLevofloxacin tablets IP 250 mg
Each Filmcoated tablet contains :
Levofloxacin Hemihydrate IP
Eq. To Levofloxacin 250 mgLevofloxacin tablets IP 500 mg
Each Filmcoated tablet contains :
Levofloxacin Hemihydrate IP
Eq. To Levofloxacin 500 mgLevofloxacin Tablets 750 mg
Each Film Coated Tablet Contains :
Levofloxacin Hemihydrate
Eq To Levofloxacin 750 mgLevofloxacin tablets 250 mg
Each Filmcoated tablet contains :
Levofloxacin Hemihydrate
Eq. To Levofloxacin 250 mgLevofloxacin tablets 500 mg
Each Filmcoated tablet contains :
Levofloxacin Hemihydrate
Eq. To Levofloxacin 500 mgUsage: – Variety of bacterial infections
Category: – Antibiotic Drugs
Therapeutic category: – Antibiotic
Levothyroxine Tablets
FreeThyrostim Tablets
Levothyroxine Sodium Tablets BP
(Thyroxine Sodium tablets BP)
Each Uncoated tablet contains :
Levothyroxine Sodium BP
Eq. To Anhydrous Levothyroxine Sodium 0.1 mgTHYROXINE SODIUM TABLETS BP (LEVOTHYROXINE SODIUM TABLETS BP 0.25 MG)
EACH UNCOATED TABLET CONTAINS:
LEVOTHYROXINE SODIUM EQ. TO ANHYDROUS LEVOTHYROXINE SODIUM BP 0.25 MG
EXCIPIENTS Q.S.
Levothyroxine Sodium Tablets IP 50 MCG Each Uncoated tablet contains:Levothyroxine Sodium IP Eq. to Anhydrous Levothyroxine Sodium IP (50 mcg)
EXCIPENT (- QS)
THYROXINE 25 MCG
EACH UNCOATED TABLET CONTAINS:
LEVOTHYROXINE SODIUM EQ TO ANHYDROUS LEVOTHYROXINE SODIUM IP 25 MCGLevothyroxine Tablets IP 25 mcg Each uncoated tablet contains:
LEVOTHYROXINE SODIUM EQ TO ANHYDROUS LEVOTHYROXINE SODIUM IP (25 mcg)
Excipient (- QS)Levothyroxine Sodium Tablets IP 50 MCG Each Uncoated tablet contains: Levothyroxine Sodium IP Eq. to Anhydrous Levothyroxine Sodium IP (50 mcg)
EXCIPENT (- QS)
Usage: – Treat hypothyroidismCategory: – Thyroid Hormones drugs
Therapeutic category: – Thyroid Hormones
Lidocaine 2% Ampoules
FreeProduct Composition & Strength
We supply this product as a Sterile, Preservative-Free Solution in glass ampoules.
Active Ingredient Strength Total Content (2ml) Lidocaine Hydrochloride USP/BP 2% (20 mg/ml) 40 mg Total Content (5ml) 2% (20 mg/ml) 100 mg Excipients Sodium Chloride / Water for Injection Isotonic Solution *Available in 2ml, 5ml, and 10ml Type I Glass Ampoules.
Lidocaine Cartridges
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Type I Glass Cartridges.
Active Ingredients Strength Cartridge Volume Lidocaine Hydrochloride 2% (20 mg/ml) 1.8 ml / 2.2 ml Adrenaline (Epinephrine) 1:80,000 / 1:100,000 – Stabilizer Sodium Metabisulfite Antioxidant *Pack Sizes: Box of 50 or 100 Cartridges (Blister packed for sterility).
Lidocaine Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Glass Ampoules or Multi-dose Vials.
Active Ingredient Strength Common Application Lidocaine Hydrochloride 1% (10 mg/ml) Infiltration / Minor Suturing / Diagnostic Blocks Lidocaine Hydrochloride 2% (20 mg/ml) Major Nerve Blocks / Epidural / Anti-arrhythmic Excipients Sodium Chloride / Methylparaben (in vials) Isotonicity / Preservative *Available in preservative-free (PF) versions for spinal/epidural use.
Lignocaine Adrenaline Dental Cartridges
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Glass Cartridges with siliconized plungers.
Active Ingredients Strength Standard Cartridge Volume Lignocaine Hydrochloride 2% (20 mg/ml) 1.8 ml (Standard) Adrenaline (Epinephrine) 1:80,000 / 1:100,000 2.2 ml (Extended) Excipients Sodium Metabisulfite / NaCl Antioxidant / Isotonicity *Pack Sizes: Box of 50 Cartridges or 100 Cartridges (Blister packed).
