Showing 501–1000 of 1289 results
Enrofloxacin veterinary injection
FreeComposition
Enrofloxacin INJ. 50 ML
Usage: –It’s crucial to use enrofloxacin injections only under the guidance of a veterinarian
Category: – veterinary injection
Therapeutic category: – veterinary medicine
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Ergometrine Tablets
FreeErgometrine Tablets IP 250 mcg
Each Sugarcoated Tablet Contains:
Ergometrine Maleate IP 250 mcgUsage: – Postpartum haemorrhage and post abortion hemorrhage in patients with uterine atony
Category: – Generic Drugs and Medicine
Therapeutic category: –
Ergotamine & Caffeine ,Paracetamol & Prochlorperazine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Quad-API Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the sensitive ergot alkaloid and phenothiazine components.
Active Ingredient Strength Primary Clinical Function Ergotamine Tartrate USP/Ph.Eur. 1 mg The Vascular Terminator (Micro-Dose): The foundational ergot alkaloid required to aggressively constrict pulsing cranial arteries. Caffeine Anhydrous USP/Ph.Eur. 100 mg The Absorption Catalyst: Enhances Ergotamine bioavailability and accelerates onset of action. Paracetamol (Acetaminophen) USP/Ph.Eur. 250 mg The Central Analgesic (Macro-Dose): Provides immediate, broad-spectrum relief from accompanying head and neck pain. Prochlorperazine Maleate USP/Ph.Eur. 2.5 mg The Antiemetic Anchor (Ultra-Micro-Dose): Lethally blocks the CTZ to prevent vomiting and ensure full gastrointestinal absorption of the FDC. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Povidone K-30 / Magnesium Stearate / Premium Polymeric Film Diluent / Superdisintegrant / Binder / Lubricant (Engineered utilizing extreme multi-stage geometric dilution to flawlessly blend a microscopic 1mg and 2.5mg payload into a massive 250mg/100mg base, guaranteeing absolute content uniformity and zero toxic ‘hot spots’) *Pack Sizes: 10×10 Alu-Alu Blisters (Optimized specifically for strict, heavily limited acute neurology dispensing to prevent ergot toxicity).
Erlotinib Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips or heavy-gauge HDPE bottles to ensure the absolute chemical stability of the targeted antineoplastic API.
Active Ingredient Strength Primary Clinical Function Erlotinib (as Hydrochloride) USP/Ph.Eur. 100 mg The Pancreatic Oncology Standard: The definitive daily maintenance dose utilized globally in combination with Gemcitabine for advanced, unresectable, or metastatic pancreatic cancer. Erlotinib (as Hydrochloride) USP/Ph.Eur. 150 mg The Thoracic Oncology Standard: Massive therapeutic payload utilized as a first-line, maintenance, or second-line daily monotherapy for EGFR-mutated Non-Small Cell Lung Cancer. Excipients Lactose Monohydrate / Microcrystalline Cellulose / Sodium Starch Glycolate / Sodium Lauryl Sulfate / Magnesium Stearate / Premium Polymeric Film Diluent / Binder / Superdisintegrant / Surfactant / Heavy Film-Coating (Engineered specifically to maximize dissolution of this highly insoluble compound while locking the potent cytotoxic API safely inside the tablet, completely preventing handler exposure to carcinogenic dust) *Pack Sizes: 3×10 Alu-Alu Blisters or Bottles of 30 (Optimized specifically for strict 30-day chronic oncology dispensing regimens).
Erythromycin Capsules
FreeErythromycin capsules are macrolide antibiotics used to treat various bacterial infections. Because erythromycin base is easily destroyed by stomach acid, these capsules are typically formulated as delayed-release systems containing enteric-coated pellets
Erythromycin Tablets
FreeErythromycin Stearate Tablets IP 250 mg
Each Film coated tablet contains:
Erythromycin Stearate IP
Eq. To Erythromycin 250 mg
Color: ErythrosineErythromycin Stearate Tablets IP 500 mg
Each Film coated tablet contains:
Erythromycin Stearate IP
Eq. To Erythromycin 500 mg
Excipients q.sElthro – 250
Erythromycin Stearate Tablets IP 250 mg
Each Filmcoated tablet contains:
Erythromycin Stearate IP
Eq. To Erythromycin 250 mgERYTHROMYCIN STEARATE TABLETS IP 500 MG
Each Film Coated Tablet Contains:
Erythromycin Stearate IP (- )
Eq. to Erythromycin (500 mg)
Excipients (- QS)Eritro C
(Erythromycin Stearate Tablets BP)
Each Filmcoated tablet contains :
Erythromycin Stearate BP
Eq. To Erythromycin 500 mgYALLETHRO BP 500 (ERYTHROMYCIN STEAATE TABLETS BP 500 MG) Each film coated tablets contains: Erythromycin stearate eq. To Erythromycin BP 500 mg.
Usage: – Chest infections
Category: – Antibiotic Drugs
Therapeutic category: – Antibiotic
Erythropoietin Prefilled Syringe Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Liquid in a Prefilled Syringe (PFS) with a fine needle.
Active Ingredient Strength (IU) Patient Profile Recombinant Human Erythropoietin 2000 IU / 0.5 ml Maintenance / Low Weight Recombinant Human Erythropoietin 4000 IU / 0.4 ml Standard Dialysis Dose Recombinant Human Erythropoietin 10000 IU / 1.0 ml Chemotherapy / High Dose Stabilizer Human Albumin / Polysorbate Prevents Protein Aggregation *Pack Sizes: Blister Pack of 1 PFS or Box of 6 PFS.
Escitalopram Oxalate & Clonazepam Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Microdose FDC Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of both active pharmaceutical ingredients.
Active Ingredient Strength Primary Clinical Function Escitalopram (as Oxalate) USP/Ph.Eur. 5 mg / 10 mg The Serotonin Anchor (Macro-Dose): The foundational, highly selective SSRI required to cure the underlying clinical depression and generalized anxiety. Clonazepam USP/Ph.Eur. 0.25 mg / 0.5 mg The Anxiolytic Bridge (Micro-Dose): The highly potent, long-acting benzodiazepine required to suppress acute panic attacks and SSRI-induced activation syndrome. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Povidone K-30 / Colloidal Silicon Dioxide / Magnesium Stearate / Premium Polymeric Film Diluent / Superdisintegrant / High-Shear Binder / Glidant / Film-Coating (Engineered specifically utilizing advanced multi-stage geometric dilution to ensure the lethal 0.25mg/0.5mg micro-dose of Clonazepam is flawlessly distributed throughout the Escitalopram matrix, preventing toxic “hot spots”) *Pack Sizes: 10×10 Alu-Alu Blisters (Optimized specifically for strict 2-to-4 week short-term bridging regimens before stepping down to Escitalopram monotherapy).
Esomeprazole & Levosulpiride Capsules
FreeEsomeprazole and Levosulpiride capsules are a combination medication primarily used for gastroesophageal reflux disease (GERD) and peptic ulcers in patients who do not respond to a proton pump inhibitor (PPI) alone.
Esomeprazole Capsule
FreeEsomeprazole capsules primarily contain Esomeprazole Magnesium (as a dihydrate or trihydrate) as the active ingredient. Because esomeprazole is “acid-labile” (broken down by stomach acid), these capsules are formulated as delayed-release or gastro-resistant systems containing enteric-coated pellets.
Esomeprazole Domperidone Capsules
FreeEsomeprazole and Domperidone capsules are combination medications containing two active pharmaceutical ingredients, typically formulated as pellets within a hard gelatin capsule.
Esomeprazole Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in glass vials.
Active Ingredient Strength Form Esomeprazole Sodium USP/BP Equivalent to 40 mg Esomeprazole White to Off-White Cake Excipients Disodium Edetate / Sodium Hydroxide Chelating Agent / pH Adjuster *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials.
Esomeprazole Tablets
FreeEsomeprazole Tablets 20 mg
Each Enteric Coated Tablet Contains:
Esomeprazole Magnesium Trihydrate USP
eq. To Esomeprazole 20 mgEsomeprazole Tablets 40 mg
Each Enteric Coated Tablet Contains:
Esomeprazole Magnesium Trihydrate USP
eq. To Esomeprazole 40 mgUsage: – Heartburn, acid reflux and gastro-oesophageal reflux disease
Category: – Antacid & Antiulcer Drugs
Therapeutic category: – Antiulcer, Antacid
Ethambutol Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets. The coating is essential as Ethambutol HCl is highly hygroscopic (absorbs moisture) and has a bitter taste.
Active Ingredient Strength (Standard) Role in DOTS Therapy Ethambutol HCl USP/BP 200 mg Pediatric / Renal Adjustment Ethambutol HCl USP/BP 400 mg Standard Adult Unit Ethambutol HCl USP/BP 600 mg / 800 mg High Weight Band / Forte Ethambutol HCl USP/BP 1000 mg High Dose (Special Order) Excipients Q.S. Sorbitol / Magnesium Stearate Ethambutol Tablets
FreeEthambutol Hydrochloride Tablets IP 200 mg
Each Film Coated Tablet contains:
Ethambutol Hydrochloride IP 200 mgEthambutol tablets IP 400 mg
Each film coated tablet contains :
Ethambutol hydrochloride IP 400 mgEthambutol Hydrochloride Tablets IP 800 mg
Each Film Coated Tablet contains:
Ethambutol Hydrochloride IP 800 mgEthambutol Tablets BP 200 mg
Each Film Coated Tablet Contains :
Ethambutol Hydrochloride BP 200 mgEthambutol Tablets BP 400 mg
Each Film Coated Tablet Contains :
Ethambutol Hydrochloride BP 400 mgEthambutol Tablets BP 800 mg
Each Film Coated Tablet Contains :
Ethambutol Hydrochloride BP 800 mgUsage: – Treat Tuberculosis
Category: – Antibiotic Drugs
Therapeutic category: – Antibiotic, Anti tuberculosis
Ethamsylate tablets
FreeAn Ethamsylate tablet is a hemostatic (antihemorrhagic) medication used to prevent and control excessive bleeding, particularly from small blood vessels (capillaries).
Ethinyestradiol Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Hazard-Shielded Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the synthetic steroidal API.
Active Ingredient Strength Primary Clinical Function Ethinylestradiol USP/Ph.Eur. 0.05 mg (50 mcg) The Endocrinology Standard: The definitive micro-dose engineered specifically for female hypogonadism, primary ovarian failure, and strictly monitored estrogen replacement. Ethinylestradiol USP/Ph.Eur. 1 mg The Palliative Oncology Macrodose: Massive therapeutic payload utilized strictly for the aggressive hormonal suppression of advanced, inoperable prostate or breast cancer. Excipients Lactose Monohydrate / Microcrystalline Cellulose / Croscarmellose Sodium / Povidone K-30 / Magnesium Stearate / Premium Polymeric Film Diluent / Binder / Superdisintegrant / Lubricant / Heavy Film-Coating (Engineered specifically to form an impenetrable physical barrier over the tablet core. This completely seals the potent steroidal API, ensuring absolute safety for handlers and guaranteeing zero cross-contamination in the facility) *Pack Sizes: 10×10 Blisters or Bottles of 100 (Optimized specifically for strict chronic endocrine and oncology dispensing regimens).
Ethinylestradiol & Levonorgestrel Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Microdose FDC Tablet, packed exclusively in highly secure, moisture-resistant “Calendar Blister” packs to ensure absolute daily patient compliance and chemical stability.
Active Ingredient Strength Primary Clinical Function Ethinylestradiol USP/Ph.Eur. 0.03 mg (30 mcg) The Estrogenic Anchor: The synthetic estrogen required to suppress FSH release and stabilize the uterine lining to prevent irregular breakthrough bleeding. Levonorgestrel USP/Ph.Eur. 0.15 mg (150 mcg) The Progestin Powerhouse: The highly potent, second-generation progestin required to suppress the LH surge and thicken the cervical mucus. Excipients Lactose Monohydrate / Maize Starch / Povidone K-30 / Magnesium Stearate / Premium Sugar/Film Coating Diluent / Disintegrant / High-Shear Binder / Lubricant (Engineered specifically using elite multi-stage geometric dilution to guarantee the microscopic 30 mcg and 150 mcg payloads are flawlessly distributed, preventing lethal hormonal drops that cause contraceptive failure) *Pack Sizes: 21-Day Active Blisters OR 28-Day Calendar Blisters (21 Active Hormonal Tablets + 7 Placebo/Iron Tablets to enforce daily habit-forming compliance).
Ethionamide Tablets
FreeEthionamide Tablets IP 125 mg
Each Film Coated Tablet contains:
Ethionamide IP 125 mgEthionamide Tablets IP 250 mg
Each Film Coated Tablet contains:
Ethionamide IP 250 mgETHIONAMIDE TABLETS 250 MG
Each filmcoated tablet contains:
Ethionamide BP 250 mgUsage: – Treat Tuberculosis
Category: – Antibiotic Drugs
Therapeutic category: – Antibiotic, Ant tuberculosis
Etizolam Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Microdose Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the ultra-low-dose API.
Active Ingredient Strength Primary Clinical Function Etizolam JP/Ph.Eur. 0.25 mg The Geriatric & Daytime Standard: The definitive ultra-low-dose unit utilized for daytime anxiety suppression without severe sedation, or for highly sensitive elderly patients. Etizolam JP/Ph.Eur. 0.5 mg / 1 mg The Panic & Insomnia Standard: High-efficacy therapeutic payload utilized for the immediate termination of acute panic attacks and the induction of sleep in severe insomnia. Excipients (MD Formulation) Mannitol / Crospovidone / Aspartame / Peppermint Flavor / Magnesium Stearate Diluent / Superdisintegrant / Sweetener / Flavoring (Engineered specifically using elite “Rapid-Burst” superdisintegrants to ensure the tablet shatters and dissolves in saliva within seconds, masking the chemical taste for maximum patient compliance during severe distress) *Pack Sizes: 10×10 Alu-Alu Blisters (Optimized specifically for strict, short-term psychiatric dispensing regimens).
Etmsylate Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Glass Ampoules.
Active Ingredient Strength Volume Etamsylate USP/BP 250 mg 2 ml Ampoule (125 mg/ml) – Standard Etamsylate USP/BP 125 mg 2 ml Ampoule – Pediatric Excipients Sodium Metabisulfite Antioxidant *Pack Sizes: Tray of 10 Ampoules, 50 Ampoules, or 100 Ampoules.
Etofylline Theophylline Tablets
FreeEt Phylline Retard Plus
Etofylline & Theophylline Tablets
Each Sugar Coated Sustained Release Tablet Contains:
Etofylline BP 231 mg
Theophylline BP 69 mgET Phyllin Retard
Etofylline & Theophylline Tablets
Each Sustained release sugarcoated tablet contains:
Etofylline IP 115 mg
Theophylline IP 35 mgET Phyllin
Etofylline & Theophylline Tablets
Each sugarcoated tablet contains:
Etofylline IP 77 mg
Theophylline IP 23 mgUsage: – Treatment and prevention of asthma and chronic obstructive pulmonary disease
Category: – Anti Allergic Drugs
Therapeutic category: – Antiasthmatic
Etophylline & Theophylline injection
FreeComposition
Each ml contains- Etophylline IP 84.7 mg + Theophylline
(anhydrous) eq. to Theophylline (hydrated)
25.3 mg + Benzyl alcohol ( as preservative )
0.2% v/v + WFI IP q.s INJECTION (2 ML)
Usage: – commonly used in the treatment of asthma
Category: – Pulmonary injection (COPD)
Therapeutic category: –Pulmonary Disease (COPD)
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Etoricoxib Tablets
FreeEtoricoxib tablets 90 mg
Each film coated tablet contains:
Etoricoxib 90 mgUsage: – Pain relief
Category: – Anti Analgesic Painkiller Drugs
Therapeutic category: – NSAID, Analgesic, Generic Drugs
Everolimus Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Antioxidant-Stabilized Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical survival of the highly oxidative, potent API.
Active Ingredient Strength Primary Clinical Function Everolimus USP/Ph.Eur. 0.25 mg / 0.5 mg / 0.75 mg Global Transplant Standard: Ultra-micro doses utilized for strict, lifelong immunosuppression following renal or hepatic allografts. Requires extreme content uniformity to maintain the narrow therapeutic blood trough levels (3 to 8 ng/mL). Everolimus USP/Ph.Eur. 5 mg / 10 mg Global Oncology Standard: Massive-dose antineoplastic payload utilized for the aggressive suppression of Renal Cell Carcinoma, Neuroendocrine Tumors (NETs), and HR+/HER2- Breast Cancer. Excipients Butylated Hydroxytoluene (BHT) / Hypromellose / Lactose Anhydrous / Crospovidone / Magnesium Stearate Antioxidant / Binder / Diluent / Superdisintegrant / Lubricant (Engineered specifically utilizing BHT (Butylated Hydroxytoluene) to prevent the rapid oxidative degradation of the Everolimus molecule, guaranteeing maximum clinical shelf-life and potency across harsh climates) *Pack Sizes: 10×10 Alu-Alu Blisters (Transplant) or 3×10 Alu-Alu Blisters (Oncology – 30 Day Supply).
Exemestane Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the potent steroidal antineoplastic agent.
Active Ingredient Strength Primary Clinical Function Exemestane USP/Ph.Eur. 25 mg Global Adult Oncology Standard: The definitive daily maintenance dose required to maintain absolute, continuous destruction of peripheral aromatase enzymes and keep estrogen levels crashed. Excipients Mannitol / Copovidone / Crospovidone / Sodium Starch Glycolate / Magnesium Stearate / Premium Polymeric Film Diluent / High-Shear Binder / Superdisintegrant / Surfactant / Heavy Film-Coating (Engineered specifically to lock the highly potent, teratogenic steroidal API safely inside the tablet, completely preventing handler and pharmacist exposure to the hazardous dust) *Pack Sizes: 10×10 Blisters or Bottles of 30 (Optimized specifically for strict 30-day chronic oncology dispensing regimens).
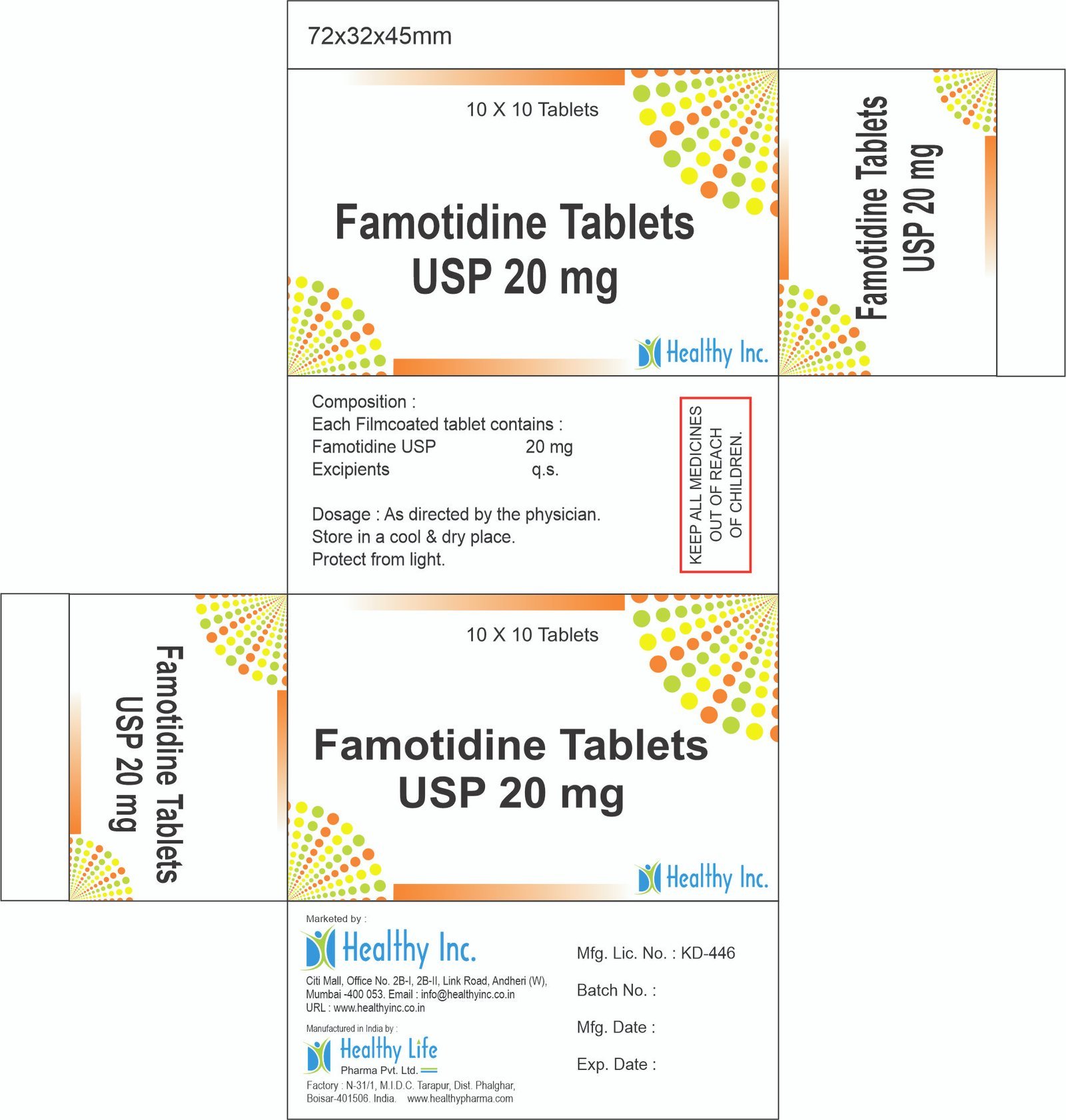
Famotidine Tablets
FreeFamotidine Tablets USP 20 mg
Each Filmcoated tablet contains :
Famotidine USP 20 mgUsage: – Prevent and treat heartburn due to acid indigestion and sour stomach
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Ferric Hydroxide injection
FreeComposition
Ferric Hydroxide (Iron) 20 mg INJ 1 X 5 ML
Usage: –it is commonly used to treat iron deficiency anemia
Category: – anemia injection
Therapeutic category: –deficiency anemia
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Ferrous Folic Tablets
FreeFerrous Sulfate With Folic Acid Tablets
Each Enteric-Coated Tablet Contains:
Dried Ferrous Sulfate IP 150 mg
eq to elemental Iron 45 mg
Folic Acid IP 0.4 mg
Excipent ….Q.s.
Colour : Indigo carmineFerrous Sulfate With Folic Acid Tablets
(IFA large)
Each Enteric-Coated Tablet Contains:
Dried Ferrous Sulfate IP 335 mg
eq to elemental Iron 100 mg
Folic Acid IP 0.5 mg
Excipent q.s
Colour: Red Oxide of IronIron-Folate Tablets
(Ferrous Sulphate With Folic Acid Tablets)
Each film coated tablet contains:
Dried Ferrous Sulphate IP 335 mg
eq. To Elemental Iron 100 mg
Folic Acid IP 0.5 mgFerrous Sulfate With Folic Acid Tablets
(large)
Each Enteric-Coated Tablet Contains:
Dried Ferrous Sulfate IP 335 mg
eq to elemental Iron 100 mg
Folic Acid IP 0.5 mg Colour: Blue Coloured (Indigo Carmine)FERROUS SULPHATE WITH FOLIC ACID ( LARGE) Each enteric coated tablet contains: Dried ferrous sulphate IP 335 mg Equivalent to elemental iron IP 100 mg Folic acid IP 0.5 mg Blue coloured (indigo Carmine) excipients q.s.
Ferrous Sulphate with Folic Acid Tablets
(Pediatric)
Each Film Coated tablet contains:
Dried Ferrous Sulphate IP 67 mg
Eq.to elemental iron 20 mg
Folic Acid IP 100 mcgFerro Force
(Folic Acid & Ferrous Sulphate Tablets)
Each Sugarcoated tablet contains:
Dried Ferrous Sulphate BP 200 mg
Eq.to elemental iron 60 mg
Folic Acid BP 0.25 mgFERROUS SULFATE WITH FOLIC ACID TABLETS
Each Enteric-Coated Tablet Contains:
Dried Ferrous Sulfate IP 335 mg
eq to elemental Iron 100 mg
Folic Acid IP 0.5 mgFESFO- E
(Ferrous Sulphate With Folic Acid Tablets)
Each Enteric Coated Tablet Contains:
Dried Ferrous Sulphate IP 100 mg
eq. To Elemental Iron 30 mg
Folic Acid IP 0.1 mgColour: Indigo Carmine lake
Ferrous Sulfate With Folic Acid Tablets
Each Enteric-Coated Tablet Contains:
Dried Ferrous Sulfate IP 150 mg
eq to elemental Iron 45 mg
Folic Acid IP 0.4 mg
Excipent ….Q.s.
Colour : Indigo carmineFerrous Sulphate with Folic Acid Tablets
(Pediatric)
Each Film Coated tablet contains:
Dried Ferrous Sulphate IP 67 mg
Eq.to elemental iron 20 mg
Folic Acid IP 100 mcgUsage: – Treat or prevent low blood levels of iron
Category: – Generic Drugs and Medicine
Therapeutic category: – Vitamin supplement
Ferrous Sulphate Tablets
FreeFerrous Sulphate Tablets IP 200 mg
Each Sugarcoated tablet contains :
Dried Ferrous sulphate IP 200 mg
Eq.To Elemental Iron 60 mgFerrous sulphate Tablets IP 100 mg
Each Film coated tablet contains:
Dried ferrous sulphate IP 100 mgFerrous Sulphate Tablets IP 300 mg
Each Sugarcoated tablet contains :
Dried Ferrous sulphate IP 300 mgFerrous Sulphate Tablets BP 200 mg
Each Sugarcoated tablet contains :
Dried Ferrous sulphate BP 200 mg
Eq.To Elemental Iron 60 mgUsage: – Treat and prevent iron deficiency anemia
Category: – Generic Drugs and Medicine
Therapeutic category: – Vitamin supplement
Finasteride Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Hazard-Shielded Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the highly potent active pharmaceutical ingredient.
Active Ingredient Strength Primary Clinical Function Finasteride USP/Ph.Eur. 1 mg The Global Dermatology Standard: The definitive micro-dose engineered specifically for the lifelong, daily maintenance of Androgenetic Alopecia (Male Pattern Hair Loss). Finasteride USP/Ph.Eur. 5 mg The Global Urology Standard: High-efficacy macro-dose utilized for the aggressive shrinkage of the prostate gland in symptomatic Benign Prostatic Hyperplasia (BPH). Excipients Lactose Monohydrate / Microcrystalline Cellulose / Sodium Starch Glycolate / Docusate Sodium / Magnesium Stearate / Premium Polymeric Film Diluent / Binder / Superdisintegrant / Surfactant / Heavy Opadry Film-Coating (Engineered specifically to form an impenetrable physical barrier over the tablet core. This completely seals the teratogenic API, ensuring absolute safety for female pharmacists and caregivers handling the medication) *Pack Sizes: 10×10 Blisters or Bottles of 30/100 (Optimized specifically for strict, lifelong chronic urology and dermatology dispensing regimens).
Flucloxacillin and Amoxicillin Capsules
FreeFlucloxacillin and amoxicillin combination capsules (often referred to by trade names like Flamox (cosmos-pharm.com) or Amoxam) contain two distinct penicillin-type antibiotics (hra.nhs.uk) that work together to treat a broader range of bacterial infections.
Flucloxacillin Capsules
FreeFlucloxacillin capsules contain flucloxacillin sodium as the active ingredient, along with specific excipients that vary slightly by manufacturer but typically include a lubricant, a glidant, and sodium.
Flucloxacillin Injection
FreeFlucloxacillin injection is a narrow-spectrum penicillin antibiotic used to treat infections caused by staphylococci. It is typically supplied as a white to off-white sterile powder for reconstitution.
Fluconazole & Ivermectin Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Microdose FDC Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of both active pharmaceutical ingredients.
Active Ingredient Strength Primary Clinical Function Fluconazole USP/Ph.Eur. 150 mg The Antifungal Anchor (Macro-Dose): The foundational triazole payload required to systematically eradicate widespread dermatophytes and yeasts. Ivermectin EP/USP 6 mg / 12 mg The Antiparasitic Assassin (Micro-Dose): The highly potent, targeted neurotoxin required to paralyze burrowing mites, lice, and severe helminthic infections. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Povidone K-30 / Colloidal Silicon Dioxide / Magnesium Stearate / Premium Polymeric Film Diluent / Superdisintegrant / High-Shear Binder / Glidant / Film-Coating (Engineered specifically utilizing advanced multi-stage geometric dilution to ensure the lethal 6mg micro-dose of Ivermectin is flawlessly distributed throughout the massive Fluconazole matrix, preventing toxic “hot spots”) *Pack Sizes: Single-Dose Blister Packs (1×1 or 1×2) or 10×10 Blisters (Optimized specifically for rapid NGO deployment and “stat” dosing protocols).
Fluconazole Capsules
FreeFluconazole capsules (commonly known by the brand name Diflucan (pfizermedical.com)) contain the active antifungal ingredient fluoxetine hydrochloride along with a powder fill of inactive excipients and a hard gelatin shell.
Fluconazole Tablets
FreeFLUCONAZOLE TABLETS IP 150 MG
Each uncoated tablet contains:
Fluconazole IP 150 mg Excipients q.s.Nuflucon – 200
Fluconazole Tablets IP 200 mg
Each Uncoated Tablet contains :
Fluconazole IP 200 mgUsage: – Prevent and treat a variety of fungal and yeast infections
Category: – Antifungal Drugs
Therapeutic category: – Antifungal
Fludac
FreeFludac is a brand name for capsules manufactured by Cadila Pharmaceuticals Ltd (amazon.in), used primarily as an antidepressant. It belongs to the class of Selective Serotonin Reuptake Inhibitors (SSRIs) (1mg.com).
Flunarazine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Microdose Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the dihydrochloride salt.
Active Ingredient Strength Primary Clinical Function Flunarizine (as Dihydrochloride) USP/Ph.Eur. 5 mg The Pediatric & Geriatric Standard: The definitive low-dose unit required for migraine prophylaxis in children or for treating vertigo in elderly patients who are highly sensitive to drug accumulation. Flunarizine (as Dihydrochloride) USP/Ph.Eur. 10 mg The Global Adult Migraine Standard: High-efficacy adult maintenance dose utilized for maximum suppression of chronic, refractory migraine attacks. Excipients Lactose Monohydrate / Microcrystalline Cellulose / Crospovidone / Colloidal Silicon Dioxide / Magnesium Stearate Diluent / Binder / Superdisintegrant / Glidant / Lubricant (Engineered utilizing elite low-dose geometric dilution to guarantee that the 5mg or 10mg micro-dose is flawlessly distributed, preventing accidental drug-accumulation spikes) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict, heavily monitored 3-to-6 month neurology dispensing regimens).
Fluoxetine Capsules
FreeFluoxetine capsules, commonly known by the brand name Prozac (accessdata.fda.gov), contain the active antidepressant fluoxetine hydrochloride along with several inactive ingredients that vary by manufacturer and dosage strength.
Flupentixol & Melitracen Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Microdose FDC Tablet, packed exclusively in highly secure, light-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the highly photolabile thioxanthene derivative.
Active Ingredient Strength Primary Clinical Function Flupentixol (as Dihydrochloride) USP/Ph.Eur. 0.5 mg The Anxiolytic Activator (Ultra-Micro-Dose): The foundational presynaptic autoreceptor antagonist required to rapidly resolve apathy, fatigue, and physical tension. Melitracen (as Hydrochloride) USP/Ph.Eur. 10 mg The Mood Elevator (Micro-Dose): The highly tolerable TCA required to lift the baseline depressive neurosis. Excipients Lactose Monohydrate / Microcrystalline Cellulose / Croscarmellose Sodium / Magnesium Stearate / Premium Opaque Polymeric Film Diluent / Binder / Superdisintegrant / Lubricant / UV-Barrier Film (Engineered utilizing elite multi-stage geometric dilution to guarantee the lethal 0.5mg micro-dose of Flupentixol is flawlessly distributed throughout the matrix, finished with a heavy, opaque coat to prevent UV degradation) *Pack Sizes: 10×10 Blisters (Optimized specifically for rapid-response, short-to-medium term psychiatric and general medicine dispensing regimens).
Fluphenazine Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Oily Solution in Amber Glass Ampoules or Vials.
Active Ingredient Strength Type Fluphenazine Decanoate USP/BP 25 mg 1 ml Ampoule / Vial Fluphenazine Decanoate USP/BP 12.5 mg 0.5 ml Ampoule (Starter Dose) Vehicle Sesame Oil Depot Base Preservative Benzyl Alcohol 1.5% v/v (In Vials) *Pack Sizes: Tray of 5 Ampoules, 10 Ampoules, or 10 ml Multi-Dose Vial.
Folic Acid, Niacinamide Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated or Sugar Coated Tablets (typically Yellow or White). The coating protects the photosensitive vitamins from degradation.
Active Ingredient Strength (Standard) Therapeutic Role Folic Acid USP/BP 1.5 mg / 5 mg DNA Repair / Anemia Prevention Niacinamide USP/BP 20 mg / 50 mg / 100 mg Metabolic Support / Skin Barrier Excipients Q.S. Dibasic Calcium Phosphate / Starch Filler / Disintegrant Fortified Procaine Peniciilin Injection with Streptomycin
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials.
Active Ingredient Strength Role Procaine Penicillin G 300,000 IU Sustained Release (24 Hours) Benzyl Penicillin Sodium 100,000 IU Rapid Onset (Peak Levels) Streptomycin Sulfate Equivalent to 0.5 g (500 mg) Base Gram-Negative Coverage Total Formulation 400,000 IU + 0.5 g Standard Adult Dose *Pack Sizes: Tray of 50 Vials or 100 Vials.
Fortified Procaine Penicillin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials.
Active Ingredient Proportion Role Procaine Penicillin G 300,000 IU (75%) Long Acting (Depot) Benzyl Penicillin Sodium 100,000 IU (25%) Rapid Acting (Peak) Total Strength 400,000 IU (4 Lac) Standard Adult Dose Total Strength 4,000,000 IU (40 Lac) High Dose / Veterinary *Pack Sizes: Tray of 50 Vials or 100 Vials.
FRUSEMIDE INJECTION
FreeComposition
Each ml. contains: Frusemide I.P. ……..……….. 10 mg.
Benzyl Alcohol I.P. ………….1.0 % w/v
(As Preserva9ve )
Water for Injec9on I.P ……….. Q.S 2 mlUsage: – High blood pressure, heart failure and edema
Category: – Diuretic injection
Therapeutic category: – Diuretic
Pcd pharma franchise:-
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Frusemide Tablets
FreeFrusemide Tablets IP 40 mg
Each Uncoated tablet contains:
Frusemide IP 40 mgFurosemide Tablets
Frusemide Tablets BP 40 mg
Each Uncoated tablet contains:
Frusemide BP 40 mgUsage: – High blood pressure, heart failure and edema
Category: – Diuretic drugs
Therapeutic category: – Diuretic
Frusemide veterinary injection
FreeComposition
Frusemide VET INJ. 10ML
Usage: –It is crucial to follow the veterinarian’s prescription and dosage instructions precisely.
Category: – veterinary practice
Therapeutic category: – veterinary practice
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Furosemide & Spironolactone Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of both active pharmaceutical ingredients.
Active Ingredient Strength Primary Clinical Function Furosemide USP/Ph.Eur. 20 mg The Loop Diuretic (Fluid Clearance): Rapidly eliminates severe fluid buildup (edema) from the lungs, abdomen, and peripheral tissues. Spironolactone USP/Ph.Eur. 50 mg The Potassium-Sparing Shield: Blocks aldosterone, prevents toxic cardiac fibrosis, and heavily protects the patient against lethal hypokalemia. Excipients Microcrystalline Cellulose / Lactose Monohydrate / Sodium Starch Glycolate / Povidone K-30 / Magnesium Stearate / Premium Polymeric Film Diluent / Binder / Superdisintegrant / Slipper-Coating (Engineered utilizing advanced wet-granulation to ensure both the rapid-acting Furosemide and the slower-acting Spironolactone dissolve at precise, synchronized rates to guarantee the electrolyte-sparing synergy) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict chronic cardiology and hepatology dispensing regimens).
Furosemide Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Amber Glass Ampoules.
Active Ingredient Strength Volume Furosemide USP/BP 20 mg 2 ml Ampoule (10 mg/ml) – Standard Furosemide USP/BP 40 mg / 4 ml 4 ml Ampoule – High Dose Furosemide USP/BP 250 mg / 25 ml 25 ml Vial – Renal Failure Excipients Sodium Hydroxide / NaCl pH Adjuster / Isotonicity *Pack Sizes: Tray of 10 Ampoules, 50 Ampoules, or 100 Ampoules.
Gabapentin & Amitriptyline Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Macrodose FDC Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of both active pharmaceutical ingredients.
Active Ingredient Strength Primary Clinical Function Gabapentin USP/Ph.Eur. 300 mg / 400 mg The Neuromodulator (Macro-Dose): The foundational calcium-channel inhibitor required to stabilize hyperactive nerve terminals. Amitriptyline (as Hydrochloride) USP/Ph.Eur. 10 mg The Synaptic Enhancer (Micro-Dose): The highly potent TCA required to amplify descending pain inhibition and restore sleep patterns. Excipients Microcrystalline Cellulose / Crospovidone / Copovidone / Colloidal Silicon Dioxide / Magnesium Stearate / Premium Polymeric Film Diluent / Superdisintegrant / High-Shear Binder / Glidant / Film-Coating (Engineered specifically utilizing advanced multi-stage geometric dilution to ensure the lethal 10mg micro-dose of Amitriptyline is flawlessly distributed throughout the massive Gabapentin matrix, preventing toxic “hot spots”) *Pack Sizes: 10×10 Alu-Alu Blisters (Optimized specifically for strict 30-day chronic neurology dispensing regimens).
Gabapentin & Nortriptyline Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Macrodose FDC Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of both active pharmaceutical ingredients.
Active Ingredient Strength Primary Clinical Function Gabapentin USP/Ph.Eur. 400 mg The Neuromodulator (Macro-Dose): The foundational calcium-channel inhibitor required to stabilize hyperactive nerve terminals. Nortriptyline (as Hydrochloride) USP/Ph.Eur. 10 mg The Synaptic Enhancer (Micro-Dose): The highly potent TCA required to amplify descending pain inhibition and restore sleep patterns. Excipients Microcrystalline Cellulose / Crospovidone / Copovidone / Colloidal Silicon Dioxide / Magnesium Stearate / Premium Polymeric Film Diluent / Superdisintegrant / High-Shear Binder / Glidant / Film-Coating (Engineered specifically utilizing advanced multi-stage geometric dilution to ensure the lethal 10mg micro-dose of Nortriptyline is flawlessly distributed throughout the massive 400mg Gabapentin matrix, preventing toxic “hot spots”) *Pack Sizes: 10×10 Alu-Alu Blisters (Optimized specifically for strict 30-day chronic neurology dispensing regimens).
Gabapentin Capsules
FreeGabapentin capsules (commonly 100 mg, 300 mg, and 400 mg) contain the active pharmaceutical ingredient gabapentin USP, along with a variety of inactive excipients that aid in the drug’s delivery and stability.
Gabapentine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Macrodose Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute physical stability of the heavily compressed active pharmaceutical ingredient.
Active Ingredient Strength Primary Clinical Function Gabapentin USP/Ph.Eur. 600 mg Global Neuropathy Standard: The definitive high-dose unit utilized for the aggressive, long-term maintenance of severe diabetic peripheral neuropathy and postherpetic neuralgia. Gabapentin USP/Ph.Eur. 800 mg Max-Dose Seizure & Refractory Pain Standard: Massive therapeutic payload utilized for high-dose titration (up to 3600 mg/day) in refractory partial-onset seizures and extreme nerve pain. Excipients Copovidone / Crospovidone / Microcrystalline Cellulose / Magnesium Stearate / Premium Polymeric Film High-Shear Binder / Superdisintegrant / Diluent / Slipper-Coating (Engineered specifically utilizing advanced wet-granulation to compress the massive, fluffy 800mg API into a dense, smooth, swallowable tablet, preventing the pill from becoming a dangerous choking hazard for elderly patients) *Pack Sizes: 10×10 Blisters or Bottles of 100 (Optimized specifically for chronic, high-volume daily dispensing regimens).
Gefitinib tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips or heavy-gauge HDPE bottles to ensure the absolute chemical stability of the targeted antineoplastic agent.
Active Ingredient Strength Primary Clinical Function Gefitinib USP/Ph.Eur. 250 mg Global Adult Oncology Standard: The definitive, highly precise daily maintenance dose required to maintain constant, paralyzing pressure on the mutated EGFR protein to keep the lung cancer in deep molecular remission. Excipients Lactose Monohydrate / Microcrystalline Cellulose / Croscarmellose Sodium / Sodium Lauryl Sulfate / Magnesium Stearate / Premium Polymeric Film Diluent / Binder / Superdisintegrant / Surfactant / Heavy Film-Coating (Engineered specifically to maximize the dissolution of this poorly soluble compound while locking the cytotoxic API safely inside the tablet, completely preventing handler and pharmacist exposure to the carcinogenic dust) *Pack Sizes: 10×10 Alu-Alu Blisters or Bottles of 30 (Optimized specifically for strict 30-day chronic oncology dispensing regimens).
Gentamicin Injection
FreeGentamicin injection (brand name Garamycin) is a sterile, clear, colourless to slightly yellow aqueous solution of an aminoglycoside antibiotic. It is used to treat serious gram-negative bacterial infections.
Gentamicin injection
FreeComposition
Each 2 ml Contains :Gentamicin Sulphate I.P.
Equiv. To. Gentamicin base 80 mg
Methyl Paraben I.P 0.18 % W/V ( As Preserva9ve )
Propyl Paraben I.P 0.02 % W/V ( As Preserva9ve )
Water for Injection I.P. Q.S. 2 ml AMP, 10 ml, 20 ml, 30 mlEach 2 ml Contains :Gentamicin Sulphate I.P.
Equi. To. Gentamicin base 20 mg
Methyl Paraben I.P 0.18 % W/V( As Preserva9ve )
Propyl Paraben I.P 0.02 % W/V( As Preserva9ve )
Water for Injec9on I.P. Q.S. 2 ml AMPsage: – It is commonly used to treat various bacterial infections
Category: – antibiotic injection
Therapeutic category: – antibiotic medication
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Gentamicin Veterinary injection
FreeComposition
Gentamicin VET INJ. 30 M
Gentamicin VET INJ. 100 ML
Usage: – It is commonly used to treat various bacterial infections Animal
Category: – Veterinary injection
Therapeutic category: – Veterinary medicine
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Glibenclamide & Metformin Hydrochloride Tablets
FreeProduct Composition & Standard Strengths
We supply this product in standardized Fixed-Dose Combinations (FDCs) designed for flexible clinical titration.
Active Ingredient Standard Strengths (per Tablet) Primary Clinical Function Metformin HCl 400 mg / 500 mg The Foundation: Reduces glucose production. Glibenclamide (Glyburide) 2.5 mg / 5 mg The Stimulator: Enhances insulin secretion. Excipients Pharma Grade The Matrix: Ensures stability of the FDC. Glibenclamide Tablets
FreeGlibenclamide Tablets IP 5 mg Each Uncoated tablet contains: Glibenclamide IP 5 mg
Excipients – QSGLIBENCLAMIDE & METFORMIN HYDROCHLORIDE SR TABLETS
Each sustained release tablet contains:: – Glibenclamide 5 mg
– Metformin Hydrochloride 500 mg
– Excipients – QSGilben
Glibenclamide Tablets BP 5 mg
Each Uncoated tablet contains :
Glibenclamide BP 5 mgTrippleace Glibenclamide – 5
Glibenclamide Tablets BP 5 mg
Each Uncoated tablet contains :
Glibenclamide BP 5 mgUsage: – Along with diet and exercise to improve blood sugar control
Category: – Anti Diabetic drugs
Therapeutic category: – Anti Diabetic
Gliclazide Tablets
FreeGLICAZIDE TABLETS BP 80 mg Each Uncoated tablet contains : GLICLAZIDE BP 80 mg
Usage: – Type 2 diabetes is an illness
Category: – Anti Diabetic drugs
Therapeutic category: – Anti Diabetic
Glimepiride Tablets
FreeGlimepiride Tablets IP 1 mg
Each Filmcoated tablet contains :
Glimepiride IP 1 mgGlimepiride Tablets IP 2 mg
Each Filmcoated tablet contains :
Glimepiride IP 2 mgGlimepiride Tablets 1 mg
Each Filmcoated tablet contains :
Glimepiride BP 1 mgGlimepiride Tablets 2 mg
Each Filmcoated tablet contains :
Glimepiride BP 2 mgGlimepiride Tablets 4 mg
Each Film Coated Tablet Contains:
Glimepiride BP 4 mgSMEGLIP -1 / GLIMEPIRIDE TABLETS BP 1 MG Each Filmcoated Tablet Contains: Glimepiride BP 1 mg
Excipients – QSSMEGLIP -2 / GLIMEPIRIDE TABLETS BP 2 MG Each Filmcoated Tablets Contains: Glimepiride BP 2 mg
Excipients – QSGLIMEPIRIDE,PIOGLITAZONE & METMIN HYDROCHLORIDE SR TABLET
Each Sustained Release Tablet Contains :
Glimepiride 2 mg
Pioglitazone 15 mg
Metmin Hydrochloride 500 mg
Excipients q.sUsage: – Type 2 diabetes is a condition where the body doesn’t make enough insulin, or the insulin that it makes doesn’t work properly
Category: – Anti Diabetic drugs
Therapeutic category: – Anti Diabetic
Glimepiride, Pioglitazone & Metformin Hydrochloride Tablets
FreeProduct Composition & Standard Strengths
We supply this product in standardized Fixed-Dose Combinations (FDCs) designed for flexible clinical titration.
Active Ingredient Common Strength Primary Clinical Function Metformin HCl (SR/IR) 500 mg / 1000 mg The Foundation: Reduces glucose production. Glimepiride 1 mg / 2 mg The Stimulator: Enhances insulin secretion. Pioglitazone 15 mg The Sensitizer: Fixes insulin resistance. Glipizide Tablets
FreeGlipizide Tablet IP
Each uncoated Tablet contains :
Glipizide IP 5 mgGynoglid Tablets
Glipizide Tablets BP 5 mg
Each Uncoated tablet contains :
Glipizide BP 5 mgUsage: – Lowers blood sugar by causing the pancreas to produce insulin
Category: – Anti Diabetic drugs
Therapeutic category: – Anti Diabetic
Glucosamine ,Chondroitin & Diacerein Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Oblong Caplet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the highly hygroscopic sulfate salts.