Lignocaine Injection
FreeLignocaine injection (also known as Lidocaine) is a sterile, aqueous solution primarily used as a local anaesthetic and antiarrhythmic agent. Its composition varies depending on whether it is a single-dose or multi-dose formulation and whether it includes a vasoconstrictor.
Lincomycin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Glass Ampoules or Vials.
Active Ingredient Strength Volume Lincomycin Hydrochloride USP/BP 600 mg 2 ml Ampoule (300 mg/ml) – Standard Lincomycin Hydrochloride USP/BP 300 mg 1 ml Ampoule (300 mg/ml) – Pediatric Preservative Benzyl Alcohol 0.945% v/v *Pack Sizes: Tray of 10 Ampoules, 50 Ampoules, or 100 Ampoules.
Linezolide Tablets
FreeLinezolide Tablets
Each Film Coated Tablet Contains
Linezolide 600 mgUsage: – Treat different types of bacterial infections
Category: – Antibiotic drugs
Therapeutic category: – Antibiotics
Lisinopril Tablets
FreeLisinopril Tablets IP 2.5 mg
Each Film coated tablet contains:
Lisinopril IP 2.5 mgLisinopril Tablets IP 5 mg
Each Film coated tablet contains:
Lisinopril IP 5 mgLowpril
Lisinopril Tablet BP
Each Film Coated Tablet Contains:
Lisinopril Dihydrate BP
eq. To Lisinopril Anhydrous 20 mg
Usage: – Treat high blood pressure and heart failureCategory: – Anti Hypertensive / Cardiac Drugs
Loperamide Tablets
FreeLoperamide Tablets
Each Film Coated Tablet Contains:
Loperamide Hydrochloride USP 2 mg
Usage: – Treat diarrhoea (runny poo)Category: – Antidiarrheal drugs
Therapeutic category: – Antidiarrheal
Loratidine Tablets
FreeLotadin
Loratidine Tablets
Each Uncoated tablet contains :
Loratidine 10 mgUsage: – Temporarily relieve the symptoms of hay fever
Category: – Anti Allergic Drugs
Therapeutic category: – Antihistamine, Anti Allergic
Lorazepam Tablets
FreeLozepam 1
Lorazepam tablets BP 1 mg
Each Uncoated tablet contains:
Lorazepam BP 1 mgLozepam 2
Lorazepam tablets BP
Each Uncoated tablet contains:
Lorazepam BP 2 mgLOZEPAM – 2
Lorazepam Tablets USP 2 mg
Each Uncoated Tablet Contains:
Lorazepam USP 2 mgUsage: – Treat anxiety and sleeping problems
Category: – Anti Anxiety Drugs
Therapeutic category: – Anti Anxiety
Losartan Potassium, Amlodipine Besylate Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets (typically White or Yellow, Oval/Round). The formulation uses the Besylate salt of Amlodipine for maximum stability.
Active Ingredient Strength (Standard) Therapeutic Role Losartan Potassium USP/BP 50 mg Vasodilator (ARB) Amlodipine Besylate USP/BP 5 mg (Eq. to Base) Vasodilator (CCB) (High Strength Variant) 50 mg Losartan + 10 mg Amlodipine
100 mg Losartan + 5 mg AmlodipineResistant Hypertension Excipients Q.S. Microcrystalline Cellulose / Sodium Starch Glycolate Filler / Super-Disintegrant Losartan Tablets
FreeLosartan Potassium Tablets IP 25 mg
Each Film Coated Tablets Contains:
Losartan Potassium IP 25 mgLosartan Potassium Tablet IP 50 mg
Each Film Coated Tabet Contains :
Losartan Potasium IP 50 mgLOSARTAN POTASSIUM & HYDROCHLOROTHIAZIDE TABLETS IP
Each Film Coated Tablet Contains :: Losartan potassium IP 50 mg
Hydrochlorothiazide IP 12.5 mg – Excipients – QSLOSARTAN POTASSIUM & AMLODIPINE TABLET IP
Each Film Coated Tablet Contains :
Losartan potassium IP 50mg
Amlodipine Besylate IP Eq. To Amlodipine 5 mg
Excipients q.sUsage: – Treat high blood pressure (hypertension)
Category: – Anti Cardiovascular Drugs
Therapeutic category: – Cardiovascular Agent, Antihypertensive
Magnesium Sulphate Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Hypertonic Solution in Glass Ampoules.