Active Ingredient Strength Primary Clinical Function Glucosamine Sulfate Potassium Chloride USP/Ph.Eur. 750 mg The Structural Foundation: Massive therapeutic payload required to stimulate raw cartilage synthesis. Chondroitin Sulfate Sodium USP/Ph.Eur. 200 mg / 250 mg The Hydraulic Restorer: Pulls water into the joint matrix to restore physical shock-absorption and elasticity. Diacerein USP/Ph.Eur. 50 mg The Biochemical Brake: Direct Interleukin-1 (IL-1) inhibition to halt the enzymatic degradation of the joint space. Excipients Microcrystalline Cellulose / Povidone K-30 / Croscarmellose Sodium / Magnesium Stearate / Premium Moisture-Barrier Film Diluent / High-Shear Binder / Superdisintegrant / Lubricant / Slipper-Coating (Engineered specifically to compress over 1000mg of bulky, powdery API into a single, dense, swallowable “caplet” without crumbling, heavily coated to mask the bitter taste and lock out tropical humidity) *Pack Sizes: 10×10 Alu-Alu Blisters (Optimized specifically for strict 3-to-6 month chronic orthopedic dispensing regimens).
Glucosamine Capsule
FreeGlucosamine capsules typically contain one or more forms of glucosamine, an amino sugar essential for building cartilage and joint tissues. These active ingredients are often derived from the shells of shellfish (crabs, shrimp, and lobsters) or produced from vegetarian sources like fermented corn or fungi.
Glucosamine Sulphate Chondroitin Sulphate Tablets
FreeGlucosamine Sulphate And Chondroitin Sulphate Tablets
Each Film Coated Tablet Contains:
Glucosamine Sulphate 500 mg
added as Glucosamine Sulphate Sodium Chloride
Chondroitin Sulphate 400 mg
added as Chondroitin Sulphate SodiumUsage: – slow or prevent the degeneration of joint cartilage
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Glucose Infusion 10%
FreeActive IngredientStrength (per 100 ml)Primary Clinical FunctionGlucose (Dextrose) IP/BP/USP10.0 gThe Fuel: Provides 34 kcal per 100 ml.ExcipientsWater for Injection (WFI)The Vehicle: Sterile and pyrogen-free.Osmolarity~505 mOsm/LThe Physics: Hypertonic solution.pH Range3.5 to 6.5The Stability: Chemically optimized for shelf-life.
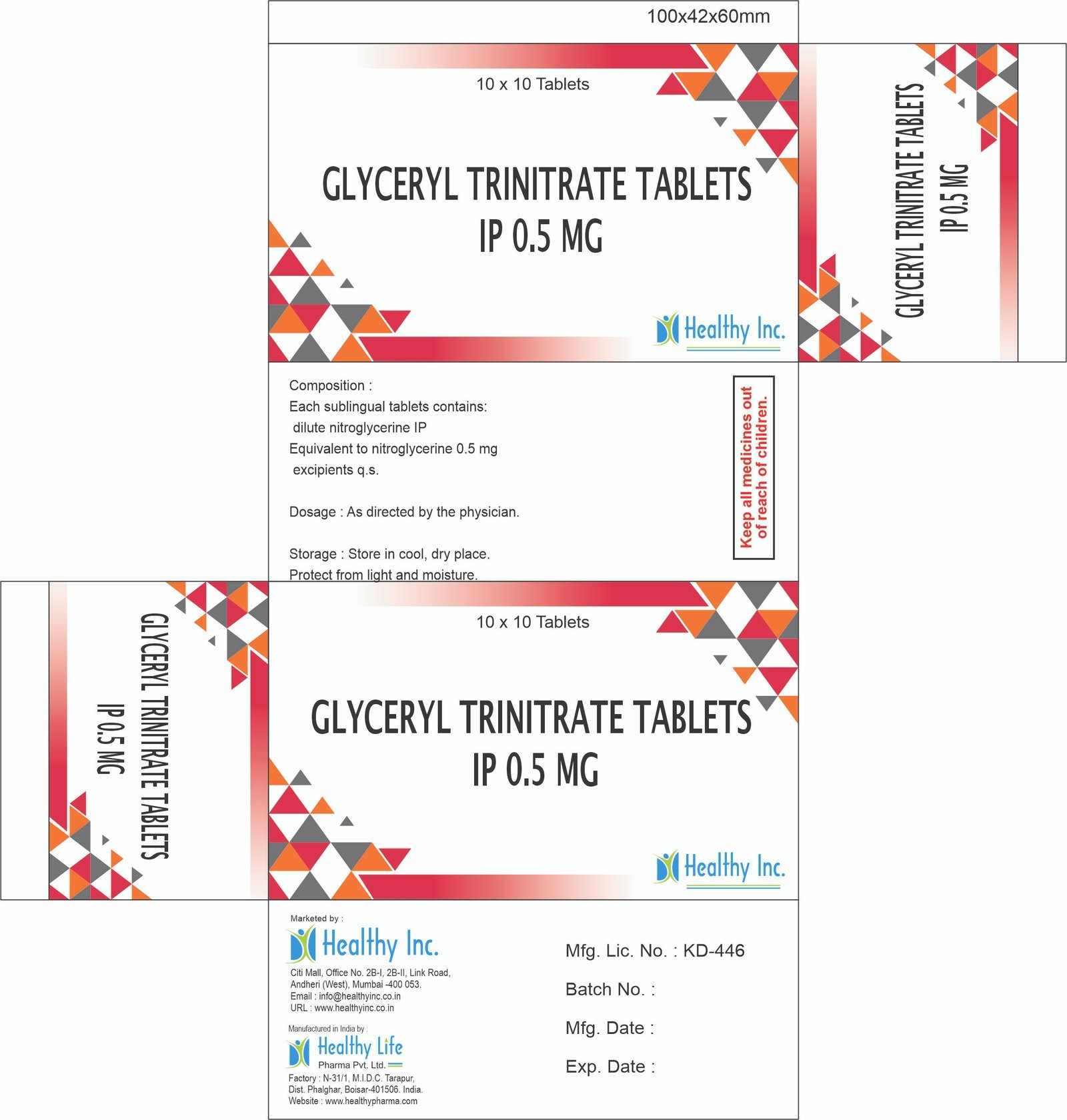
Glyceryl Trinitrate Tablets
FreeGLYCERYL TRINITRATE TABLETS IP 0.5 MG
Each sublingual tablets contains: Dilute Nitroglycerin IP Equivalent to Nitroglycerin 0.5 mg
Excipients q.s.Usage: – Treat angina (chest pain)
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic
Griseofulvin Tablets
FreeGriseofulvin Tablets IP 125 mg
Each Uncoated tablet contains :
Griseofulvin IP 125 mg
Excipients q.sGRIDORN – 125
(Griseofulvin Tablets IP 125 mg)
Each Uncoated tablet contains :
Griseofulvin IP 125 mgGRISEOFULVIN TABLETS IP 250 MG Each uncoated tablet contains : Griseofulvin IP 250 mg Excipients q.s.
Griseofulvin Tablets IP 500 mg
Each Uncoated tablet contains :
Griseofulvin IP 500 mgGRIDORN-125
(Griseofulvin tablets BP 125 mg)
Each uncoated tablet contains :
Griseofulvin BP 125 mgUsage: – Treat skin infections such as jock itch, athlete’s foot, and ringworm
Category: – Antifungal Drugs
Therapeutic category: – Antifungal
Haematinic Capsules
FreeHaematinic capsules are combination supplements containing essential nutrients required for the production of healthy red blood cells and haemoglobin. While specific formulations vary by brand, they typically contain the following categories of ingredients:
Haloperidol Injection
FreeComposition
Each ml Contains :Haloperidol I.P. …………… 5 mg
Methyl Paraben I.P 0.18 % W/V( As Preserva9ve )
Propyl Paraben I.P 0.02 % W/V( As Preserva9ve )
Water for Injec9on I.P. Q.S. 1mlUsage: – Treat certain mental/mood disorders
Category: – Antipsychotic / CNS injection
Therapeutic category: –Haloperidol Injection
Haloperidol Tablets
FreeHaloperidol Tablets BP 1.5 mg
Each Uncoated tablet contains :
Haloperidol BP 1.5 mgH PERIDOL-2
Haloperidol Tablets BP 2 mg
Each Uncoated tablet contains :
Haloperidol BP 2 mgHaloperidol Tablets BP 5 mg
Each Uncoated tablet contains :
Haloperidol BP 5 mgHaloperidol Tab BP 5 mg Haloperidol BP 5 mg Pantone Excipent QS
Haloperidol Tablets BP 5 mg
Each uncoated tablet contains:
Haloperidol B.P. 5 mg.Usage: – Treat certain mental/mood disorders
Category: – Antipsychotic / CNS Drugs
Therapeutic category: –
Heparin Sodium Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Glass Vials.
Active Ingredient Strength Volume Heparin Sodium USP/BP 5,000 IU (1,000 IU/ml) 5 ml Multi-Dose Vial Heparin Sodium USP/BP 25,000 IU (5,000 IU/ml) 5 ml Multi-Dose Vial Preservative Benzyl Alcohol 0.95% v/v *Available in preservative-free versions for neonatal use upon request.
HP Kit (Pantoprazole,Metronidazole & Clarithromycin Tablets)
FreeProduct Composition & Strength
We supply this product as a Precision-Packed, Multi-Component Combi-Kit, packed exclusively in highly secure, day-labeled Alu-Alu blister strips to ensure the absolute chemical stability of all three distinct APIs while forcing flawless patient compliance.
Active Component (Per Daily Blister) Strength & Form Primary Clinical Function Pantoprazole Sodium USP/Ph.Eur.
(2 Tablets per kit)40 mg (Gastro-Resistant / Enteric Coated) The Environmental Modulator: Suppresses acid, heals the ulcer, and creates the required pH for antibiotic survival. (Must be heavily enteric-coated to survive the stomach and absorb in the duodenum). Clarithromycin USP/Ph.Eur.
(2 Tablets per kit)500 mg (Film-Coated) The Primary Bacteriostatic: Halts bacterial protein synthesis. Heavy film-coating masks the intensely bitter, metallic macrolide taste. Metronidazole USP/Ph.Eur.
(2 Tablets per kit)400 mg / 500 mg (Film-Coated) The Bactericidal Finisher: Destroys bacterial DNA. Crucial alternative to Amoxicillin for penicillin-allergic demographics or regions with high amoxicillin resistance. *Pack Sizes: 1 Day Kit (containing 6 tablets total: 1 of each for Morning, 1 of each for Evening). Generally supplied in 7-Day or 14-Day Full Therapy Cartons.
Hucog
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection accompanied by a solvent (Sodium Chloride Injection).
Brand Reference Strength Primary Use Hucog / Corion 2000 IU Pediatric Cryptorchidism / Male Fertility Hucog / Corion 5000 IU Standard Ovulation Trigger Hucog / Corion 10000 IU IVF / Assisted Reproductive Technology (ART) Solvent 0.9% NaCl 1 ml or 2 ml Ampoule *Pack Sizes: Single Vial with Ampoule or Tray of 10.
Hydrochlorothiazide Tablets
FreeHydrochlorothiazide Tablets IP 50 mg
Each Uncoated Tablet contains :
Hydrochlorothiazide IP 50 mgHydrochlorothiazide Tabtes BP 25 mg Each uncoated tablet contains : Hydrochlorothiazide BP 25 mg , Excipients q.s.
Hydrochlorothiazide tablets USP
Each Uncoated tablet contains :
Hydrochlorothiazide USP 25 mgHydrochlorothiazide Tablets USP 50 mg
Each Uncoated Tablet contains :
Hydrochlorothiazide USP 50 mgUsage: – Treat edema
Category: – Diuretic Drugs
Therapeutic category: – Diuretic
Hydrocortisone Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in glass vials.
Active Ingredient Strength Primary Use Hydrocortisone Sodium Succinate USP/BP Equivalent to 100 mg Hydrocortisone Standard Adult / Pediatric Emergency Hydrocortisone Sodium Succinate USP/BP Equivalent to 250 mg Severe Asthma / Sepsis Pulse Hydrocortisone Sodium Succinate USP/BP Equivalent to 500 mg High-Dose ICU Protocol Excipients Sodium Phosphate / Sodium Hydroxide Buffer / pH Adjuster *Pack Sizes: Tray of 1 Vial + 1 Ampoule (WFI), 10 Vials, or 50 Vials.
Product Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in glass vials.
Active Ingredient Strength Primary Use Hydrocortisone Sodium Succinate USP/BP Equivalent to 100 mg Hydrocortisone Standard Adult / Pediatric Emergency Hydrocortisone Sodium Succinate USP/BP Equivalent to 250 mg Severe Asthma / Sepsis Pulse Hydrocortisone Sodium Succinate USP/BP Equivalent to 500 mg High-Dose ICU Protocol Excipients Sodium Phosphate / Sodium Hydroxide Buffer / pH Adjuster *Pack Sizes: Tray of 1 Vial + 1 Ampoule (WFI), 10 Vials, or 50 Vials.
Hydrocortisone injection
FreeComposition
Hydrocortisone Sodium 100mg vial+wfi
Usage: – used to treat a variety of medical conditions.
Category: –Allergic injection
Therapeutic category: – Allergic Reactions
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Hydrocortisone Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Scored Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the micronized steroid payload.
Active Ingredient Strength Primary Clinical Function Hydrocortisone USP/Ph.Eur. 5 mg / 10 mg (Scored) Titration & Afternoon Standard: Highly precise, low-dose units engineered for pediatric CAH dosing and the critical “afternoon taper” in adult replacement therapy. Hydrocortisone USP/Ph.Eur. 20 mg (Scored) Morning Surge & Acute Flare Standard: High-efficacy dosing to replicate the natural morning cortisol spike, or for short-term suppression of acute severe allergic or inflammatory conditions. Excipients Lactose Monohydrate / Microcrystalline Cellulose / Sodium Starch Glycolate / Magnesium Stearate Diluent / Binder / Superdisintegrant / Lubricant (Engineered utilizing Micronized API—the steroid particles are milled to a microscopic size before blending to guarantee immediate, consistent gastric absorption, which is critical to avoid unpredictable hormone spikes) *Pack Sizes: Bottles of 100 or 10×10 Blisters (Optimized specifically for strict chronic, lifelong endocrinology dispensing regimens).
Hydroxy Progesterone Caproate veterinary injection
FreeComposition
Hydroxy Progesterone Caproate INJ. 2 ML
Hydroxy Progesterone Caproate INJ. 3 ML
Usage: – used to synchronize estrus (heat) cycles in female animals
Category: – veterinary medicine
Therapeutic category: – veterinary medicine
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Hyoscine Butylbromide Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Glass Ampoules.
Active Ingredient Strength Volume Hyoscine Butylbromide USP/BP 20 mg 1 ml Ampoule (Standard Dose) Excipients Sodium Chloride / Water for Injection Isotonic Solution *Pack Sizes: Tray of 5 Ampoules, 10 Ampoules, or 100 Ampoules.
Hyoscine Butylbromide Tablets
FreeProduct Composition & Strength
We supply this product as Sugar Coated Tablets or Film Coated Tablets. The coating is essential to mask the naturally bitter taste of the active ingredient and ensure patient compliance.
Active Ingredient Strength (Standard) Therapeutic Role Hyoscine Butylbromide BP/USP 10 mg Muscarinic Antagonist (Antispasmodic) Excipients Q.S. Calcium Hydrogen Phosphate / Maize Starch Stabilizing Core Matrix Coating Agent Q.S. Sucrose / Talc / Acacia Traditional Sugar Coating Ibuprofen & Paracetamol Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets. The coating protects the gastric mucosa from direct contact with Ibuprofen and masks the bitter taste of the active ingredients.
Active Ingredient Strength (Standard) Therapeutic Role Ibuprofen BP/USP 200 mg NSAID (Anti-inflammatory/Analgesic) Paracetamol BP/USP 325 mg Analgesic & Antipyretic Excipients Q.S. Starch / Croscarmellose Sodium Disintegrant & Binder Ibuprofen Solution For Infusion
FreeProduct Composition & Strength
We supply this product in two formats: Ready-to-Use Infusion and Concentrate for Dilution.
Active Ingredient Strength Format Ibuprofen (with Arginine) 400 mg / 100 ml Ready-to-Use (IV Bag/Bottle) Ibuprofen (with Arginine) 400 mg / 4 ml Concentrate (Vial – Must Dilute) Solubilizer L-Arginine Ensures Water Solubility *Pack Sizes: 100 ml Bottle (Single) or Tray of 10 Vials.
Ibuprofen Tablets
FreeIbuprofen Tablets IP 200 mg
Each Sugar Coated tablet contains :
Ibuprofen IP 200 mgIbuprofen Tablets IP 400 mg
Each Sugar Coated tablet contains :
Ibuprofen IP 400 mgIbupramol
Ibuprofen & Paracetamol Tablets
Each Uncoated tablet contins :
Ibuprofen IP 200 mg
Paracetamol IP 325 mgIBUPROFEN TABLETS IP 400 MG Each Filmcoated Tablet contains: – Ibuprofen IP 400 mg
– Excipients – QSMakupar – 200
Ibuprofen Tablets BP
Each Sugarcoated Tablet contains :
Ibuprofen BP 200 mgMakupar – 400
Ibuprofen Tablets BP
Each Sugarcoated Tablet contains :
Ibuprofen BP 400 mgIbu – 600
Ibuprofen Tablets BP
Each Filmcoated tablet contains :
Ibuprofen BP 600 mgIBUPROFEN TABLETS BP 400 MG
Each Filmcoated Tablets Contains:
Ibuprofen BP 400 mg
– Erythrosine –
– Excipients – QSUsage: – Painkiller
Category: – Anti Analgesic Painkiller Drugs
Therapeutic category: – NSAID, Analgesic, Antipyretic, Anti Inflammatory
Ibuprofen with Paracetamol Tablets
FreeProduct Composition & Standard Strengths
We supply this product in standardized Fixed-Dose Combinations (FDCs) designed for maximum therapeutic synergy and patient safety.
Active Ingredient Standard Strengths (per Tablet) Primary Clinical Function Ibuprofen IP/BP/USP 200 mg / 400 mg The Anti-Inflammatory: Reduces physical swelling. Paracetamol IP/BP/USP 325 mg / 500 mg The Analgesic: Raises the chemical pain threshold. Excipients Pharma Grade The Matrix: Ensures stability and rapid release. Imatinib Capsules
FreeImatinib capsules contain the active oncology drug and a specific set of inactive ingredients designed to stabilize the compound for oral delivery.
Imatinib Mesylate Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips or heavy-gauge HDPE bottles to ensure the absolute chemical stability of the mesylate salt.
Active Ingredient Strength Primary Clinical Function Imatinib (as Mesylate) USP/Ph.Eur. 100 mg Pediatric & Titration Standard: Base therapeutic unit utilized for weight-based pediatric leukemia dosing, or for highly precise adult dose adjustments during acute hematological toxicity. Imatinib (as Mesylate) USP/Ph.Eur. 400 mg Global Adult Oncology Standard: High-efficacy adult maintenance dose, typically taken once daily for the chronic phase of CML, or twice daily for accelerated phases and GIST. Excipients Microcrystalline Cellulose / Crospovidone / Hypromellose / Colloidal Silicon Dioxide / Magnesium Stearate / Premium Iron-Oxide Film Diluent / Superdisintegrant / Binder / Glidant / Heavy Film-Coating (Engineered specifically to lock the cytotoxic API safely inside the tablet, completely preventing handler and pharmacist exposure to the carcinogenic dust) *Pack Sizes: 10×10 Alu-Alu Blisters or Bottles of 30 (Optimized specifically for strict 30-day chronic oncology dispensing regimens).
Imidapril Hydrochloride Tablets
FreeIMAPRIL TABLETS 5 MG
IMIDAPRIL HYDROCHLORIDE 5 MG
EACH FILM COATED TABLET CONTAINS :
IMIDAPRIL HYDROCHLORIDE 5 MGIMAPRIL TABLETS 10 MG
IMIDAPRIL HYDROCHLORIDE 10 MG
EACH FILM COATED TABLET CONTAINS :
IMIDAPRIL HYDROCHLORIDE 10 MGUsage: – reduce high blood pressure
Category: – Hypertensive / Cardiac Drugs
Therapeutic category: – Hypertensive
Imipenem & Cilastatin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Buffered Lyophilized Powder for Injection in glass vials.
Active Ingredient Strength Role Imipenem USP/BP 500 mg Antibiotic (Carbapenem) Cilastatin Sodium USP/BP Equivalent to 500 mg Cilastatin DHP-I Inhibitor Buffer Sodium Bicarbonate pH Stabilizer *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials.
Imipramine Tablets
FreeFraneel 10 Tablets
Imipramine tablets BP 10 mg
Each Sugar Coated tablet contains :
Imipramine Hydrochloride BP 10 mgFRANEEL 25 MG IMIPRAMINE TABLET 25 MG
EACH SUGAR COATED TABLET CONTAINS: IMIPRAMINE HYDROCHLORIDE BP 25 MG EXCIPIENTS- QSUsage: – Treat depression
Category: – Antidepressant drugs
Iron Folic Acid Tablets
FreeIRON WITH FOLIC ACID TABLETS IP
Each Enteric Coated Tablet Contains: –
Dried Ferrous Sulphate 335 mg
Eq. to Elemental iron IP (100 mg)
Folic Acid IP (0.5 mg)
Indigo Caramine (- )
Excipients (- )Iron with Folic acid Tablets IP
Each sugarcoated tablet contains:Ferrous Sulphate IP (200 mg)
Eq. to Elemental Iron IP (60 mg)
Folic Acid IP (0.5 mg)
Ponceau 4R (In House) (- )
Excipients (- QS)Usage: – Treat or prevent low iron in the body
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs / Medicine
Isoniazid & Pyridoxine Hydrochloride Tablets
FreeProduct Composition & Strength
We supply this product as Uncoated or Film Coated Tablets. The ratio of Isoniazid to Pyridoxine is carefully calibrated to meet international clinical guidelines for neuroprotection.
Active Ingredient Strength (Standard) Therapeutic Role Isoniazid BP/USP 300 mg Primary Antitubercular (Bactericidal) Pyridoxine Hydrochloride BP/USP 10 mg / 25 mg / 50 mg Neuroprotective Vitamin (B6) Excipients Q.S. Starch / Lactose / Magnesium Stearate Tablet Core Matrix Isoniazid & Pyridoxine Hydrochloride Tablets
FreeProduct Composition & Strength
We supply this product as Uncoated or Film Coated Tablets. The ratio of Isoniazid to Pyridoxine is carefully calibrated to meet international clinical guidelines for neuroprotection.
Active Ingredient Strength (Standard) Therapeutic Role Isoniazid BP/USP 300 mg Primary Antitubercular (Bactericidal) Pyridoxine Hydrochloride BP/USP 10 mg / 25 mg / 50 mg Neuroprotective Vitamin (B6) Excipients Q.S. Starch / Lactose / Magnesium Stearate Tablet Core Matrix Isoniazid Tablets
FreeIsoniazide Tablets IP
Each Coated Tablet Contains :
Isoniazide IP 50 mgIsoniazid Tablet IP 100 mg
Each Coated Tablets Contains :
Isoniazid IP 100 mgIsoniazid Tablet IP 300 mg
Each Coated Tablets Contains :
Isoniazid IP 300 mgPYRINH ISONIAZIDE WITH PYRIDOXINE TABLETS Each Uncoated tablet contain: – Isoniazide IP 100 MG
– Pyridoxin Hyrochloride IP 5 MGISONIAZID TABLETS BP 100 MG
Each Film Coated Tablet contains:
Isoniazid BP 100 mgISONIAZID TABLETS BP 300 MG
Each filmcoated tablet contains:
Isoniazid BP 300 mgUsage: – Treat active tuberculosis (TB) infections
Category: – Anti tuberculosis drugs
Therapeutic category: –
Isosorbide Mononitrate Tablets
FreeProduct Composition & Strength
We supply this product as Immediate Release Tablets (20 mg). We also offer Extended Release (ER/SR) variants for once-daily dosing through our contract manufacturing division.
Active Ingredient Strength (Standard) Therapeutic Role Isosorbide Mononitrate BP/USP 20 mg Organic Nitrate / Vasodilator Excipients Q.S. Microcrystalline Cellulose / Lactose Stabilizing Matrix Isosorbide Tablets
FreeProduct Composition & Variants
We supply both Mononitrate and Dinitrate forms in immediate and sustained-release formats.
Variant Type Standard Strengths Primary Clinical Use Isosorbide Mononitrate 10mg, 20mg, 40mg Prophylaxis: Long-term prevention of angina. Isosorbide Dinitrate 5mg, 10mg, 20mg Acute & Chronic: Short and long-term heart failure. Sustained Release (SR) 30mg, 60mg Once-Daily: For improved patient compliance. Isosorbide Tablets
FreeIsosorbide Dinitrate Tablets IP 5 mg
Each Uncoated tablet contains :
Diluted Isosorbide Dinitrate IP
Eq. To Isosorbide Dinitrate 5 mgIsosorbide Dinitrate Tablets IP 10 mg
Each Uncoated tablet contains :
Diluted Isosorbide Dinitrate IP
Eq.To Isosobide Dinitrate 10 mgIsosorbide Dinitrate Tablets BP 5 mg
Each uncoated tablet contains:
Diluted Isosorbide Dinitrate BP
Eq. to Isosorbide Dinitrate 5 mgIsosorbide Mononitrate Tablets 10 mg
Each Uncoated tablet contains :
Diluted Isosorbide Mononitrate
Eq.To Isosobide mononitrate 10 mgM- Trate tablets
Isosorbide 5 -Mononitrate Tablet BP
Each Uncoated tablet contains :
Diluted Isosorbide 5- Mononitate BP
eq to Isosorbide 5- Mononitate 20 mgUsage: – Prevent chest pain caused by angina
Category: – Anti Cardiovascular drugs
Therapeutic category: – Cardiovascular Agent
Isotretinoin Capsules
FreeProduct Composition & Strengths
We supply this product as Liquid-Filled Soft Gelatin Capsules, optimized for maximum systemic absorption.
Active Ingredient Strength (per Softgel) Primary Clinical Target Isotretinoin IP/BP/USP 10 mg Standard Starting Dose: For mild-to-moderate systemic treatment. Isotretinoin IP/BP/USP 20 mg Therapeutic Dose: For severe, cystic, or conglobate acne. Excipients Soybean Oil / Beeswax The Vehicle: Ensures lipid-solubility for better absorption. Isoxsuprine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Immediate-Release or Sustained-Release Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the active pharmaceutical ingredient.
Active Ingredient Strength Primary Clinical Function Isoxsuprine Hydrochloride USP/Ph.Eur. 10 mg (IR) Acute Titration Standard: Immediate-release base unit used for the rapid initiation of therapy or carefully titrated maintenance in sensitive patients. Isoxsuprine Hydrochloride USP/Ph.Eur. 40 mg (SR) Obstetrics Maintenance Standard: High-efficacy, slow-release matrix designed specifically to provide 12-hour continuous tocolysis, vastly improving maternal compliance and protecting the cardiovascular system. Excipients Hypromellose (HPMC) / Microcrystalline Cellulose / Lactose Monohydrate / Colloidal Silicon Dioxide / Magnesium Stearate ER Polymer / Diluent / Binder / Glidant / Lubricant (Engineered specifically utilizing premium Hypromellose matrices in the 40mg variant to form a robust hydrogel that slowly erodes in the gastrointestinal tract, ensuring steady-state plasma levels) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict obstetric and chronic vascular dispensing regimens).
Itopride Tablets
FreeItopride Tablets
Each Film Coated Tablet Contains:
Itopride Hydrochloride 50 mgUsage: – treatment of heartburn and nausea
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Itraconazole Capsules
FreeItraconazole capsules contain a combination of the active antifungal drug and specialized inactive ingredients designed to overcome the medication’s poor solubility in water.
Ivermactin veterinary injection
FreeComposition
Ivermactin INJ. 10 ML
Ivermactin INJ. 50 ML
Usage: – It is also used to control external parasites such as mites, lice, and ticks in animals.
Category: – veterinary injection medicine
Therapeutic category: –veterinary injection medicine
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Ivermectin & Clorsulon veterinary INJECTION
FreeCOMPOSITION
Ivermectin 1% & Clorsulon 10% VET INJ. 10 ML
Usage: – Treat or prevent Common Veterinary Uses
Category: –Common Veterinary injection
Therapeutic category: Veterinary injection
Pcd pharma franchise:-
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Ivermectin Tablets
FreeIvermectin Tablets 3 mg
Each tablet contains :
Ivermectin BP 3 mgIvermectin Tablets 6 mg
Each Uncoated tablet contains :
Ivermectin IP 6 mgUsage: – Parasitic infections
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic, Antiparasitic
Ketoconazole Tablets
FreeKetoconazole Tablets IP
Each Uncoated Tablet contains :
Ketoconazole IP 200 mgKetoconazole Tablets USP
Each Uncoated Tablet contains :
Ketoconazole USP 200 mgUsage: – Some kinds of fungal infections
Category: – Antifungal drugs
Therapeutic category: – Antifungal
Ketoprofen veterinary injection
FreeComposition
Ketoprofen VET INJ. 15 ML
Ketoprofen VET INJ. 100 ML
Usage: –It is used in various animal species, including dogs, cats, horses, and livestock.
Category: – veterinary injection
Therapeutic category: –veterinary injection
Pcd pharma franchise:-
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Ketorolac Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Amber Glass Ampoules.
Active Ingredient Strength Volume Ketorolac Tromethamine USP/BP 30 mg 1 ml Ampoule (Standard Dose) Ketorolac Tromethamine USP/BP 60 mg 2 ml Ampoule (High Dose) Excipients Alcohol USP / Sodium Chloride Solubilizer / Isotonicity *Pack Sizes: Tray of 5 Ampoules, 10 Ampoules, or 50 Ampoules.
Ketorolac injection
FreeComposition
INJ (1 ML) Each ml contains –
Ketorolac Tromethamine 30 mg + WFI IP Q.S.
Usage: – Relieve moderately severe pain
Category: – Anti-inflammatory injection (NSAIDs)
Therapeutic category: – NSAIDs
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
L-Methylfolate Calcium ,Pyridoxal-5-Phosphate & Mecobalamin Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Photostable Film-Coated Tablet, packed exclusively in highly secure, light-blocking Alu-Alu blister strips to ensure the absolute chemical survival of the three delicate coenzymes.
Active Ingredient Strength Primary Clinical Function L-Methylfolate Calcium USP/Ph.Eur. 1 mg Active Vitamin B9: Direct methyl-donor for homocysteine remethylation and massive neurotransmitter synthesis. Pyridoxal-5-Phosphate (P5P) USP/Ph.Eur. 0.5 mg Active Vitamin B6: Immediate catalyst for transsulfuration; drastically reduces the risk of paradoxical sensory neuropathy seen with cheap Pyridoxine. Mecobalamin (Methylcobalamin) USP/Ph.Eur. 1500 mcg Active Vitamin B12: Direct peripheral nerve repair, myelin synthesis, and essential co-factor for homocysteine neutralization. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Magnesium Stearate / Titanium Dioxide / Premium Opaque Opadry Film Diluent / Superdisintegrant / Lubricant / Heavy UV-Blocking Coating (Engineered specifically to form an impenetrable light barrier, completely preventing the photolabile degradation of the active B12 and folate components) *Pack Sizes: 10×10 Alu-Alu Blisters (Optimized specifically for strict, 3-to-6 month chronic neurology and diabetic dispensing regimens).
L-Ornithine-L-Aspartate Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Hypertonic Solution in Glass Ampoules. (Must be diluted).
Active Ingredient Strength Concentration L-Ornithine-L-Aspartate 5 g (5000 mg) 500 mg/ml (10 ml Ampoule) Excipients Water for Injection – *Pack Sizes: Tray of 5 Ampoules or 10 Ampoules.
Lactobacillus SporogenesTablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Low-Moisture Matrix Tablet, packed exclusively in highly secure Alu-Alu blister strips or tightly sealed Alu-PVC blisters to prevent premature ambient moisture from triggering spore germination prior to ingestion.
Active Ingredient Strength Primary Clinical Function Lactobacillus sporogenes (Bacillus coagulans) Spores 60 Million Spores (CFU) Global Microbiome Standard: High-density, survival-guaranteed spore count to aggressively recolonize the intestinal lining, halt acute diarrhea, and outcompete opportunistic pathogens following heavy antibiotic therapy. Excipients Microcrystalline Cellulose / Lactose Anhydrous / Croscarmellose Sodium / Colloidal Silicon Dioxide / Magnesium Stearate Diluent / Superdisintegrant / Glidant / Lubricant (Engineered specifically utilizing ultra-low moisture excipients during dry-blending to ensure the dormant spores are not accidentally “woken up” and destroyed by water activity on the factory floor) *Pack Sizes: 10×10 Blisters or Bottles of 100 (Optimized specifically for massive-volume retail pharmacy and pediatric clinic dispensing).
Lamotrigine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet or Rapid-Dispersible (DT) Tablet, packed exclusively in highly secure Alu-PVC or Alu-Alu blister strips, frequently customized into highly regulated Titration Starter Kits.
Active Ingredient Strength Primary Clinical Function Lamotrigine USP/Ph.Eur. 25 mg (Starter Kit) The SJS-Prevention Standard: Mandatory base unit for the strict, slow titration schedule required to prevent fatal cutaneous reactions. Often formulated as a Dispersible Tablet (DT) for pediatric epilepsy. Lamotrigine USP/Ph.Eur. 50 mg / 100 mg / 200 mg Global Maintenance Standard: High-efficacy adult maintenance doses for lifelong seizure control and bipolar stabilization. Excipients Microcrystalline Cellulose / Sodium Starch Glycolate / Povidone / Sucralose & Flavoring (If DT) / Magnesium Stearate Diluent / Superdisintegrant / Binder / Sweetener (Engineered specifically to either swallow intact or disperse rapidly in a teaspoon of water for children or elderly psychiatric patients suffering from dysphagia) *Pack Sizes: 10×10 Blisters, Bottles of 100, or Dedicated 28-Day Titration Calendar Packs (Optimized specifically to eliminate caregiver dosing errors during the critical initiation phase).
Lamotrigine Tablets
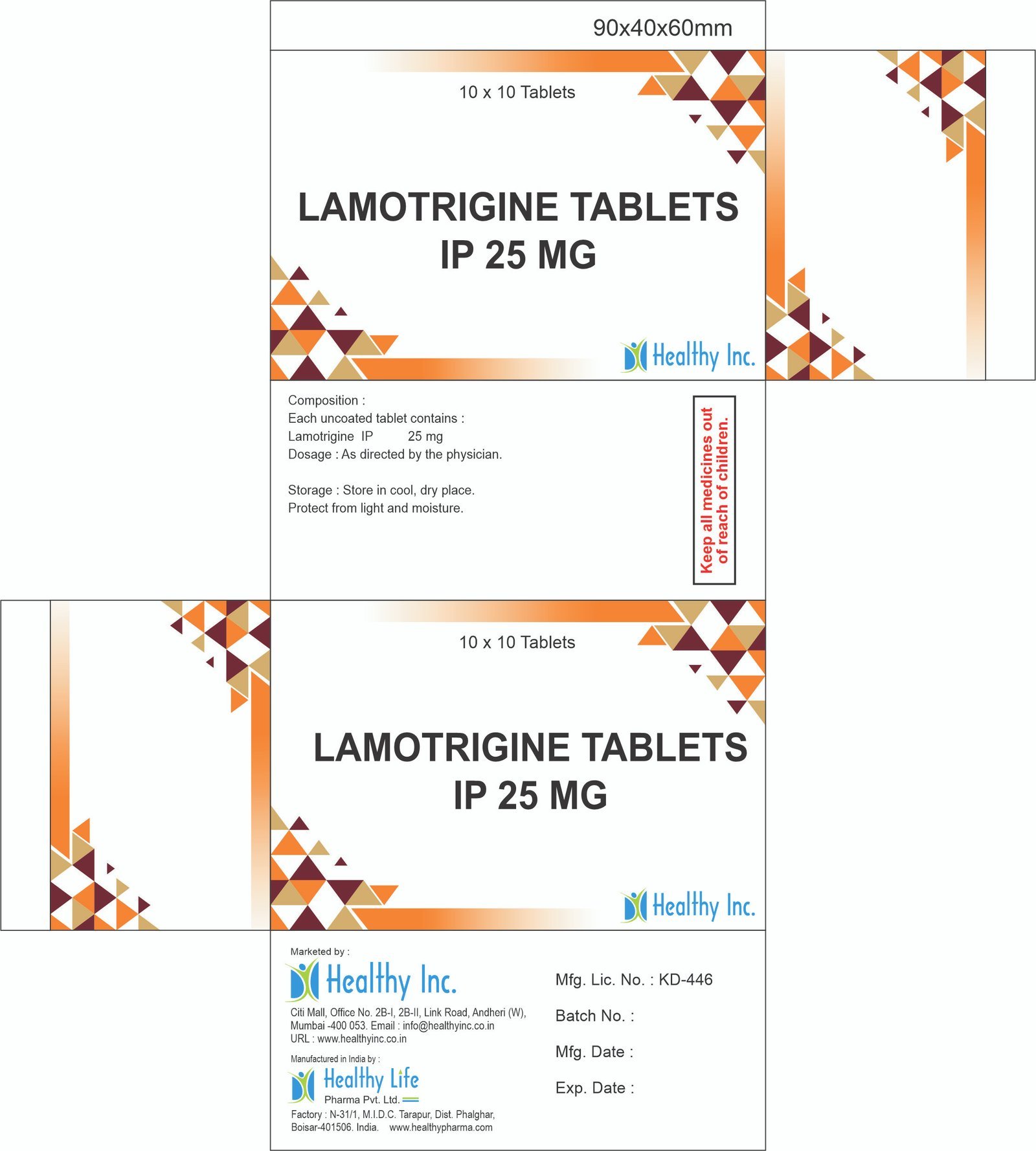
FreeLamotrigine Tablets IP 25 mg
Each uncoated tablet contains :
Lamotrigine IP 25 mgUsage: – Prevent and control seizures
Category: – Antiepileptic drugs
Therapeutic category: –
Lansoprazole Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in glass vials.
Active Ingredient Strength Form Lansoprazole Sodium USP/BP Equivalent to 30 mg Lansoprazole White Lyophilized Cake Excipients Mannitol / Meglumine / Sodium Hydroxide Bulking Agent / pH Stabilizer *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials.
Leflunomide Tablets
FreeLeflunomide Tablets
Each Film Coated Tablet Contains:
Leflunomide BP 20 mgUsage: – Rheumatoid arthritis
Category: – Antiarrhytmics drugs
Therapeutic category: –
Lenalidomide Capsules
FreeLenalidomide capsules (brand name Revlimid) are a potent immunomodulatory medication used to treat certain types of cancers, such as multiple myeloma and myelodysplastic syndromes.
Letrozole Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the micro-dosed endocrine disruptor.
Active Ingredient Strength Primary Clinical Function Letrozole USP/Ph.Eur. 2.5 mg Global Oncology & Fertility Standard: The universal therapeutic unit utilized for 5-to-10 year daily adjuvant breast cancer therapy, or short 5-day cyclic dosing for ovulation induction. Excipients Lactose Monohydrate / Microcrystalline Cellulose / Sodium Starch Glycolate / Colloidal Silicon Dioxide / Magnesium Stearate / Premium Opadry Film Diluent / Binder / Superdisintegrant / Glidant / Slipper-Coating (Engineered specifically using elite geometric dilution to ensure the microscopic 2.5mg active payload is flawlessly distributed throughout the tablet, guaranteeing absolute content uniformity) *Pack Sizes: 10×10 Blisters or Bottles of 30 (Optimized specifically for strict, heavily monitored chronic oncology dispensing or monthly fertility cycles).
Levamisole Hydrochloride veterinary injection
FreeComposition
Levamisole Hydrochloride INJ. 30ML
Usage: – It is typically administered as an injectable solution or added to the animal’s feed.
Category: – parasitic infections veterinary injection
Therapeutic category: –parasitic infections veterinary injection
Levamisole Tablets
FreeJantrax
Levamisole Hydrochloride Tablets USP
Each Filmcoated tablet contains :
Levamisole Hydrochloride USP
Eq. To Levamisole 40 mgUsage: – Treat infections of the worm
Category: – Anthelmintic drugs
Therapeutic category: –
Levetiracetam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile, Clear, Colorless Solution in glass vials.
Active Ingredient Strength Concentration Levetiracetam USP/BP 500 mg 100 mg/ml (in 5 ml Vial) Excipients Sodium Acetate / Glacial Acetic Acid Buffer / pH Adjuster (5.5) *Pack Sizes: Tray of 5 Vials, 10 Vials, or Single Vial packs.
Levocetirizine Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets. The small, easy-to-swallow tablets are engineered for rapid disintegration to facilitate quick relief from acute allergic flares.
Active Ingredient Strength (Standard) Therapeutic Role Levocetirizine Dihydrochloride BP/USP 5 mg Potent H1-Receptor Antagonist Excipients Q.S. Microcrystalline Cellulose / Lactose Diluent & Disintegrant Coating Agent Q.S. Opadry® White Protective Film Coating Levofloxacin injection
FreeComposition
Levofloxacin Hemihydrate, 500mg + Sodium Chloride IP
900mg & Water for injection IP q.s
Levofloxacin 500mg & Dextrose Anhydrous 5.0% w/v injection
Pcd pharma franchise:-
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Levofloxacin Tablets
FreeLevofloxacin tablets IP 250 mg
Each Filmcoated tablet contains :
Levofloxacin Hemihydrate IP
Eq. To Levofloxacin 250 mgLevofloxacin tablets IP 500 mg
Each Filmcoated tablet contains :
Levofloxacin Hemihydrate IP
Eq. To Levofloxacin 500 mgLevofloxacin Tablets 750 mg
Each Film Coated Tablet Contains :
Levofloxacin Hemihydrate
Eq To Levofloxacin 750 mgLevofloxacin tablets 250 mg
Each Filmcoated tablet contains :
Levofloxacin Hemihydrate
Eq. To Levofloxacin 250 mgLevofloxacin tablets 500 mg
Each Filmcoated tablet contains :
Levofloxacin Hemihydrate
Eq. To Levofloxacin 500 mgUsage: – Variety of bacterial infections
Category: – Antibiotic Drugs
Therapeutic category: – Antibiotic
Levonorgestrel Emergency Contraceptive Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Single-Dose Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the potent steroid molecule.
Active Ingredient Strength Primary Clinical Function Levonorgestrel USP/Ph.Eur. 1.5 mg Global Emergency Contraceptive Standard: A massive, single-dose payload designed to instantly halt ovulation within the critical 72-hour post-coital window. Excipients Lactose Monohydrate / Maize Starch / Povidone K30 / Colloidal Silicon Dioxide / Magnesium Stearate Diluent / Binder / Glidant / Lubricant (Engineered specifically utilizing advanced micronized API to ensure ultra-rapid gastric dissolution, guaranteeing the hormone hits the bloodstream fast enough to beat the LH surge) *Pack Sizes: 1-Tablet Blister Packs (Optimized specifically for discrete, over-the-counter (OTC) retail and single-dispense NGO crisis kits).
Levosulpiride Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Rapid-Burst Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the benzamide derivative.
Active Ingredient Strength Primary Clinical Function Levosulpiride USP/Ph.Eur. 25 mg Global Gastroenterology Standard: Base therapeutic unit taken prior to meals for the rapid relief of functional dyspepsia, bloating, and mild gastroparesis. Levosulpiride USP/Ph.Eur. 50 mg / 75 mg Psychiatric & Severe Dysmotility Standard: High-efficacy adult therapy used for severe, treatment-resistant diabetic gastroparesis or utilized by psychiatrists for somatoform disorders and mild depression. Excipients Microcrystalline Cellulose / Sodium Starch Glycolate / Crospovidone / Colloidal Silicon Dioxide / Magnesium Stearate Diluent / Superdisintegrant / Binder / Glidant (Engineered specifically without heavy polymeric retardants to guarantee immediate, complete gastric dissolution within 10 to 15 minutes of ingestion, preparing the stomach for the incoming meal) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict, meal-dependent acute and chronic dispensing regimens).
Levothyroxine Tablets
FreeThyrostim Tablets
Levothyroxine Sodium Tablets BP
(Thyroxine Sodium tablets BP)
Each Uncoated tablet contains :
Levothyroxine Sodium BP
Eq. To Anhydrous Levothyroxine Sodium 0.1 mgTHYROXINE SODIUM TABLETS BP (LEVOTHYROXINE SODIUM TABLETS BP 0.25 MG)
EACH UNCOATED TABLET CONTAINS:
LEVOTHYROXINE SODIUM EQ. TO ANHYDROUS LEVOTHYROXINE SODIUM BP 0.25 MG
EXCIPIENTS Q.S.
Levothyroxine Sodium Tablets IP 50 MCG Each Uncoated tablet contains:Levothyroxine Sodium IP Eq. to Anhydrous Levothyroxine Sodium IP (50 mcg)
EXCIPENT (- QS)
THYROXINE 25 MCG
EACH UNCOATED TABLET CONTAINS:
LEVOTHYROXINE SODIUM EQ TO ANHYDROUS LEVOTHYROXINE SODIUM IP 25 MCGLevothyroxine Tablets IP 25 mcg Each uncoated tablet contains:
LEVOTHYROXINE SODIUM EQ TO ANHYDROUS LEVOTHYROXINE SODIUM IP (25 mcg)
Excipient (- QS)Levothyroxine Sodium Tablets IP 50 MCG Each Uncoated tablet contains: Levothyroxine Sodium IP Eq. to Anhydrous Levothyroxine Sodium IP (50 mcg)
EXCIPENT (- QS)
Usage: – Treat hypothyroidismCategory: – Thyroid Hormones drugs
Therapeutic category: – Thyroid Hormones
Lidocaine 2% Ampoules
FreeProduct Composition & Strength
We supply this product as a Sterile, Preservative-Free Solution in glass ampoules.
Active Ingredient Strength Total Content (2ml) Lidocaine Hydrochloride USP/BP 2% (20 mg/ml) 40 mg Total Content (5ml) 2% (20 mg/ml) 100 mg Excipients Sodium Chloride / Water for Injection Isotonic Solution *Available in 2ml, 5ml, and 10ml Type I Glass Ampoules.
Lidocaine Cartridges
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Type I Glass Cartridges.
Active Ingredients Strength Cartridge Volume Lidocaine Hydrochloride 2% (20 mg/ml) 1.8 ml / 2.2 ml Adrenaline (Epinephrine) 1:80,000 / 1:100,000 – Stabilizer Sodium Metabisulfite Antioxidant *Pack Sizes: Box of 50 or 100 Cartridges (Blister packed for sterility).
Lidocaine Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Glass Ampoules or Multi-dose Vials.
Active Ingredient Strength Common Application Lidocaine Hydrochloride 1% (10 mg/ml) Infiltration / Minor Suturing / Diagnostic Blocks Lidocaine Hydrochloride 2% (20 mg/ml) Major Nerve Blocks / Epidural / Anti-arrhythmic Excipients Sodium Chloride / Methylparaben (in vials) Isotonicity / Preservative *Available in preservative-free (PF) versions for spinal/epidural use.
Lignocain with Adrenaline veterinary injection
FreeComposition
Lignocain with Adrenaline VET 30ML
Usage: –combination medication commonly used in veterinary medicine for various purposes.
Category: – veterinary injection
Therapeutic category: – veterinary medicine
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Lignocaine Adrenaline Dental Cartridges
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Glass Cartridges with siliconized plungers.