Active Ingredient Concentration Strength (per 2ml) Magnesium Sulphate BP/USP 50% w/v (Standard) 1 Gram (in 2 ml) Magnesium Sulphate BP/USP 25% w/v 0.5 Gram (in 2 ml) Magnesium Sulphate BP/USP 10% w/v 2 Grams (in 20 ml) Osmolarity Approx. 4060 mOsmol/L Highly Hypertonic *Pack Sizes: Tray of 10 Ampoules, 25 Ampoules, or 100 Ampoules.
Maxgalin
FreeMaxgalin is a brand of medication manufactured by Sun Pharmaceutical Industries Ltd., primarily used to treat neuropathic (nerve) pain, fibromyalgia, and certain types of seizures.
Mebendazole Tablets
FreeEmzole
Mebendazole Tablets USP 100 mg
Each Uncoated tablet contains :
Mebendazole USP 100 mgMeben – 500
Mebendazole Tablets USP
Each Uncoated tablet contains :
Mebendazole USP 500 mgUsage: – Roundworm and whipworm infections
Category: – Anthelmintic Drugs
Therapeutic category: – Anthelmintic, Antiprotozoal, Antiparasitic
Mebeverine Tablets
FreeMebeverine Hydrochloride Tablets IP
Each filmcoated tablet contains :
Mebeverine Hydrochloride IP 135 mgUsage: – Helps with muscle spasms
Category: – Anti Analgesic Painkiller Drugs
Therapeutic category: – Painkiller
Mefenamic Acid Tablets
FreeMefenamic Acid Tablets
Each Uncoated Tablet contains :
Mefenamic Acid IP 500 mgMefenamic Acid Tablets
Each Uncoated Tablet contains :
Mefenamic Acid BP 500 mgUsage: – Relieve mild to moderate pain, including menstrual pain
Category: – Anti Analgesic Painkiller Drugs
Therapeutic category: – NSAID, Analgesic, Antipyretic, Anti Inflammatory
Mefloquine Tablets
FreeMefloquine Tablets
Each Filmcoated tablet contains:
Mefloquine Hydrochloride IP
Eq. To Mefloquine 250 mgMefloqine tablets
Each Filmcoated Tablets Contains:
Mefloquine Hydrochloride IP
Eq To Mefloquine 500 mgMefloquine tablets
Each Filmcoated Tablets Contains:
Mefloquine Hydrochloride BP
eq to Mefloquine 500 mgMefloquine tablets
Each Filmcoated Tablets Contains:
Mefloquine Hydrochloride BP
eq to Mefloquine 750 mgUsage: – Treat malaria
Category: – Antimalarial drugs
Therapeutic category: – Antimalarial
Meropenem & Sulbactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in glass vials.
Active Ingredient Strength Role Meropenem USP/BP 1000 mg (1 g) Broad-Spectrum Carbapenem Sulbactam Sodium USP/BP Equivalent to 500 mg Beta-Lactamase Inhibitor Total Strength 1.5 g (1500 mg) Per Single Dose Vial *Pack Sizes: Single Vial with or without Sterile Water for Injection.
Meropenem Injection
FreeMeropenem injection (brand name Keppra) is a broad-spectrum carbapenem antibiotic supplied as a sterile white to pale yellow powder for reconstitution. It is used to treat severe bacterial infections
Metformin Hydrochloride & Glimepiride Tablets
FreeProduct Composition & Strength
We supply this product in Bilayer Tablets or Matrix Tablets packed in Alu-Alu or PVC/PVDC Blisters.
Product Strength Metformin HCl (SR) Glimepiride Common Name 500/1 mg 500 mg 1 mg G1 Tablet 500/2 mg 500 mg 2 mg G2 Tablet 1000/2 mg 1000 mg 2 mg Forte / DS Tablet 1000/4 mg 1000 mg 4 mg High Strength Metformin Hydrochloride & Pioglitazone Tablets
FreeProduct Composition & Strength
We supply this product as Sustained Release (SR) Matrix Tablets packed in Alu-Alu Blisters.
Product Strength Metformin HCl (SR) Pioglitazone Common Name 500/15 mg 500 mg 15 mg Pio-M 15 500/30 mg 500 mg 30 mg Pio-M 30 1000/15 mg 1000 mg 15 mg Pio-M Forte 1000/30 mg 1000 mg 30 mg Pio-M DS