Active Ingredients Strength Standard Cartridge Volume Lignocaine Hydrochloride 2% (20 mg/ml) 1.8 ml (Standard) Adrenaline (Epinephrine) 1:80,000 / 1:100,000 2.2 ml (Extended) Excipients Sodium Metabisulfite / NaCl Antioxidant / Isotonicity *Pack Sizes: Box of 50 Cartridges or 100 Cartridges (Blister packed).
Lignocaine Hydrochloride Injection
FreeComposition
Each ml Contains : Lignocaine Hydrochloride I.P. …21.33 mg
Sodium Chloride I.P. ……….. 6 mg
Methyl Paraben I.P ………… 1 mg
Water for Injection I.P. Q.S.Each ml Contains :
Lignocaine Hydrochloride I.P. ..21.33 mg
Adrenaline (As Adrenaline Bitartrate I.P)
……………………………………………0.01mg
Sodium Metabisulphate I.P. …… 1 mg
Methyl Paraben I.P ……… 1 mg
( As Preserva9ve )
Water for Injection I.P. Q.S.Usage: – It is primarily used to induce local anesthesia, meaning it temporarily numbs a specific area of the body
Category: –administered via injection
Therapeutic category: –anesthetic medicine
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Lignocaine Injection
FreeLignocaine injection (also known as Lidocaine) is a sterile, aqueous solution primarily used as a local anaesthetic and antiarrhythmic agent. Its composition varies depending on whether it is a single-dose or multi-dose formulation and whether it includes a vasoconstrictor.
Lincomycin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Glass Ampoules or Vials.
Active Ingredient Strength Volume Lincomycin Hydrochloride USP/BP 600 mg 2 ml Ampoule (300 mg/ml) – Standard Lincomycin Hydrochloride USP/BP 300 mg 1 ml Ampoule (300 mg/ml) – Pediatric Preservative Benzyl Alcohol 0.945% v/v *Pack Sizes: Tray of 10 Ampoules, 50 Ampoules, or 100 Ampoules.
Lincomycin Injection
FreeComposition
Each 2 ml Contains: Lincomycin Hydrochloride I.P. Equi. To
Lincomycin Base 300 mg.
Benzyl Alcohol I.P. 9 mg
Water for Injection I.P. Q.S.Usage: – used for the treatment of various bacterial infections
Category: – anti bacteria injection
Therapeutic category: – susceptible bacteria.
Pcd pharma franchise:-
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Linezolid injection
FreeComposition
Linezolid 600mg &
Dextrose Anhydrous 5% w/v injection
Usage: – Treat different types of bacterial infections
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Linezolide Tablets
FreeLinezolide Tablets
Each Film Coated Tablet Contains
Linezolide 600 mgUsage: – Treat different types of bacterial infections
Category: – Antibiotic drugs
Therapeutic category: – Antibiotics
Lisinopril Tablets
FreeLisinopril Tablets IP 2.5 mg
Each Film coated tablet contains:
Lisinopril IP 2.5 mgLisinopril Tablets IP 5 mg
Each Film coated tablet contains:
Lisinopril IP 5 mgLowpril
Lisinopril Tablet BP
Each Film Coated Tablet Contains:
Lisinopril Dihydrate BP
eq. To Lisinopril Anhydrous 20 mg
Usage: – Treat high blood pressure and heart failureCategory: – Anti Hypertensive / Cardiac Drugs
Lithium Carbonate Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute physical stability of the controlled-release polymers.
Active Ingredient Strength Primary Clinical Function Lithium Carbonate USP/Ph.Eur. 300 mg (IR) Acute Mania Standard: Immediate-release base unit used for the rapid, aggressive titration of serum lithium levels during an active, severe manic episode. Lithium Carbonate USP/Ph.Eur. 400 mg / 450 mg (ER/SR) Global Maintenance Standard: Extended-release therapy designed to flatten the pharmacokinetic curve, preventing toxic blood spikes and minimizing severe hand tremors and nausea during lifelong maintenance therapy. Excipients Hypromellose (HPMC) / Microcrystalline Cellulose / Sodium Starch Glycolate / Magnesium Stearate / Premium Opadry Film Hydrophilic ER Polymer / Diluent / Disintegrant / Lubricant (Engineered specifically using high-shear wet granulation to form a highly reliable, slow-eroding matrix that trickles the elemental lithium into the bloodstream over 12 hours) *Pack Sizes: Bottles of 100 or 10×10 Blisters (Optimized specifically for strict, massive-volume lifelong psychiatric dispensing regimens).
Loperamide Capsules
FreeLoperamide capsules (most commonly known by the brand name Imodium) are an anti-diarrheal medication used to slow down an overactive bowel. They contain a specific active salt and various inactive ingredients that form the capsule and stabilize the medicine
Loperamide Tablets
FreeLoperamide Tablets
Each Film Coated Tablet Contains:
Loperamide Hydrochloride USP 2 mg
Usage: – Treat diarrhoea (runny poo)Category: – Antidiarrheal drugs
Therapeutic category: – Antidiarrheal
Loratidine Tablets
FreeLotadin
Loratidine Tablets
Each Uncoated tablet contains :
Loratidine 10 mgUsage: – Temporarily relieve the symptoms of hay fever
Category: – Anti Allergic Drugs
Therapeutic category: – Antihistamine, Anti Allergic
Lorazepam Tablets
FreeLozepam 1
Lorazepam tablets BP 1 mg
Each Uncoated tablet contains:
Lorazepam BP 1 mgLozepam 2
Lorazepam tablets BP
Each Uncoated tablet contains:
Lorazepam BP 2 mgLOZEPAM – 2
Lorazepam Tablets USP 2 mg
Each Uncoated Tablet Contains:
Lorazepam USP 2 mgUsage: – Treat anxiety and sleeping problems
Category: – Anti Anxiety Drugs
Therapeutic category: – Anti Anxiety
Losartan Potassium Hydrochlorothiazide Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets (typically Yellow or White, Oval). The coating protects the hygroscopic Losartan from moisture.
Active Ingredient Strength (Standard) Therapeutic Role Losartan Potassium USP/BP 50 mg Vasodilator (ARB) Hydrochlorothiazide USP/BP 12.5 mg Diuretic (Thiazide) (High Strength Variant) 100 mg Losartan + 25 mg HCTZ Severe Hypertension Excipients Q.S. Microcrystalline Cellulose / Lactose / Magnesium Stearate Filler / Lubricant Losartan Potassium, Amlodipine Besylate Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets (typically White or Yellow, Oval/Round). The formulation uses the Besylate salt of Amlodipine for maximum stability.
Active Ingredient Strength (Standard) Therapeutic Role Losartan Potassium USP/BP 50 mg Vasodilator (ARB) Amlodipine Besylate USP/BP 5 mg (Eq. to Base) Vasodilator (CCB) (High Strength Variant) 50 mg Losartan + 10 mg Amlodipine
100 mg Losartan + 5 mg AmlodipineResistant Hypertension Excipients Q.S. Microcrystalline Cellulose / Sodium Starch Glycolate Filler / Super-Disintegrant Losartan Tablets
FreeLosartan Potassium Tablets IP 25 mg
Each Film Coated Tablets Contains:
Losartan Potassium IP 25 mgLosartan Potassium Tablet IP 50 mg
Each Film Coated Tabet Contains :
Losartan Potasium IP 50 mgLOSARTAN POTASSIUM & HYDROCHLOROTHIAZIDE TABLETS IP
Each Film Coated Tablet Contains :: Losartan potassium IP 50 mg
Hydrochlorothiazide IP 12.5 mg – Excipients – QSLOSARTAN POTASSIUM & AMLODIPINE TABLET IP
Each Film Coated Tablet Contains :
Losartan potassium IP 50mg
Amlodipine Besylate IP Eq. To Amlodipine 5 mg
Excipients q.sUsage: – Treat high blood pressure (hypertension)
Category: – Anti Cardiovascular Drugs
Therapeutic category: – Cardiovascular Agent, Antihypertensive
Magnesium Sulphate Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Hypertonic Solution in Glass Ampoules.
Active Ingredient Concentration Strength (per 2ml) Magnesium Sulphate BP/USP 50% w/v (Standard) 1 Gram (in 2 ml) Magnesium Sulphate BP/USP 25% w/v 0.5 Gram (in 2 ml) Magnesium Sulphate BP/USP 10% w/v 2 Grams (in 20 ml) Osmolarity Approx. 4060 mOsmol/L Highly Hypertonic *Pack Sizes: Tray of 10 Ampoules, 25 Ampoules, or 100 Ampoules.
Maxgalin
FreeMaxgalin is a brand of medication manufactured by Sun Pharmaceutical Industries Ltd., primarily used to treat neuropathic (nerve) pain, fibromyalgia, and certain types of seizures.
Mebendazole Tablets
FreeEmzole
Mebendazole Tablets USP 100 mg
Each Uncoated tablet contains :
Mebendazole USP 100 mgMeben – 500
Mebendazole Tablets USP
Each Uncoated tablet contains :
Mebendazole USP 500 mgUsage: – Roundworm and whipworm infections
Category: – Anthelmintic Drugs
Therapeutic category: – Anthelmintic, Antiprotozoal, Antiparasitic
Mebeverine Tablets
FreeMebeverine Hydrochloride Tablets IP
Each filmcoated tablet contains :
Mebeverine Hydrochloride IP 135 mgUsage: – Helps with muscle spasms
Category: – Anti Analgesic Painkiller Drugs
Therapeutic category: – Painkiller
Medroxyprogesterone Acetate Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Micronized Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the delicate steroid molecule.
Active Ingredient Strength Primary Clinical Function Medroxyprogesterone Acetate USP/Ph.Eur. 2.5 mg / 5 mg HRT Maintenance Standard: Continuous or sequential low-dose therapy for the prevention of endometrial hyperplasia in postmenopausal women receiving estrogen. Medroxyprogesterone Acetate USP/Ph.Eur. 10 mg Acute Gynecology Standard: High-efficacy adult dosing for the rapid arrest of abnormal uterine bleeding and the induction of withdrawal bleeding in secondary amenorrhea. Excipients Lactose Monohydrate / Microcrystalline Cellulose / Crospovidone / Magnesium Stearate Diluent / Binder / Superdisintegrant / Lubricant (Engineered specifically utilizing advanced micronized API to overcome the severe hydrophobicity of the steroid, ensuring maximum, predictable gastric absorption) *Pack Sizes: 10×10 Blisters or 14-Day Calendar Packs (Optimized specifically for strict cyclic or continuous gynecological dispensing regimens).
Mefanamic Acid & Dicyclomine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Rapid-Burst Tablet, packed exclusively in highly secure, moisture-resistant PVC/PVDC or Alu-Alu blister strips to ensure the absolute chemical stability of the combined APIs.
Active Ingredient Strength Primary Clinical Function Mefenamic Acid USP/Ph.Eur. 250 mg The Anti-Inflammatory Anchor: Rapidly reduces prostaglandin-induced pain, uterine inflammation, and heavy menstrual bleeding. Dicyclomine Hydrochloride USP/Ph.Eur. 10 mg The Antispasmodic Catalyst: Instantly relaxes violently contracting smooth muscle in the gastrointestinal and biliary tracts, and the uterus. Excipients Microcrystalline Cellulose / Maize Starch / Sodium Starch Glycolate / Colloidal Silicon Dioxide / Magnesium Stearate Diluent / Binder / Superdisintegrant / Glidant (Engineered specifically to ensure the bulky Mefenamic Acid powder shatters and dissolves instantly upon reaching the gastric fluid, ensuring the 30-minute rapid onset required for acute pain rescue) *Pack Sizes: 10×10 Blisters (Optimized specifically for high-volume, over-the-counter (OTC) or acute prescription dispensing regimens).
Mefenamic Acid & Paracetamol veterinary injection
FreeComposition
Mefenamic Acid & Paracetamol INJ. 30 ML
Mefenamic Acid & Paracetamol INJ. 100 ML
Usage: – Always consult with a veterinarian before using any medication in animals.
Category: – Anti Analgesic veterinary injection
Therapeutic category: – NSAID, Analgesic, veterinary injection
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Mefenamic Acid Tablets
FreeMefenamic Acid Tablets
Each Uncoated Tablet contains :
Mefenamic Acid IP 500 mgMefenamic Acid Tablets
Each Uncoated Tablet contains :
Mefenamic Acid BP 500 mgUsage: – Relieve mild to moderate pain, including menstrual pain
Category: – Anti Analgesic Painkiller Drugs
Therapeutic category: – NSAID, Analgesic, Antipyretic, Anti Inflammatory
Mefloquine Tablets
FreeMefloquine Tablets
Each Filmcoated tablet contains:
Mefloquine Hydrochloride IP
Eq. To Mefloquine 250 mgMefloqine tablets
Each Filmcoated Tablets Contains:
Mefloquine Hydrochloride IP
Eq To Mefloquine 500 mgMefloquine tablets
Each Filmcoated Tablets Contains:
Mefloquine Hydrochloride BP
eq to Mefloquine 500 mgMefloquine tablets
Each Filmcoated Tablets Contains:
Mefloquine Hydrochloride BP
eq to Mefloquine 750 mgUsage: – Treat malaria
Category: – Antimalarial drugs
Therapeutic category: – Antimalarial
Meloxicam veterinary injection
FreeComposition
Meloxicam VET INJ. 30 ML
Meloxicam VET INJ. 100 ML
Usage: –commonly used in veterinary medicine to alleviate pain and reduce inflammation in animals
Category: – veterinary injection
Therapeutic category: – veterinary medicine
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Meloxicam with Paracetamol veterinary injection
FreeComposition
Meloxicam with Paracetamol VET INJ. 100ML
Usage: – used to manage pain and inflammation in animals
Category: – veterinary injection
Therapeutic category: – veterinary medicine
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Melphalan Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Film-Coated Tablet, packed exclusively in highly secure, handler-protective Amber Glass Bottles or specialized Alu-Alu cold-chain blisters to ensure the absolute chemical survival of the highly unstable alkylating agent.
Active Ingredient Strength Primary Clinical Function Melphalan USP/Ph.Eur. 2 mg Global Oncology Standard: Base therapeutic unit allowing for highly precise, weight-based or Body Surface Area (BSA) calculated “pulsed” dosing regimens. Excipients Microcrystalline Cellulose / Crospovidone / Colloidal Silicon Dioxide / Magnesium Stearate / Premium Opadry Film Diluent / Superdisintegrant / Glidant / Slipper-Coating (Engineered specifically with a heavy film-coat to ensure immediate gastric transit and to completely seal the highly toxic, mutagenic core away from nurses and caregivers) *Pack Sizes: Amber Glass Bottles of 25/50 Tablets (Optimized specifically for strict, highly monitored hematology dispensing regimens).
Memantine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips, frequently customized into 4-week Titration Starter Packs.
Active Ingredient Strength Primary Clinical Function Memantine (as Hydrochloride) USP/Ph.Eur. 5 mg Initiation / Titration Standard: Base therapeutic unit utilized exclusively for the mandatory 4-week step-up protocol to build patient tolerance and prevent severe CNS adverse effects. Memantine (as Hydrochloride) USP/Ph.Eur. 10 mg Global Neurology Standard: High-efficacy adult maintenance dose, typically taken twice daily, for the sustained neuroprotection of the degenerating brain. Excipients Microcrystalline Cellulose / Lactose Monohydrate / Croscarmellose Sodium / Magnesium Stearate / Premium Opadry Film Diluent / Binder / Superdisintegrant / Slipper-Coating (Engineered specifically with a heavy, smooth film-coat to ensure immediate gastric transit and absolute ease of swallowing for elderly patients suffering from severe dysphagia) *Pack Sizes: 10×10 Blisters, Bottles of 60, or Customized 28-Day Titration Starter Kits (Optimized specifically for strict chronic geriatric dispensing regimens).
Meropenem & Sulbactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in glass vials.
Active Ingredient Strength Role Meropenem USP/BP 1000 mg (1 g) Broad-Spectrum Carbapenem Sulbactam Sodium USP/BP Equivalent to 500 mg Beta-Lactamase Inhibitor Total Strength 1.5 g (1500 mg) Per Single Dose Vial *Pack Sizes: Single Vial with or without Sterile Water for Injection.
Meropenem Injection
FreeMeropenem injection (brand name Keppra) is a broad-spectrum carbapenem antibiotic supplied as a sterile white to pale yellow powder for reconstitution. It is used to treat severe bacterial infections
Meropenem injection
FreeComposition
Meropenem 1 g & Sodium 90.2 mg injection
Usage: – used to treat severe intra-abdominal infections caused by susceptible bacteria
Category: – bacterial injection
Therapeutic category: – bacterial infections
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Mesalazine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, pH-Targeted PR Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the highly oxidative 5-ASA molecule.
Active Ingredient Strength Primary Clinical Function Mesalazine (5-ASA) USP/Ph.Eur. 400 mg (PR) Initiation / Titration Standard: Base therapeutic unit for mild presentations or pediatric titration in pediatric gastroenterology. Mesalazine (5-ASA) USP/Ph.Eur. 800 mg / 1200 mg (PR) Global Gastroenterology Standard: High-efficacy adult maintenance doses designed to improve patient compliance by drastically reducing the daily pill burden. Excipients Methacrylic Acid Copolymer Type B & C (Eudragit L/S) / Hypromellose / Triethyl Citrate / Iron Oxide Colors / Talc Enteric & Prolonged-Release Polymers / Plasticizer / Glidant (Engineered specifically to form an impenetrable, acid-resistant shield that strictly delays drug release until reaching the high-pH environment of the lower gastrointestinal tract) Metformin Hydrochloride & Glimepiride Tablets
FreeProduct Composition & Strength
We supply this product in Bilayer Tablets or Matrix Tablets packed in Alu-Alu or PVC/PVDC Blisters.
Product Strength Metformin HCl (SR) Glimepiride Common Name 500/1 mg 500 mg 1 mg G1 Tablet 500/2 mg 500 mg 2 mg G2 Tablet 1000/2 mg 1000 mg 2 mg Forte / DS Tablet 1000/4 mg 1000 mg 4 mg High Strength Metformin Hydrochloride & Pioglitazone Tablets
FreeProduct Composition & Strength
We supply this product as Sustained Release (SR) Matrix Tablets packed in Alu-Alu Blisters.
Product Strength Metformin HCl (SR) Pioglitazone Common Name 500/15 mg 500 mg 15 mg Pio-M 15 500/30 mg 500 mg 30 mg Pio-M 30 1000/15 mg 1000 mg 15 mg Pio-M Forte 1000/30 mg 1000 mg 30 mg Pio-M DS Methoxsalen Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Rapid-Dissolution Tablet or Liquid-Filled Softgel, packed exclusively in highly secure, light-blocking Alu-PVC or Amber Blister strips to ensure the absolute chemical stability of the highly photoactive molecule.
Active Ingredient Strength Primary Clinical Function Methoxsalen (8-Methoxypsoralen) USP/Ph.Eur. 10 mg Global Phototherapy Standard: Weight-based dosing unit designed for exact pharmacokinetic synchronization with scheduled clinical UVA light exposure. Excipients Microcrystalline Cellulose / Pregelatinized Starch / Colloidal Silicon Dioxide / Magnesium Stearate (If Tablet) OR Macrogol (PEG 400) / Gelatin (If Softgel) Fast-Dissolving Carrier / Disintegrant / Glidant (Engineered specifically to guarantee complete gastric breakdown and maximum systemic absorption within 60 to 90 minutes, ensuring the patient is perfectly “primed” for the light booth) *Pack Sizes: Bottles of 30 or 10×10 Blisters (Optimized specifically for strict, monitored bi-weekly dermatological dispensing regimens).
Methyl Prednisolone Tablets
FreeMETHYL PREDNISOLONE TABLETS IP 4 MG Each uncoated tablets contains: Methylprednisolone IP 4 mg Excipients q.s.
METHYL PREDNISOLONE TABLETS IP 8 MG Each uncoated tablets contains:
Methylprednisolone IP 8 mg Excipients q.s.Usage: – Treat certain forms of arthritis
Category: – Anti Allergic Drugs
Therapeutic category: – Steroid
Methyl Ergometrine Maleate veterinary injection
FreeComposition
Methyl Ergometrine Maleate VET INJ. 5ML
Usage: –Here are some key points about Methyl Ergometrine Maleate for veterinary use
Category: – Veterinary Supervision
Therapeutic category: –Veterinary Supervision
Pcd pharma franchise:-
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Methyl Ergonovine Tablets
FreeMethyl Ergonovine maleate tablets IP
Each Sugarcoated tablet contains :
Methyl ergonovine Maleate IP 0.125 mgMethyl Ergonovine Maleate tablets USP
Each Sugarcoated tablet contains :
Methyl Ergonovine Maleate tablets USP 0.125 mgUsage: – Control bleeding from the uterus
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Methylcobalamin + Pyridoxine + Nicotinamide Injection
FreeComposition
PLUS RF (2 ML) Methylcobalamin 1000 mcg
+ Pyridoxine Hydrochloride 100 mg
+ Nicotinamide 100 mg
+ Benzyl Alcohol IP 2% V/V (as preservative)
+ Water for Injection IP q.s.
Usage: –used to treat or prevent vitamin B12 deficiency, which can lead to neurological issues
Category: – Benzyl Alcohol injection
Therapeutic category: – Benzyl Alcohol
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Methylcobalamin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Red Solution in amber glass ampoules.
Active Ingredient Strength Volume Methylcobalamin JP/USP 500 mcg 1 ml Ampoule Methylcobalamin JP/USP 1500 mcg (1.5 mg) 1 ml / 2 ml Ampoule (Standard Adult Dose) Methylcobalamin JP/USP 2500 mcg (2.5 mg) High-Dose (Intensive Therapy) Excipients Mannitol / Benzyl Alcohol Stabilizer / Preservative *Pack Sizes: Tray of 5 Ampoules, 10 Ampoules, or 100 Ampoules.
Methylcobalamin, Folic Acid & Pyridoxine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Photostable Film-Coated Tablet, packed exclusively in highly secure, light-blocking Alu-Alu blister strips to ensure the absolute chemical survival of the three distinct vitamin coenzymes.
Active Ingredient Strength Primary Clinical Function Methylcobalamin (Active B12) Ph.Eur./USP 1500 mcg Myelin Regenerator: Direct peripheral nerve repair and essential co-factor for homocysteine remethylation. Folic Acid (Vitamin B9) Ph.Eur./USP 5 mg Endothelial Shield: Mega-dose methyl-donor driving DNA synthesis, red blood cell formation, and toxic homocysteine clearance. Pyridoxine Hydrochloride (Vitamin B6) Ph.Eur./USP 3 mg Neurotransmitter Catalyst: Vital co-enzyme for the transsulfuration of homocysteine and the synthesis of GABA/Serotonin. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Magnesium Stearate / Titanium Dioxide / Premium Opaque Opadry Film Diluent / Superdisintegrant / Lubricant / Heavy UV-Blocking Coating (Engineered specifically to form an impenetrable light barrier, completely preventing the photolabile degradation of the B12 component) *Pack Sizes: 10×10 Alu-Alu Blisters (Optimized specifically for strict, 3-to-6 month chronic neurology dispensing regimens).
Methylcobalamine Folic Acid Pyridoxine Hydrochloride & DHA Capsules
FreeThis combination is a specialized nutritional supplement, often prescribed for nerve health (neuropathy), pregnancy, or cardiovascular support. It combines essential B-vitamins with a crucial fatty acid.Methylcobalamine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Photostable Film-Coated Tablet, packed exclusively in highly secure, light-blocking Alu-Alu blister strips to ensure the absolute chemical survival of the delicate coenzyme.
Active Ingredient Strength Primary Clinical Function Methylcobalamin (Mecobalamin) Ph.Eur./USP 500 mcg Maintenance / Hematology Standard: Base therapeutic unit for the treatment of Megaloblastic/Pernicious anemia and general nutritional B12 deficiency. Methylcobalamin (Mecobalamin) Ph.Eur./USP 1500 mcg Global Neurology Standard: Mega-dose adult therapy for the aggressive regeneration of peripheral nerves in Diabetic Neuropathy, Sciatica, and Trigeminal Neuralgia. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Magnesium Stearate / Titanium Dioxide / Premium Opaque Opadry Film Diluent / Superdisintegrant / Lubricant / UV-Blocking Coating (Engineered specifically to form a heavy, opaque shield that completely locks out ultraviolet light, preventing the photolabile degradation of the active coenzyme) *Pack Sizes: 10×10 Alu-Alu Blisters (Optimized specifically for strict, 3-to-6 month chronic neurology dispensing regimens).
METHYLERGOMETRINE INJECTION
FreeCOMPOSITION
Each ml. contains:Methylergometrine Maleate I.P. 0.2 mg.
Water for Injec9on I.P. Q.S 1 mlUsage: – Control bleeding from the uterus
Category: – Generic Drugs and injection
Therapeutic category: – Generic Drugs
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Methylprednisolone Acetate Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Milky White Suspension in glass vials.
Active Ingredient Strength Standard Use Methylprednisolone Acetate 40 mg / ml Standard IM / Small Joints Methylprednisolone Acetate 80 mg / ml High Dose IM / Large Joints (Knee) Excipients Polyethylene Glycol (PEG) / Myristyl-gamma-picolinium chloride Suspending Agent / Preservative *Pack Sizes: 1 ml Vial, 2 ml Vial, or 5 ml Multi-dose Vial.
Methylprednisolone Sodium Succinate Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder requiring reconstitution with the provided solvent.
Active Ingredient Strength Standard Use Methylprednisolone Sod. Succ. 40 mg Local Injection / Minor Allergic Reaction Methylprednisolone Sod. Succ. 125 mg Acute Asthma / Status Asthmaticus Methylprednisolone Sod. Succ. 500 mg / 1000 mg (1 gm) Pulse Therapy (Lupus / Transplant Rejection) Diluent Bacteriostatic Water Provided with Act-O-Vial or Separate Ampoule *Pack Sizes: Single Vial with Diluent.
Metoclopramide Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Isotonic Solution in glass ampoules.
Active Ingredient Strength Volume Metoclopramide HCl USP/BP 5 mg / ml 2 ml Ampoule (Total 10 mg) Metoclopramide HCl USP/BP 5 mg / ml 10 ml Multi-dose Vial Preservative Sodium Metabisulfite Antioxidant *Pack Sizes: Tray of 10 Ampoules, 50 Ampoules, or 100 Ampoules.
Metoclopramide injecton
FreeComposition
Each ml Contains : Metoclopramide Mono Hydrochloride I.P.
Equi. To Anhydrous Metoclopramide
Hcl…………………………………….. 5mg
Methyl Paraben I.P 0.15 % W/V ( As Preserva9ve )
Propyl Paraben I.P 0.02 % W/V ( As Preserva9ve )
Water for Injec9on I.P. Q.S 10 ml , 30 mlUsage: – Anti-sickness medicine
Category: – Antacid & Anti ulcer injection
Therapeutic category: – Antiprotozoal,Anti Vomiting
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Metoclopramide Tablets
FreeZerinorm
Metoclopramide tablets BP 10mg
Each Uncoated tablet contains :
Metoclopramide hydrocloride BP
Eq To Anhydrous Metoclopramide Hydrocloride 10 mgUsage: – Anti-sickness medicine
Category: – Antacid & Anti ulcer drugs
Therapeutic category: – Antiprotozoal,Anti Vomiting
Metoprolol & Amlodipin Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered, API-Segregated Bilayer Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu or high-density PVC/PVDC blister strips to ensure the absolute chemical stability of the combined molecules.
Active Ingredient Strength Primary Clinical Function Metoprolol (as Tartrate/Succinate ER) Ph.Eur./USP 25 mg / 50 mg Cardioselective Base: Reduces heart rate, cardiac workload, and neutralizes CCB-induced reflex tachycardia. Amlodipine (as Besylate) Ph.Eur./USP 5 mg Calcium Channel Blocker (CCB): Forces profound peripheral arterial vasodilation, dropping severe blood pressure and relieving coronary spasms. Excipients Microcrystalline Cellulose / Dibasic Calcium Phosphate / Crospovidone / Magnesium Stearate / Premium Opadry Film / Iron Oxide Colors Diluent / Stabilizing Buffer / Superdisintegrant / Lubricant (Engineered specifically using bilayer press technology to physically separate the slightly acidic Metoprolol salt from the basic Amlodipine, preventing destructive chemical cross-talk) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict 30-day chronic cardiology dispensing regimens).
Metoprolol & Ramipril Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered, API-Segregated Bilayer Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the highly degradable Ramipril molecule.
Active Ingredient Strength Primary Clinical Function Metoprolol (as Succinate/Tartrate) Ph.Eur./USP 25 mg / 50 mg Cardioselective Base: Reduces heart rate, cardiac output, and myocardial oxygen demand; protects against sudden cardiac death. Ramipril Ph.Eur./USP 2.5 mg / 5 mg ACE Inhibitor Driver: Forces profound systemic vasodilation, protects the kidneys (renoprotection), and halts pathological cardiac remodeling. Excipients Microcrystalline Cellulose / Sodium Stearyl Fumarate / Hypromellose / Sodium Bicarbonate / Iron Oxide Colors Low-Moisture Diluent / Lubricant / Film-Coating / Polymeric Stabilizer (Engineered specifically to physically isolate the acidic Ramipril from cross-reacting with the Metoprolol salt, preventing the formation of toxic diketopiperazine impurities) *Pack Sizes: 10×10 Alu-Alu Blisters (Optimized specifically for strict 30-day chronic cardiology dispensing regimens).
Metoprolol Tablets
FreeMETOPROLOL TARTRATE TABLET IP Each Tablet Contains: Metoprolol tartrate IP 50 MG
XYPROL – T TABLETS
Metoprolol Tartrate Tablet IP
Each Tablet Contains:
Metoprolol Tartrate IP 50 mgUsage: – Treat high blood pressure
Category: – Anti Cardiovascular drugs
Therapeutic category: – Cardiovascular Agent, Anti hypertensive
Metronidazole Capsule 500mg
FreeMetronidazole 500 mg capsules are an antibiotic and antiprotozoal medication used to treat a variety of bacterial and parasitic infections. The capsule contains a specific amount of the active drug and various inactive ingredients that help with its delivery and stability.
Metronidazole injection
FreeComposition
Each 100 ml contains
Metronidazole 100 mg +Sodium Chloride 900 mg
Metronidazole injection is 500 mg to 1 gram
Pcd pharma franchise:-
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Metronidazole Tablets
FreeMetronidazole Tablets IP 200 mg
Each Uncoated tablet contains :
Metronidazole IP 200 mgMetronidazole Tablets IP 400 mg
Each Uncoated tablet contains :
Metronidazole IP 400 mgMetronidazole Tablets IP 200 mg
Each Filmcoated Tablets contains :
Metronidazole IP 200 mg
Excipients q.sMetronidazole Tablets IP 400 mg
Each film coated tablet contains :
Metronidazole IP 400 mg Excipients Q.S.Metro V – 250
Metronidazole Vaginal Tablets
Each Uncoated tablets
Metronidazole BP 250 mgMetronidazole tablets BP
Each Sugarcoated tablet contains :
Metronidazole BP 250 mgMetronidazole Tablets USP
Each Uncoated tablet contains :
Metronidazole USP 200 mgMetronidazole Tablets USP
Each Uncoated tablet contains :
Metronidazole USP 400 mgMetronidazole Tablets USP
Each Uncoated tablet contains :
Metronidazole USP 500 mgMetronidazole tab BP 250 mg Metronidazole BP 250 mg Titanium dioxide bp Excipent
Cirogyl – 400
Metronidazole Tablets BP 400 mg
Each Film coated Tablets contains :
Metronidazole BP 400 mgMetronidazole Tab BP 500 mg Metronidazole BP 500 mg Brilliant blue Excipents
Usage: – Skin infections, rosacea and mouth infections
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic
Metronidazole Vaginal Tablets
FreeProduct Composition & Strength
We supply this product as compressed Vaginal Tablets (Pessaries) in blister packs.
Active Ingredient Strength (Standard) Form Role Metronidazole IP/BP/USP 500 mg Uncoated / Film Coated Tablet Antiprotozoal / Antibacterial Lactic Acid Q.S. (Optional Additive) pH Regulator Excipients Q.S. Disintegrants / Binders Base for Tablet Midazolam Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Rapid-Burst Immediate-Release Tablet, packed exclusively in highly secure, tamper-evident Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the highly soluble maleate salt.
Active Ingredient Strength Primary Clinical Function Midazolam (as Maleate) Ph.Eur./USP 7.5 mg Initiation / Geriatric Standard: Base therapeutic unit for pre-surgical anxiety reduction and sleep induction in elderly or debilitated patients. Midazolam (as Maleate) Ph.Eur./USP 15 mg Global Anesthesiology Standard: High-efficacy adult dose for profound pre-procedural conscious sedation and the treatment of severe, treatment-refractory insomnia. Excipients Microcrystalline Cellulose / Lactose Anhydrous / Croscarmellose Sodium / Magnesium Stearate / Premium Opadry Film Diluent / Fast-Dissolving Carrier / Superdisintegrant / Lubricant (Engineered specifically to shatter upon contact with gastric fluid, ensuring peak plasma concentration is reached within 20 to 30 minutes) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict, highly monitored hospital and short-term dispensing regimens).
Midodrine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Rapid-Burst Immediate-Release Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the highly soluble hydrochloride salt.
Active Ingredient Strength Primary Clinical Function Midodrine (as Hydrochloride) USP/Ph.Eur. 2.5 mg / 5 mg Initiation / Titration Standard: Base therapeutic units for careful dose-titration to elevate standing blood pressure without triggering dangerous supine hypertension. Midodrine (as Hydrochloride) USP/Ph.Eur. 10 mg Global Dysautonomia Standard: High-efficacy adult maintenance dose for severe neurogenic orthostatic hypotension. Excipients Microcrystalline Cellulose / Pregelatinized Starch / Colloidal Silicon Dioxide / Magnesium Stearate Diluent / Disintegrant / Glidant / Lubricant (Engineered specifically without heavy polymeric retardants to guarantee rapid, complete gastric dissolution, ensuring the prodrug reaches the liver instantly for conversion) *Pack Sizes: 10×10 Blisters or Bottles of 100 (Optimized specifically for strict, daytime-only chronic dispensing regimens).
Minocycline Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Hydrophilic Matrix ER Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips or opaque HDPE bottles to ensure the absolute chemical stability of the photosensitive hydrochloride salt.
Active Ingredient Strength Primary Clinical Function Minocycline (as Hydrochloride) USP/Ph.Eur. 50 mg (ER) Dermatology Maintenance Standard: Long-term, low-dose therapy for controlling inflammatory acne and severe Rosacea with minimal side effects. Minocycline (as Hydrochloride) USP/Ph.Eur. 100 mg (ER) Acute Clearance Standard: High-dose therapy for severe cystic acne breakouts, CA-MRSA skin infections, and Rheumatoid Arthritis flares. Excipients Hypromellose (HPMC) / Microcrystalline Cellulose / Magnesium Stearate / Premium Opaque Opadry Film Hydrophilic ER Polymer / Diluent / Lubricant / UV-Blocking Coating (Engineered specifically to pace the gastric release over several hours, preventing the rapid blood-brain-barrier saturation that triggers vertigo) *Pack Sizes: 10×10 Alu-Alu Blisters or Bottles of 30 (Optimized specifically for strict 12-to-16 week chronic dermatology dispensing regimens).
Mirabegron Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Hydrophilic Matrix ER Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute physical stability of the extended-release polymers.
Active Ingredient Strength Primary Clinical Function Mirabegron Ph.Eur./USP 25 mg (ER) Initiation / Renal Standard: Base therapeutic unit for assessing patient tolerance and for use in patients with severe renal or hepatic impairment. Mirabegron Ph.Eur./USP 50 mg (ER) Global Urology Standard: High-efficacy adult maintenance therapy for maximum suppression of urge urinary incontinence and detrusor overactivity. Excipients Polyethylene Oxide / Hypromellose (HPMC) / Butylated Hydroxytoluene (BHT) / Magnesium Stearate / Premium Opadry Film Swellable ER Polymer / Hydrophilic Matrix Base / Antioxidant / Lubricant (Engineered specifically to form an impenetrable, slow-eroding gel matrix in the GI tract, strictly metering the release of the active ingredient over 24 hours) *Pack Sizes: 10×10 Alu-Alu Blisters or Bottles of 30 (Optimized specifically for strict 30-day chronic urology dispensing regimens).
Misoprostol Tablets
FreeMisoprostol Tablets 100 mcg
Each Uncoated Tablet Contains:
Misoprostol 100 mcgMISOPROSTOL TABLETS IP 200 MG
Each uncoated Tablet Contains:
misoprostol IP 200 mcg Excipients q.s.Misoprostol Tablets 200 mcg
Each Uncoated tablet contains :
Misoprostol 200 mcgMisoprostol Tablets 600 mcg
Each Uncoated Tablet Contains:
Misoprostol 600 mcg
Excipients q.sMisoprostol Tablets 200 mcg
Each Uncoated tablet contains :
Misoprostol 200 mcgModafinil Tablets
FreeMODAFINIL TABLETS USP 100 MG EACH UNCOATED TABLET CONTAINS: MODAFINIL BP 100 MG: – MODAFINIL BP 100 mg
EXCIPIENTS QSUsage: – Treat sleepiness caused by narcolepsy, shift work sleep disorder, or obstructive sleep apnea
Category: – Generic Drugs and Medicine
Therapeutic category: – Nootropic, CNS Drugs, Generic Drugs
Morphine Sulphate Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Extended-Release Matrix Tablet, packed exclusively in highly secure, tamper-evident Alu-PVC or Alu-Alu blister strips to ensure the absolute physical stability of the controlled-release polymers.
Active Ingredient Strength Primary Clinical Function Morphine Sulphate USP/Ph.Eur. 15 mg (SR) Initiation / Titration Standard: Base therapeutic unit for opioid-naïve patients starting around-the-clock chronic pain management. Morphine Sulphate USP/Ph.Eur. 30 mg / 60 mg (SR) Global Oncology Standard: High-dose adult maintenance therapy for severe, opioid-tolerant terminal cancer and intractable neuropathic pain. Excipients Hypromellose (HPMC) / Cetostearyl Alcohol / Lactose Monohydrate / Magnesium Stearate / Premium Color-Coded Film Hydrophilic Polymer / Lipophilic Retardant / Diluent / Lubricant (Engineered specifically to form an impenetrable, slow-eroding gel matrix in the GI tract, strictly metering the release of the narcotic over 12 hours) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict 30-day chronic oncology dispensing regimens; frequently color-coded by strength to prevent fatal dispensing errors).
Moxifloxacin & Cefixime Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the highly reactive antibiotic compounds.
Active Ingredient Strength Primary Clinical Function Moxifloxacin (as Hydrochloride) Ph.Eur./USP 400 mg 4th-Gen Fluoroquinolone: Deep tissue and lung penetration to eradicate DNA replication in resistant respiratory and intra-abdominal pathogens. Cefixime (as Trihydrate) Ph.Eur./USP 400 mg 3rd-Gen Cephalosporin: Massive Gram-negative cell wall destruction, specifically targeting enteric pathogens like Salmonella typhi. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Povidone K30 / Magnesium Stearate / Premium Opadry Film Diluent / Superdisintegrant / Binder / Moisture-Barrier Coating (Engineered specifically to protect the hygroscopic Cefixime Trihydrate and mask the intensely bitter taste of Moxifloxacin) *Pack Sizes: 10×10 Alu-Alu Blisters (Optimized specifically for strict 7-to-14 day acute critical-care anti-infective dispensing regimens).
Moxifloxacin Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the highly reactive hydrochloride salt.
Active Ingredient Strength Primary Clinical Function Moxifloxacin (as Hydrochloride) Ph.Eur./USP 400 mg Global Pulmonology Standard: High-dose adult therapy for the aggressive eradication of resistant respiratory, skin, and intra-abdominal pathogens. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Lactose Monohydrate / Magnesium Stearate / Premium Opadry Film Diluent / Superdisintegrant / Binder / Moisture-Barrier Coating (Engineered specifically to protect the highly hygroscopic API and completely mask its intensely bitter taste) *Pack Sizes: 1×5 or 10×10 Alu-Alu Blisters (Optimized specifically for strict 5-to-14 day acute critical-care dispensing regimens).
Multivitamin & Multimineral Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated or Sugar Coated Tablets (typically Red, Maroon, or Brown). The coating is an absolute necessity to prevent the oxidative degradation of vitamins by the mineral salts.
Category Ingredient (BP Grade) Strength (Standard) Fat-Soluble Vitamins Vitamin A (Retinol Acetate/Palmitate) BP 2500 IU / 5000 IU Vitamin D3 (Cholecalciferol) BP 200 IU / 400 IU Vitamin E (Tocopheryl Acetate) BP 10 mg / 15 IU Water-Soluble Vitamins Vitamin B1 (Thiamine Mononitrate) BP 2 mg / 10 mg Vitamin B2 (Riboflavin) BP 2 mg / 10 mg Vitamin B3 (Nicotinamide) BP 25 mg / 50 mg Vitamin B6 (Pyridoxine HCl) BP 1 mg / 3 mg Vitamin B12 (Cyanocobalamin) BP 1 mcg / 15 mcg Folic Acid BP 1 mg Vitamin C (Ascorbic Acid) BP 50 mg / 100 mg Minerals & Trace Elements Ferrous Fumarate / Sulphate BP 30 mg / 50 mg (Elemental Iron) Zinc Sulphate BP 15 mg (Elemental Zinc) Copper Sulphate BP 0.5 mg / 2 mg Selenium (as Sodium Selenite) BP 60 mcg / 70 mcg Iodine (as Potassium Iodide) BP 100 mcg / 150 mcg Multivitamin & Multimineral Tablets (Complete A-Z Formula)
FreeProduct Composition & Strength
We supply this product as Sugar Coated or Film Coated Tablets (typically Red, Maroon, or Brown). The coating is an absolute necessity to prevent the oxidative degradation of vitamins by the mineral salts.
Category Ingredient Strength (Standard) Fat-Soluble Vitamins Vitamin A (Acetate/Palmitate) 2500 IU / 5000 IU Vitamin D3 (Cholecalciferol) 200 IU / 400 IU Vitamin E (Acetate) 10 mg / 15 mg Water-Soluble Vitamins Vitamin B1 (Thiamine Mononitrate) 2 mg / 10 mg Vitamin B2 (Riboflavin) 2 mg / 10 mg Vitamin B6 (Pyridoxine HCl) 1 mg / 3 mg Vitamin B12 (Cyanocobalamin) 1 mcg / 15 mcg Vitamin C (Ascorbic Acid) 50 mg / 100 mg Nicotinamide (Vit B3) 25 mg / 50 mg Folic Acid (Vit B9) 1000 mcg (1 mg) Minerals & Trace Elements Ferrous Sulphate (Iron) 30 mg / 50 mg (Elemental) Zinc Sulphate Monohydrate 15 mg / 22.5 mg (Elemental) Copper Sulphate 0.5 mg / 2 mg Selenium Dioxide 60 mcg / 70 mcg Potassium Iodide 100 mcg / 150 mcg Multivitamin 4G Softgels
Free4G multivitamin softgels are specialized health supplements that get their “4G” name from four key herbal extracts—Ginseng, Green Tea, Ginkgo Biloba, and Grape Seed—combined with Omega-3 fatty acids and a comprehensive range of vitamins and minerals. These softgels are designed to support cardiovascular health, cognitive function, and immunity.
Multivitamin Injection
FreeProduct Composition & Strength
We supply this product as a Dual-Ampoule Set or a Single-Vial Emulsion (depending on market regulation).
Vitamin Group Active Ingredients Standard Adult Dose (per 10 ml) Fat-Soluble Retinol (A) / Ergocalciferol (D) / Tocopherol (E) 3300 IU / 200 IU / 10 IU Water-Soluble (C) Ascorbic Acid (C) 100 mg – 200 mg Water-Soluble (B) Thiamine (B1) / Riboflavin (B2) / Pyridoxine (B6) 3 mg / 3.6 mg / 4 mg Others Niacinamide / Pantothenic Acid / Folic Acid / B12 / Biotin 40 mg / 15 mg / 400 mcg / 5 mcg / 60 mcg *Pack Sizes: Tray of 5 Dual-Ampoules or Box of 10 Vials.
Multivitamin Tablets
FreeProduct Composition & Strength
We supply this product as Sugar Coated Tablets (typically Red, Yellow, or Brown). The sugar coating is mandatory to mask the strong odor of B-vitamins and protect the labile Vitamin A and C from oxidation.
Active Ingredient Strength (Standard IP/NFI) Therapeutic Role Vitamin A (Acetate) IP 2500 IU Vision / Immunity Vitamin B1 (Thiamine Mononitrate) IP 2 mg Carbohydrate Metabolism Vitamin B2 (Riboflavin) IP 2 mg Mucosal Health Vitamin B6 (Pyridoxine HCl) IP 1 mg Protein Synthesis Niacinamide IP 25 mg Cellular Energy Calcium Pantothenate IP 5 mg Metabolic Support Vitamin C (Ascorbic Acid) IP 50 mg Antioxidant / Collagen Vitamin D3 (Cholecalciferol) IP 200 IU Bone Mineralization Folic Acid IP 1 mg DNA Synthesis / Anemia Excipients Q.S. Gelatin / Sucrose / Talc Binder / Coating Multivitamin Tablets (Vitamin A, D3 & B-Complex)
FreeProduct Composition & Strength
We supply this product as Sugar Coated Tablets (typically Red, Yellow, or Brown). The sugar coating is critical to mask the strong odor of B-vitamins and protect Vitamin A from oxidation.
Active Ingredient Strength (Standard Prophylactic) Therapeutic Role Vitamin A (Acetate/Palmitate) 2500 IU / 5000 IU Vision / Immunity Vitamin D3 (Cholecalciferol) 200 IU / 400 IU Bone Mineralization Thiamine Mononitrate (Vit B1) 2 mg / 10 mg Nerve Function Riboflavin (Vit B2) 2 mg / 10 mg Cellular Energy Pyridoxine HCl (Vit B6) 1 mg / 3 mg Protein Metabolism Niacinamide 20 mg / 50 mg Skin Health / Energy Calcium Pantothenate 5 mg / 50 mg Metabolic Support Folic Acid 1 mg / 1.5 mg DNA Synthesis Excipients Q.S. Gelatin / Sucrose / Talc Binder / Coating Multivitamin Vitamin B Complex Tablets
FreeB Complex Tablet With B-12
Each film Coated Tablet Contains
Vitamin B1 IP ( 1 mg )
Vitamin B2 IP ( 0.5 mg )
Vitamin B6 IP ( 0.5mg )
Calcium Pantothenate IP (1 mg)
Niacinamide IP ( 10 mg )
Vitamin B12 IP (0.5 mcg)
Sunset Yellow (-) Excipients (–)Obiplex (For ProphylacticUse)
(Vitamin B Complex Tablets)
Each Sugar coated tablet contains:
Viatmin B1 Mononitrate IP 0.5 mg
Viatmin B2 IP 0.5 mg
Niacinamide B3 IP 7.5 mg
Dosage : Two Tablet daily or As directed by the
PhysicianObiplex Forte tablets
(Vitamin B Complex tablet
Each Uncoated tablet contains :
Thiamine mononitrate IP 3 mg
Riboflavin IP 1.0 mg
Pyridoxine Hydrochloride IP 0.5 mg
Calcium Pantothenate IP 3 mg
Niacinamide IP 15 mg
Shelf life: One yeaVita + Iron Tablets (Multivitamin Tablets )
Each Filmcoated tablet contains :
Vitamin A BP 800 IU
Vitamin D BP 200 IU
Vitamin E BP 10 mg
Vitamin B1 BP 1.4 mg
Viatmin B2 BP 1.4 mg
Vitamin B6 BP 1.9 mg
Vitamin B12 BP 2.6 mcg
Vitamin C BP 70.0 mg
Nicotinamide BP 18.0 mg
Folic Acid BP 400.0 mcg
Copper Sulphate Eq. To Elemental Copper 2.0 mg
Ferrous sulphate eq To Elemental Iron 30.0 mg
Selinium dioxide Eq. To elemental selinium 65.0 mcg
Zinc Sulphate eq To elemental Zinc 15.0 mg
Potassium Iodide eq.to elemental Iodine 150.0 mcgHydntamins
(Prophylactic Use )
(Multivitamin Tablets )
Each Sugar Coated tablet contains :
Vitamin A BP 800 IU
Vitamin D3 BP 100 IU
Vitamin B1 BP 0.5 mg
Vitamin B2 BP 0.5 mg
Niacinamide BP 7.5 mg
Shelf Life : 2 years
Dosage : As directed by the PhysicianMULTIVITAMIN TABLETS (FOR PROPHYLACTIC USE )
Each Sugarcoated tablet Contains: VITAMIN A IP (500 IU)
– Vitamib D3 IP (50 IU)
– Vitamin B1 IP (0.5 mg)
– Vitamin B2 IP (0.5 mg)
– Niacinamide IP (7.5 mg)
– Colour : Erythrosine Supra / Carmosine Supra (- )MULTIVITAMIN TABLETS (FOR PROPHYLACTIC USE)
Each Filmcoated tablet contains:
Vitamin A IP (800 IU)
– Cholecalciferol IP (100 IU)
– Vitamin B1 IP (1.0 mg)
– Vitamin B2 IP (0.5 mg)
– Vitamin B6 IP (0.5 mg)
– Calcium Pantothenate IP (1.0 mg)
– Niacinamide IP (10 mg)
– Folic Acid IP (0.1 mg)
– Colour : Brilliant Blue / Erythrosine (- )MULTIVITAMIN TABLETS (FOR PROPHYLACTIC
USE) Each Filcoated Tablet contains: – Vitamin A IP
(800 IU)
– Cholecalciferol IP (100 IU)
– Thiamine Mononitrate (vit. B1) IP (1.0 mg)
– Riboflavin (Vit B2) IP (0.5 mg)
– Pyridoxine Hydrochloride (Vit.B6) IP (0.5 mg)
-Calcium Pantothenate IP (1.0 mg)
– Niacinamide IP (10 mg)
– Folic acid IP (0.1 mg)
– Ponceau 4 R (- )
– Excipients (- QS)Multivitamin Tablets NFI
(For Prophylactic Use)
Each Sugarcoated tablet contains :
Vitamin A IP 2500 IU
Vitamin B1 IP 2 mg
Viatmin B2 IP 2 mgMultivitamin Tablets NFI
(For Prophylactic Use)
Each Sugarcoated tablet contains :
Vitamin A IP 2500 IU
Vitamin B1 IP 2 mgMultivitamin Tablets NFI (For Prophylactic Use)
Each Sugarcoated tablet contains:
Vitamin A IP (2500 IU)
– Vitamin B1 IP (2 mg)
– Viatmin B2 IP (2 mg)
– Viatmin B6 IP (0.5 mg)
– Niacinamide IP (25 mg)
– Calcium Pantothenate IP (1 mg)
– Vitamin C IP (50 mg)
– Vitamin D3 IP (200 IU)
– Folic Acid IP (0.2 mg)
– Colour : Red Oxide of Iron (- )Multivitamins & Multiminerals Capsules
FreeMultivitamin and multimineral capsules contain a broad spectrum of active nutrients designed to fill dietary gaps, along with inactive excipients that provide structure and stability to the capsule. While specific formulations vary by brand and target audience (such as men, women, or seniors), most contain a combination of 13 essential vitamins and several key minerals.
Mycophenolate Mofetil Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant and handler-protective Alu-Alu blister strips.
Active Ingredient Strength Primary Clinical Function Mycophenolate Mofetil USP/Ph.Eur. 250 mg Dose-Titration / Pediatric Standard: Base therapeutic unit for precise plasma-level titration and pediatric transplant management. Mycophenolate Mofetil USP/Ph.Eur. 500 mg Global Transplant Standard: High-dose adult maintenance therapy for the aggressive prevention of solid organ rejection. Excipients Microcrystalline Cellulose / Povidone K-90 / Croscarmellose Sodium / Magnesium Stearate / Premium Opadry Film Diluent / Binder / Superdisintegrant / Heavy Film-Coating (Engineered specifically to create a dense, impenetrable barrier over the cytotoxic API core, ensuring absolute handling safety for nurses and pharmacists) *Pack Sizes: 10×10 Alu-Alu Blisters (Optimized specifically for strict, chronic post-transplant dispensing regimens).
N-Acetyl Cysteine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Effervescent Tablet or Odor-Masked Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant heavy-gauge Alu-Alu or Strip packs to ensure the absolute chemical stability of the highly hygroscopic API.
Active Ingredient Strength Primary Clinical Function N-Acetyl Cysteine (NAC) USP/Ph.Eur. 600 mg Global Pulmonology Standard: High-dose adult therapy for the rapid liquefaction of thick respiratory mucus and systemic antioxidant replenishment. Excipients (Effervescent Matrix) Citric Acid Anhydrous / Sodium Bicarbonate / Sucralose / Premium Orange-Lemon Flavoring / Macrogol 6000 Acid-Base Effervescent Couple / Sweetener / Odor-Masking Flavor / Lubricant (Engineered specifically to trigger a violent, complete dissolution in water within 60 seconds, creating a clear, pleasant-tasting respiratory therapeutic drink) *Pack Sizes: 10-Tablet Tubes, 10×10 Strip Packs, or Alu-Alu Blisters (Optimized specifically to absolutely lock out ambient moisture).
Nalidixic Acid Tablets
FreeNalidixic Acid Tablets IP
Each Tablet contains :
Nalidixic Acid IP 500 mgUsage: – Treatment of bacterial infections
Category: – Antibiotic Drugs
Therapeutic category: – Antibiotic
Naloxone Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Glass Ampoules or Pre-filled Syringes.
Active Ingredient Strength Format Naloxone Hydrochloride USP/BP 400 mcg (0.4 mg) 1 ml Ampoule (Standard Adult) Naloxone Hydrochloride USP/BP 20 mcg (0.02 mg) 1 ml Ampoule (Neonatal/Pediatric) Excipients Methylparaben / Propylparaben Preservative (in Multi-dose Vials) *Pack Sizes: Tray of 10 Ampoules or Box of 10 Vials.
Naltrexone Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Taste-Masked Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the hydrochloride salt.
Active Ingredient Strength Primary Clinical Function Naltrexone Hydrochloride USP/Ph.Eur. 50 mg Global Addiction Standard: Single daily maintenance dose to ensure continuous, 24-hour blockade of opioid receptors and suppression of alcohol cravings. Excipients Microcrystalline Cellulose / Lactose Monohydrate / Crospovidone / Magnesium Stearate / Colloidal Silicon Dioxide / Premium Opadry Film Diluent / Superdisintegrant / Glidant / Heavy Film-Coating (Engineered specifically to completely mask the violently bitter taste of the API, preventing the patient from spitting the pill out and ensuring total compliance) *Pack Sizes: 10×10 Blisters or Bottles of 30/100 (Optimized specifically for strictly monitored, 30-day outpatient rehabilitation dispensing regimens).
Naproxen Sodium Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Rapid-Burst Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu or high-density PVC/PVDC blister strips to ensure the absolute chemical stability of the hygroscopic sodium salt.
Active Ingredient Strength Primary Clinical Function Naproxen Sodium USP/Ph.Eur. 275 mg
(Equivalent to 250 mg Naproxen base)Acute Mild / OTC Standard: Base therapeutic unit for rapid relief of tension headaches, mild trauma, and OTC fever reduction. Naproxen Sodium USP/Ph.Eur. 550 mg
(Equivalent to 500 mg Naproxen base)Acute Severe Rx Standard: High-dose “loading” therapy to instantly abort severe acute migraines, acute gout flares, and post-surgical pain. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Povidone K30 / Talc / Magnesium Stearate / Premium Opadry Film Diluent / Superdisintegrant / Binder / Moisture-Barrier Coating (Engineered specifically to protect the hygroscopic salt from humidity in the blister, but trigger a violent, immediate structural collapse the exact second it hits the stomach) *Pack Sizes: 10×10 Blisters or Bottles of 100 (Optimized specifically for high-turnover acute dispensing and retail pharmacy OTC placement).
Naproxen Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Enteric-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the active ingredient and the protective polymer matrix.
Active Ingredient Strength Primary Clinical Function Naproxen USP/Ph.Eur. 250 mg (EC) Initiation / Mild Standard: Base therapeutic unit for mild musculoskeletal trauma, acute gout, and primary dysmenorrhea. Naproxen USP/Ph.Eur. 500 mg (EC) Global Rheumatology Standard: High-dose adult maintenance therapy for severe Rheumatoid Arthritis, Osteoarthritis, and Ankylosing Spondylitis. Excipients Methacrylic Acid Copolymer / Microcrystalline Cellulose / Croscarmellose Sodium / Povidone K30 / Triethyl Citrate / Magnesium Stearate pH-Dependent Enteric Barrier / Diluent / Superdisintegrant / Plasticizer (Engineered specifically to absolutely block dissolution in the highly acidic stomach, preventing mucosal burns, and rapidly dissolving only in the alkaline duodenum) *Pack Sizes: 10×10 Blisters or Bottles of 100/500 (Optimized specifically for strict chronic orthopedic dispensing regimens and massive government health tenders).
Nebivolol Hydrochlorothiazide Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Dual-Active Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu or high-density PVC/PVDC blister strips to ensure the absolute chemical stability of the active ingredients.
Active Ingredient Strength Primary Clinical Function Nebivolol (as HCl) Ph.Eur./USP 5 mg Cardioselective & Vasodilatory Base: Reduces cardiac workload and provides active Nitric Oxide-driven peripheral vasodilation. Hydrochlorothiazide (HCTZ) Ph.Eur./USP 12.5 mg / 25 mg Thiazide Diuretic Driver: Forces the excretion of excess sodium and water, massively amplifying the blood-pressure-lowering effect of the beta-blocker. Excipients Lactose Monohydrate / Microcrystalline Cellulose / Croscarmellose Sodium / Polysorbate 80 / Magnesium Stearate / Premium Opadry Film Diluent / Superdisintegrant / Wetting Surfactant / Moisture-Barrier Coating (Engineered specifically to protect the highly delicate racemic Nebivolol balance while ensuring rapid, synchronized gastric dissolution of the diuretic) *Pack Sizes: 10×10 Blisters or 28/30-Tablet Calendar Packs (Optimized specifically for strict 30-day chronic cardiology dispensing regimens).
Neomycin Sulphate veterinary injection
FreeComposition
Neomycin Sulphate INJ. 30 Ml
Usage: – use of neomycin in veterinary medicine is similar to its use in humans
Category: – veterinary injection
Therapeutic category: – veterinary medicine
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Neostigmine Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Isotonic Solution in glass ampoules or vials.
Active Ingredient Strength Format Neostigmine Methylsulfate USP/BP 0.5 mg / ml 1 ml / 10 ml Vial (Standard) Neostigmine Methylsulfate USP/BP 2.5 mg / ml Concentrate (Rarely used) Excipients Phenol / Methylparaben Preservative (in Multi-dose Vials) *Pack Sizes: Tray of 10 Ampoules or Box of 1/10 Vials.
Neostigmine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Uncoated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu or high-density PVC/PVDC blister strips to ensure the absolute chemical stability of the highly hygroscopic bromide salt.
Active Ingredient Strength Primary Clinical Function Neostigmine Bromide Ph.Eur./USP 15 mg Global Clinical Standard: Base therapeutic oral unit for the continuous, meticulously spaced symptomatic management of Myasthenia Gravis. Excipients Lactose Monohydrate / Maize Starch / Povidone K30 / Talc / Magnesium Stearate Diluent / Binder / Glidant / Lubricant (Engineered specifically for rapid, predictable gastric dissolution to ensure precise symptom control within 30 to 45 minutes of ingestion) *Pack Sizes: 10×10 Blisters or Bottles of 100 (Optimized specifically for strict, high-volume chronic neurology dispensing, as patients may require up to 10-15 tablets per day in divided doses).
Nicorandil Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Moisture-Shielded Tablet, packed exclusively in highly secure, heavy-gauge Alu-Alu blister strips to ensure the absolute chemical stability of the highly volatile active ingredient.
Active Ingredient Strength Primary Clinical Function Nicorandil Ph.Eur./BP 5 mg Initiation / Titration Standard: Base therapeutic unit for the mandatory slow-titration protocol required to build patient tolerance and prevent severe “nitrate headaches.” Nicorandil Ph.Eur./BP 10 mg / 20 mg Global Cardiology Standard: High-dose adult maintenance therapy for severe, refractory stable angina pectoris. Excipients Maize Starch / Croscarmellose Sodium / Stearic Acid / Mannitol Low-Moisture Diluent / Superdisintegrant / Lubricant (Engineered specifically without hygroscopic binders to guarantee the tablet remains perfectly dry and structurally intact throughout its shelf life) *Pack Sizes: 10×10 Alu-Alu Blisters (Optimized specifically for strict chronic cardiology dispensing regimens).
Nicotinamide Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Immediate-Release Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or high-density HDPE bottles to ensure the absolute chemical stability of the highly soluble vitamin amide.
Active Ingredient Strength Primary Clinical Function Nicotinamide (Niacinamide) USP/Ph.Eur. 250 mg Dermatology / Acne Standard: Base therapeutic unit for the systemic reduction of inflammatory skin lesions and mild nutritional supplementation. Nicotinamide (Niacinamide) USP/Ph.Eur. 500 mg Oncology Prophylaxis / Pellagra Standard: High-dose adult therapy for non-melanoma skin cancer prevention and acute pellagra eradication. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Stearic Acid / Magnesium Stearate / Premium Opadry Film Diluent / Superdisintegrant / Lubricant (Engineered specifically for rapid, complete gastric dissolution to ensure peak systemic NAD+ saturation within 1 to 2 hours) *Pack Sizes: 10×10 Blisters or Bottles of 60/100 (Optimized specifically for chronic, twice-daily dermatological dispensing regimens).
Nicoumalone Tablets
FreeNicoumalone Tablets IP 2 mg
Each uncoated tablet contains :
Nicoumalone IP 2 mgNicoumalone Tablets IP 4 mg
Each uncoated tablet contains :
Nicoumalone IP 4 mgUsage: – Prevent and treat harmful blood clots
Category: – Hypertensive / Cardiac Drugs
Therapeutic category: – Hypertensive
Nifedipine Tablets
FreeNifedipine Tablets IP 10 mg
Each uncoated tablet contains:
Nifedipine IP 10 mg
Excipients – QSNifin-10
Nifedipine Tablets 10 mg
Each Uncoated Tablet Contains:
Nifedipine BP 10 mgNifin-20
Nifedipine Tablets 20 mg
Each Uncoated Tablet Contains:
Nifedipine BP 20 mgNifelace – 20
Nifedipine slow release tablets
Each Filmcoated slow release tablet contains :
Nifedipine BP 20 mgUsage: – Treat high blood pressure
Category: – Anti Cardiovascular Drugs
Therapeutic category: – Cardiovascular Agent, hypertensive
Nimesulide ,Paracetamol & Chlorzoxazone Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Rapid-Burst Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of all three active ingredients.
Active Ingredient Strength Primary Clinical Function Nimesulide BP/Ph.Eur. 100 mg Preferential COX-2 NSAID: Rapid abortion of localized musculoskeletal inflammation and tissue swelling. Paracetamol (Acetaminophen) BP/Ph.Eur. 325 mg Central Analgesic: Synergistic pain threshold elevation and antipyretic control. Chlorzoxazone USP/Ph.Eur. 250 mg Central Muscle Relaxant: Direct inhibition of spinal reflex arcs to break severe, locked muscle spasms. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Povidone K30 / Magnesium Stearate / Premium Opadry Film Diluent / Superdisintegrant / Binder / Moisture-Barrier Coating (Engineered specifically to trigger a violent, immediate structural collapse in the stomach, ensuring all three APIs are absorbed simultaneously for synchronized relief) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict, short-term acute dispensing to ensure patients do not exceed the mandated 15-day Nimesulide therapy limit).
Nimesulide Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Rapid-Burst Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the active ingredient.
Active Ingredient Strength Primary Clinical Function Nimesulide BP/Ph.Eur. 100 mg Global Acute Standard: High-efficacy adult therapeutic unit for the rapid, short-term abortion of acute inflammatory pain. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Docusate Sodium / Povidone K30 / Magnesium Stearate Diluent / Superdisintegrant / Wetting Surfactant / Binder (Engineered specifically to trigger a violent, immediate structural collapse in the stomach, maximizing the drug’s surface area for ultra-fast gastrointestinal absorption) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict, short-term acute dispensing to ensure patients do not exceed the mandated 15-day therapy limit).
Nimesulide, Paracetamol & Serratiopeptidase Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Multi-Particulate Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the highly delicate enzyme component.
Active Ingredient Strength Primary Clinical Function Nimesulide BP/Ph.Eur. 100 mg Preferential COX-2 NSAID: Rapid abortion of localized musculoskeletal inflammation and tissue pain. Paracetamol (Acetaminophen) BP/Ph.Eur. 325 mg Central Analgesic: Synergistic pain threshold elevation and antipyretic control. Serratiopeptidase (as Enteric-Coated Granules) 15 mg
(Equivalent to 30,000 Serratiopeptidase Units)Proteolytic Enzyme: Direct breakdown of fibrin and inflammatory exudate to physically drain severe localized edema. Excipients Methacrylic Acid Copolymer / Microcrystalline Cellulose / Croscarmellose Sodium / Povidone K30 / Magnesium Stearate Enteric Polymer Shield / Diluent / Superdisintegrant / Binder (Engineered specifically to protect the enzyme from gastric acid while allowing the analgesics to dissolve instantly in the stomach) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict, short-term acute dispensing to ensure patients do not exceed the mandated 15-day Nimesulide therapy limit).
Nitrazepam Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Tablet, packed exclusively in highly secure, tamper-evident Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the active ingredient and prevent unauthorized access.
Active Ingredient Strength Primary Clinical Function Nitrazepam BP/Ph.Eur. 5 mg Initiation / Elderly Standard: Base therapeutic unit for the short-term management of severe insomnia, specifically titrated for elderly patients to prevent next-day falls. Nitrazepam BP/Ph.Eur. 10 mg Severe Psychiatric Standard: Maximum adult dose to force sleep in highly agitated, refractory, or acutely manic patients. Excipients Lactose Monohydrate / Maize Starch / Microcrystalline Cellulose / Magnesium Stearate / Colloidal Silicon Dioxide Diluent / Binder / Disintegrant / Lubricant (Engineered specifically for rapid gastric dissolution, inducing sleep within 30 to 60 minutes of ingestion) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict 2-to-4 week acute psychiatric dispensing regimens to prevent dependency).
Nitrofurantoin Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Dual-Release Capsule or Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the nitrofuran compounds.
Active Ingredient Strength Primary Clinical Function Nitrofurantoin (Macrocrystals) USP/Ph.Eur. 50 mg Prophylactic / Pediatric Standard: Base therapeutic unit for long-term prevention of recurrent UTIs and pediatric dosing. Nitrofurantoin (Macrocrystals 25mg + Monohydrate 75mg) USP 100 mg Global Acute UTI Standard: Premium dual-release adult maintenance dose for the rapid eradication of acute uncomplicated cystitis. Excipients Carbomer 934P / Povidone K30 / Sugar Spheres / Talc / Magnesium Stearate / Titanium Dioxide Hydrophilic Polymer Matrix / Binder / Core Substrate / Glidant (Engineered specifically to form a viscous gel upon swallowing, pacing the gastric release perfectly to prevent nausea while ensuring maximum urinary concentration) *Pack Sizes: 10×10 Blisters or Bottles of 100 (Optimized specifically for strict 5-to-7 day acute urology dispensing regimens).
Nitrofurantoin Tablets
FreeA Nitrofurantoin tablet is an antibiotic specifically used to treat and prevent urinary tract infections (UTIs), such as cystitis. It works by damaging the DNA of bacteria in the urinary tract.
Nitroglycerin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Concentrate for Infusion in glass ampoules or vials. (Must be diluted).
Active Ingredient Strength Total Content Nitroglycerin USP/BP 5 mg/ml 25 mg (in 5 ml Ampoule) Nitroglycerin USP/BP 5 mg/ml 50 mg (in 10 ml Vial) Excipients Propylene Glycol / Ethanol Solubilizers (Volatile) *Pack Sizes: Tray of 5 Ampoules or 10 Vials.
Nitroglycerin Tablets
FreeNitroglycerin Tablets USP
Each Sublingual Tablet Contains :
Dilute Nitroglycerin USP
Eq.to Nitroglycerin 0.5 mg
Excipients qsUsage: – Treat episodes of angina (chest pain)
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Norethisterone Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of the highly potent steroid hormone.
Active Ingredient Strength Primary Clinical Function Norethisterone Ph.Eur./USP 5 mg Global Gynecology Standard: High-dose therapeutic unit for acute hemorrhage cessation, endometriosis suppression, and menstrual cycle manipulation. Excipients Lactose Monohydrate / Maize Starch / Magnesium Stearate / Povidone K30 Diluent / Binder / Lubricant (Engineered specifically for rapid gastric dissolution to halt acute uterine bleeding within 24-48 hours of the first dose) Pack Sizes: 10×10 Blisters or 3×10 Calendar Packs (Optimized specifically for strict cyclical gynecology dispensing regimens).
Norfloxacin & Tinidazole Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu or Alu-PVC blister strips to ensure the absolute chemical stability of the highly photolabile active ingredients.
Active Ingredient Strength Primary Clinical Function Norfloxacin USP/Ph.Eur. 400 mg Aerobic Gut/UTI Standard: Broad-spectrum eradication of Gram-negative/positive bacteria driving acute gastroenteritis and genitourinary infections. Tinidazole USP/Ph.Eur. 600 mg Anaerobic/Protozoal Standard: Highly tolerable eradication of tissue-invasive amoebas, giardia, and pelvic anaerobes. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Povidone K30 / Magnesium Stearate / Hypromellose / Titanium Dioxide Diluent / Superdisintegrant / Binder / Premium Opaque Film-Coating (Engineered specifically to form an impenetrable barrier that completely masks the notoriously bitter, metallic taste of both APIs, guaranteeing patient compliance) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict 5-to-10 day acute anti-infective dispensing regimens).
Norfloxacin Tablets
FreeNorfloxacin Tablets IP 400 mg
Each Film Coated Tablet Contains:
Norfloxacin IP 400 mgFlonor Tablets
Norfloxacin Tablets BP 400 mg
Each Filmcoated tablet contains :
Norfloxacin BP 400 mgUsage: – Treat a variety of bacterial infections
Category: – Antibiotic Drugs
Therapeutic category: – Antibiotic
Nystatin Vaginal Tablets
FreeNygen – V
(Nystatin Vaginal tablet USP)
Each Uncoated tablet contains :
Nystatin USP 1,00,000 IUNystatin Tablets USP
Each Uncoated tablet contains :
Nystatin USP 5,00,000 IUUsage: – Treat yeast infections of the vagina
Category: – Antifungal Drugs
Therapeutic category: – Antifungal
Ofloxacin & Ornidazole Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu or Alu-PVC blister strips to ensure the absolute chemical stability of the highly photolabile active ingredients.
Active Ingredient Strength Primary Clinical Function Ofloxacin USP/Ph.Eur. 200 mg Aerobic Standard: Broad-spectrum eradication of Gram-negative/positive bacteria driving acute gastroenteritis and genitourinary infections. Ornidazole INN/Ph.Eur. 500 mg Anaerobic/Protozoal Standard: Highly tolerable eradication of tissue-invasive amoebas, giardia, and surgical-site anaerobes. Excipients Microcrystalline Cellulose / Sodium Starch Glycolate / Povidone K30 / Magnesium Stearate / Hypromellose / Titanium Dioxide Diluent / Superdisintegrant / Binder / Premium Opaque Film-Coating (Engineered specifically to form an impenetrable barrier that completely masks the notoriously bitter, metallic taste of both APIs, guaranteeing patient compliance) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict 3-to-5 day acute anti-infective dispensing regimens).
Ofloxacin injection
FreeComposition
Ofloxacin 200 mg / 100 ml
Usage: – Treat bacterial infections
Category: – Antibiotic injection
Therapeutic category: – Antibiotic
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Ofloxacin Tablets
FreeOfloxacin Tablets 200 mg
Each Filmcoated tablet contains :
Ofloxacin IP 200 mg
Excipients q.sYALLEY FLOX
Ofloxacin Tablets 200mg Each Film coated tablet contains :
Ofloxacin BP 200 mg
Excipients q.sOfnis -200
Ofloxacin Tablets
Each Filmcoated tablet contains :
Ofloxacin BP 200 mgUsage: – Treat bacterial infections
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic
Olanzapine Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder (Yellow Cake) in glass vials.
Active Ingredient Strength Reconstitution Requirement Olanzapine BP/USP 10 mg Requires 2.1 ml of Sterile Water for Injection Excipients Lactose Monohydrate / Tartaric Acid Bulking Agent / Solubilizer *Pack Sizes: Single Vial. (Produces a clear, yellow solution upon reconstitution).
Olanzepine Tablets
FreeOlanzepine Tablets IP 2.5 mg
Each film coated tablet contains:
Olanzepine IP 2.5 mgOlanzepine Tablets IP 5 mg
Each film coated tablet contains:
Olanzepine IP 5 mgOlanzepine Tablets IP 7.5 mg
Each film coated tablet contains
Olanzepine IP 7.5 mgOlanzepine Tablets IP 10 mg
Each film coated tablet contains:
Olanzepine IP 10 mgUsage: – Treat psychotic conditions such as schizophrenia and bipolar disorder (manic depression)
Category: – Anti Anxiety drugs
Therapeutic category: – Antipsychotic, Antidepressant, CNS Drugs, Anti Anxiety
Olmesartan Medoxomil Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the highly volatile ester prodrug.
Active Ingredient Strength Primary Clinical Function Olmesartan Medoxomil Ph.Eur./USP 20 mg Initiation / Global Standard: Standard adult starting and maintenance dose for the continuous control of essential hypertension. Olmesartan Medoxomil Ph.Eur./USP 40 mg Severe Refractory Standard: Maximum adult maintenance dose for patients requiring further reduction in blood pressure. Excipients Microcrystalline Cellulose / Lactose Monohydrate / Hydroxypropyl Cellulose / Magnesium Stearate / Titanium Dioxide Low-Moisture Diluent / Binder / Lubricant / Premium Moisture-Barrier Film-Coating (Engineered specifically to lock out ambient humidity and prevent the premature hydrolysis of the medoxomil group prior to ingestion) *Pack Sizes: 10×10 Alu-Alu Blisters or 3×10 Unit-Dose Blisters (Optimized specifically for strict 30-day chronic cardiology dispensing regimens).
Olmesartan, Amlodipine & Hydrochlorothiazide Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered, API-Segregated Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of all three active ingredients across their entire shelf life.
Active Ingredient Strength Primary Clinical Function Olmesartan Medoxomil Ph.Eur./USP 20 mg / 40 mg ARB Base: Blocks the renin-angiotensin-aldosterone system (RAAS) and provides critical diabetic renoprotection. Amlodipine (as Besylate) Ph.Eur./USP 5 mg / 10 mg CCB Vasodilator: Forces profound, sustained peripheral arterial relaxation. Hydrochlorothiazide (HCTZ) Ph.Eur./USP 12.5 mg / 25 mg Diuretic Volume Controller: Flushes excess sodium/water and massively amplifies the efficacy of the ARB component. Excipients Silicified Microcrystalline Cellulose / Pregelatinized Starch / Croscarmellose Sodium / Magnesium Stearate / Opadry Protective Film Moisture-Scavenging Diluent / Binder / Superdisintegrant / Premium Film-Coating (Engineered specifically to physically isolate the highly sensitive Olmesartan ester from the alkaline Amlodipine salt to prevent cross-degradation) *Pack Sizes: 10×10 Alu-Alu Blisters or 3×10 Unit-Dose Blisters (Optimized specifically for strict 30-day chronic cardiology dispensing regimens).
Omeprazole Capsules
FreeOmeprazole capsules typically contain enteric-coated pellets to prevent the stomach’s acid from breaking down the medication before it can be absorbed in the small intestine.
Omeprazole Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder (White to Off-White Cake) in clear glass vials, typically accompanied by a specialized sterile solvent.
Active Ingredient Strength Reconstitution Requirement Omeprazole Sodium USP/BP 40 mg Requires provided 10 ml solvent or specific diluent Excipients Sodium Hydroxide / Disodium Edetate Alkalizing Agent / Stabilizer *Pack Sizes: Single Vial + 10 ml Solvent Ampoule, or Tray of 10 Vials.
Omeprazole Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Enteric-Coated Capsule or Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the highly acid-labile benzimidazole derivative.
Active Ingredient Strength Primary Clinical Function Omeprazole BP/USP/Ph.Eur. 10 mg OTC / Maintenance Standard: Approved unit for over-the-counter frequent heartburn relief and long-term remission maintenance of healed esophagitis. Omeprazole BP/USP/Ph.Eur. 20 mg Global Clinical Standard: Primary adult daily dose for active GERD, gastric ulcers, and standard H. pylori eradication protocols. Omeprazole BP/USP/Ph.Eur. 40 mg Severe Refractory Standard: High-dose therapy for severe erosive esophagitis, NSAID-induced bleeding ulcers, and Zollinger-Ellison Syndrome. Excipients Methacrylic Acid Copolymer / Hypromellose (HPMC) / Sugar Spheres / Macrogol / Titanium Dioxide pH-Dependent Enteric Coating / Sub-coating Barrier / Core Substrate / Plasticizer (Engineered specifically to resist gastric fluid at pH 1.2 but dissolve instantly at duodenal pH 6.8) *Pack Sizes: 10×10 Alu-Alu Blisters or Bottles of 100 (Optimized specifically for strict 14-to-28 day acute GI healing regimens).
Ondansetron injection
FreeComposition
Each ml contains: Ondansetron Hcl
(as dihydrate) Eq. to Ondansetron 2mg ; Water for injection q.s.
Each ml Contains: Ondansetron Hydrochloride
Eq to Ondansetron 2 mg.
Water for Injection Q.S. 2 ml, 4 mlUsage: – Trigger nausea and vomiting
Category: – Antacid & Anti ulcer injection
Therapeutic category: – Antiemetic, Anti Vomiting
Ondansetron Injection
FreeProduct Composition & Strength
We supply this product as a Sterile, Clear, Aqueous Solution in glass ampoules or vials.
Active Ingredient Concentration Total Content & Format Ondansetron (as HCl Dihydrate) 2 mg / ml 4 mg in 2 ml Glass Ampoule Ondansetron (as HCl Dihydrate) 2 mg / ml 8 mg in 4 ml Glass Ampoule Excipients Citric Acid / Sodium Citrate pH Buffer System (pH 3.3 to 4.0) *Pack Sizes: Tray of 5/10 Ampoules or Box of 50 Ampoules.
Ondansetron Tablets
FreeOndansetron Tablets IP 4mg
Each uncoated tablet contains:
Ondansetron Hydrochloride Dihydrate IP
Eq. Anhydrous Ondansetron 4 mg
Excipients q.sXysetron Tablets Ondansetron Tablets 4 mg Each Uncoated Tablet contains :
Ondansetron Hydrochloride USP
Eq. To Anhydrous Ondansetron 4 mgNausedon 4
Ondansetron Tablets 4mg
Each uncoated tablet contains:
Ondansetron HCL Dihydrate BP
Eq. Anhydrous Ondansetron 4 mgXysetron 8 Tablets Ondansetron Tablets 8 mg
Each Uncoated Tablet contains :
Ondansetron Hydrochloride USP
Eq. To Anhydrous Ondansetron 8 mgNausedon 8
Ondansetron Tablets 8mg
Each uncoated tablet contains:
Ondansetron HCL Dihydrate BP
Eq. Anhydrous Ondansetron 8 mgUsage: – Trigger nausea and vomiting
Category: – Antacid & Anti ulcer drugs
Therapeutic category: – Antiemetic, Anti Vomiting
Oral Rehydration Salts (Sachets)
FreeOral Rehydration Salts IP
Each 4.2 gm contains :
Sodium Chloride IP 0.52 gm
Potassium Chloride IP 0.30 gm
Sodium Citrate IP 0.58 gm
Anhydrous Glucose IP 2.70 gm
Excipients qs
Refreshing FlavourOral Rehydration Salts IP
Each 21 gm contains :
Sodium Chloride IP 2.6 gm
Potassium Chloride IP 1.5 gm
Sodium Citrate IP 2.9 gm
Anhydrous Dextrose IP 13.5 gm
Excipients qs
Refreshing Orange FlavourOral Rehydration Salts BP
Each 4.2 gm contains :
Sodium Chloride BP 0.52 gm
Potassium Chloride BP 0.30 gm
Sodium Citrate BP 0.58 gm
Anhydrous Glucose BP 2.70 gm
Refreshing FlavourOral Rehydration Salts BP
Each 21 gm contains :
Sodium Chloride BP 2.6 gm
Potassium Chloride BP 1.5 gm
Sodium Citrate BP 2.9 gm
Anhydrous Glucose BP 13.5 gm
Refreshing Orange FlavourORASEL / ORAL REHYDRATION SALT B.P. Each 21 gm sachet contains: Anhydrous glucose BP 13.5 g Sodium Chloride BP 2.6 g Sodium citrate BP 2.9 g Potassium Chloride BP 1.5 g Refreshing orange Flavour
HEALTHY ORS /ORAL REHYDRATION SALTS BP EACH 20.5 GM CONTAINS:: POTASSIUM CHLORIDE BP 1.5 GM
– SODIUM CITRATE BP 2.9 GM
– ANHYDROUS GLUCOSE BP 13.5 GM
– SODIUM CHLORIDE BP BP 2.6 GM– EXCIPIENTS QS
Usage: – To replace salts and water that the body loses when you have dehydration caused by gastroenteritis, diarrhea, or vomiting
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Ornidazole Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu or Alu-PVC blister strips to ensure the absolute chemical stability of the highly photolabile active ingredient.
Active Ingredient Strength Primary Clinical Function Ornidazole INN/Ph.Eur. 500 mg Global Clinical Standard: Base therapeutic unit for the rapid eradication of protozoal and anaerobic pathogens. Excipients Microcrystalline Cellulose / Maize Starch / Sodium Starch Glycolate / Hypromellose / Titanium Dioxide Diluent / Binder / Superdisintegrant / Premium Film-Coating (Engineered specifically to create an impenetrable barrier that completely masks the notoriously bitter, foul taste of the nitroimidazole API, ensuring flawless patient compliance) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict 3-to-10 day acute anti-infective dispensing regimens).
ORS – Oral Rehydration Salt
FreeOral Rehydration Salts IP
Each 4.2 gm contains :
Sodium Chloride IP 0.52 gm
Potassium Chloride IP 0.30 gm
Sodium Citrate IP 0.58 gm
Anhydrous Glucose IP 2.70 gm
Excipients qs
Refreshing FlavourOral Rehydration Salts IP
Each 21 gm contains :
Sodium Chloride IP 2.6 gm
Potassium Chloride IP 1.5 gm
Sodium Citrate IP 2.9 gm
Anhydrous Dextrose IP 13.5 gm
Excipients qs
Refreshing Orange FlavourOral Rehydration Salts BP
Each 4.2 gm contains :
Sodium Chloride BP 0.52 gm
Potassium Chloride BP 0.30 gm
Sodium Citrate BP 0.58 gm
Anhydrous Glucose BP 2.70 gm
Refreshing FlavourOral Rehydration Salts BP
Each 21 gm contains :
Sodium Chloride BP 2.6 gm
Potassium Chloride BP 1.5 gm
Sodium Citrate BP 2.9 gm
Anhydrous Glucose BP 13.5 gm
Refreshing Orange FlavourORASEL / ORAL REHYDRATION SALT B.P. Each 21 gm sachet contains: Anhydrous glucose BP 13.5 g Sodium Chloride BP 2.6 g Sodium citrate BP 2.9 g Potassium Chloride BP 1.5 g Refreshing orange Flavour
HEALTHY ORS /ORAL REHYDRATION SALTS BP EACH 20.5 GM CONTAINS:: – POTASSIUM CHLORIDE BP 1.5 GM
– SODIUM CITRATE BP 2.9 GM
– ANHYDROUS GLUCOSE BP 13.5 GM
– SODIUM CHLORIDE BP BP 2.6 GM – EXCIPIENTS QSUsage: – To replace salts and water that the body loses when you have dehydration caused by gastroenteritis, diarrhea, or vomiting
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Oxacillin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Crystalline Powder buffered for stability in glass vials.
Active Ingredient Strength Sodium Content Oxacillin Sodium USP/BP 500 mg Approx. 1.4 mEq (32 mg) Oxacillin Sodium USP/BP 1000 mg (1 g) Approx. 2.8 mEq (64 mg) Oxacillin Sodium USP/BP 2000 mg (2 g) High-Dose Protocols Buffer Dibasic Sodium Phosphate Maintains pH upon reconstitution *Pack Sizes: Single Vial or Tray of 10/50 Vials. (Requires reconstitution with Sterile Water for Injection).
Oxaliplatin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile, Clear, Aqueous Concentrate in glass vials. (Also available as Lyophilized Powder depending on market preference).
Active Ingredient Strength Volume Oxaliplatin USP/BP 5 mg / ml 10 ml Vial (Total 50 mg) Oxaliplatin USP/BP 5 mg / ml 20 ml Vial (Total 100 mg) Excipients Water for Injection Preservative-Free Solution *Pack Sizes: Single Vial. (Must be handled with strict cytotoxic precautions).
Oxcarbazepine Tablets
FreeOxcarbazepine Tablets IP 150 mg
Each Uncoated tablet contains:
Oxcarbazepine IP 150 mgOxcarbazepine Tablets IP 300 mg
Each Uncoated tablet contains:
Oxcarbazepine IP 300Oxcarbazepine Tablets IP 600 mg
Each Uncoated tablet contains:
Oxcarbazepine IP 600 mgUsage: – Seizures and Pain
Category: – Antiepileptic drugs
Therapeutic category: – Antiepileptic, Anticonvulsant
Oxybutynin Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Polymeric Matrix Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips or heavy-gauge HDPE desiccant bottles to ensure the absolute chemical stability of the controlled-release hydrogel network.
Active Ingredient Strength Primary Clinical Function Oxybutynin Chloride USP/Ph.Eur. 5 mg (ER) Initiation Standard: Base once-daily titration unit for the gradual introduction of antimuscarinic therapy. Oxybutynin Chloride USP/Ph.Eur. 10 mg / 15 mg (ER) Global Clinical Standard: Advanced maintenance doses for severe, refractory Overactive Bladder and neurogenic detrusor overactivity. Excipients Hypromellose (HPMC) / Lactose Anhydrous / Macrogol (PEG) / Magnesium Stearate / Titanium Dioxide Hydrophilic Polymer Matrix / Diluent / Plasticizer / Premium Film-Coating (Engineered specifically to form a highly viscous external gel barrier upon swallowing, controlling the exact 24-hour drug diffusion rate) *Pack Sizes: Bottles of 30 or 10×10 Alu-Alu Blisters (Optimized specifically for strict, high-compliance 30-day chronic urology dispensing regimens).
Oxytetracycline injection
FreeComposition
Each ml Contains :
Oxytetracycline Hcl I.P. …….. 50 mg
Lidocain U.S.P. ……………… 2 %
Water for Injection I.P. Q.S. 30 mlUsage: – used in medicine to treat various bacterial infections
Category: – antibiotic injection
Therapeutic category: – antibiotic
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Oxytetracycline Veterinary injection
FreeComposition
Oxytetracycline 5% 100 ML vet INJ
Oxytetracycline 5% 30 ML vet INJ
Oxytetracycline 20% (LA) 50 vet ML
Usage: – used in veterinary medicine to treat various bacterial infections in animals
Category: – Veterinary injection
Therapeutic category: – Veterinary medicine
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Pancreatin & L-Ornithine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Bilayer Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute stability of the biological proteins and the highly hygroscopic amino acid.
Active Ingredient Strength Primary Clinical Function Pancreatin (Porcine Origin) Ph.Eur./USP 170 mg
(Standardized Lipase/Protease/Amylase)Gastrointestinal Standard: Intraluminal enzymatic digestion of complex macronutrients to reverse malabsorption. L-Ornithine (as L-Ornithine L-Aspartate / LOLA) 150 mg Hepatology Standard: Hepatic urea cycle activation for aggressive ammonia detoxification and liver regeneration. Excipients Methacrylic Acid Copolymer / Microcrystalline Cellulose / Crospovidone / Magnesium Stearate Enteric Barrier Coating (for Pancreatin) / Diluent / Superdisintegrant (Engineered specifically to separate the amino acid from the enzymes and ensure targeted duodenal release of the biologicals) *Pack Sizes: 10×10 Alu-Alu Blisters (Optimized specifically for strict, high-compliance chronic hepato-gastro dispensing regimens).
Pancreatin Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Enteric-Coated Tablet or Hard Capsule (containing Enteric Minimicrospheres), packed exclusively in highly secure, moisture-resistant Alu-Alu blisters or desiccant-lined HDPE bottles to ensure the absolute stability of the biological proteins.
Active Ingredient Strength (Standardized USP Units) Primary Clinical Function Pancreatin (Porcine Origin) Ph.Eur./USP 10,000 Lipase Units
(approx. 33,200 Protease / 33,200 Amylase)Pediatric / Initiation Standard: Base therapeutic unit for pediatric Cystic Fibrosis patients and meal-snack titration. Pancreatin (Porcine Origin) Ph.Eur./USP 25,000 Lipase Units
(approx. 85,000 Protease / 74,700 Amylase)Global Clinical Standard: High-dose adult maintenance therapy for severe Exocrine Pancreatic Insufficiency and Pancreatectomy. Excipients Methacrylic Acid Copolymer / Macrogol / Simethicone / Microcrystalline Cellulose / Triethyl Citrate Enteric Barrier Coating / Plasticizer / Anti-Foaming Agent / Diluent (Engineered specifically to protect the biological extract from gastric acid and ensure rapid, synchronized dissolution in the duodenum) *Pack Sizes: Bottles of 100 or 10×10 Alu-Alu Blisters (Optimized specifically for strict, high-volume daily dispensing where patients may consume 3 to 10 units daily with every meal and snack).
Panthenol & Crude veterinary injection
FreeComposion
Panthenol Choline Chloride & Crude Liver VET INJ. 100 ML
Usage: –Injections of choline chloride may be used in veterinary medicine
Category: – veterinary injection
Therapeutic category: – veterinary medicine.
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Pantoprazole injection
FreeComposition
Pantoprazole Sodium 40 mg IV (VIAL) Vial
Usage: – Heartburn, acid reflux and gastro-esophageal reflux disease
Category: – Antacid & Anti ulcer drugs
Therapeutic category: – Antiulcer, Antacid
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Pantoprazole Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder (White to Off-White Cake) in clear glass vials.
Active Ingredient Strength Reconstitution Requirement Pantoprazole Sodium USP/BP 40 mg Requires 10 ml of 0.9% Sodium Chloride Excipients Sodium Hydroxide / Edetate Disodium Alkalizing Agent / Stabilizer *Pack Sizes: Single Vial or Tray of 10 Vials. (Often packed with a 10 ml ampoule of 0.9% NaCl diluent).
Pantoprazole Tablets
FreePantoprazole Tablets IP 40 mg
Each Enteric Coated tablet contains
Pantoprazole Sodium IP
Eq. To Pantoprazole 40 mgPenta-20
Pantoprazole Tablets 20 mg
Each Enteric Coated Tablet Contains
Pantoprazole Sodium Sesquihydrate
Eq To Pantoprazole 20 mgPenta – 40
Pantoprazole Tablets 40 mg
Each Entreric Coated tablet contains
Pantoprazole Sodium Sesquihydrate
Eq. To pantoprazole 40 mgUsage: – Heartburn, acid reflux and gastro-esophageal reflux disease
Category: – Antacid & Anti ulcer drugs
Therapeutic category: – Antiulcer, Antacid
Paracetamol & Caffeine Anhydrous Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets or Uncoated Tablets. The anhydrous caffeine allows for a harder, more durable tablet suitable for bulk transport.
Active Ingredient Strength (Standard) Strength (Extra Power) Technical Grade Paracetamol IP/BP/USP 500 mg 650 mg Micronized Caffeine Anhydrous IP/BP/USP 30 mg 50 mg / 65 mg Moisture Content < 0.5% Excipients Q.S. Starch / PVP K-30 Binder Paracetamol & Diclofenac Potassium Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets (typically White, Orange, or Red). The coating is non-enteric to ensure immediate disintegration.
Active Ingredient Strength (Standard) Therapeutic Role Paracetamol IP/USP 325 mg / 500 mg Central Analgesic Diclofenac Potassium IP/USP 50 mg Rapid Anti-Inflammatory Excipients Q.S. Microcrystalline Cellulose / Sodium Starch Glycolate Binder / Super-Disintegrant Paracetamol & Diclofenac Sodium Tablets
FreeProduct Composition & Strength
We supply this product as Enteric Coated or Film Coated Tablets in blister packs.
Active Ingredient Strength (Standard) Role Diclofenac Sodium IP/BP/USP 50 mg Anti-inflammatory / Analgesic Paracetamol IP/BP/USP 500 mg Analgesic / Antipyretic Excipients Q.S. Binder / Disintegrant Paracetamol & Orphenadrine Citrate Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets in blister packs. We offer both Standard and Forte strengths.
Product Variant Paracetamol Content Orphenadrine Citrate Common Name Standard Strength 450 mg 35 mg Norgesic Generic Forte Strength 650 mg 50 mg Norgesic Forte Generic Excipients Q.S. — Binder / Filler Paracetamol Caffeine Chlorpheniramine Maleate Phenylephrine
FreeProduct Composition & Standard Strengths
We supply this product in standardized Fixed-Dose Combinations (FDCs) optimized for maximum clinical efficacy.
Active Ingredient Strength (per Tablet) Primary Clinical Function Paracetamol (Acetaminophen) 325 mg / 500 mg The Pain & Fever Reliever: Targets aches and chills. Caffeine (Anhydrous) 30 mg The Potentiator: Boosts pain relief and alertness. Chlorpheniramine Maleate 2 mg / 4 mg The Antiallergic: Stops sneezing and runny nose. Phenylephrine HCl 5 mg / 10 mg The Decongestant: Clears blocked sinuses. paracetamol caffeine phenylephrine cpm Tablets
FreeHecold
Each Uncoated Tablet contains :
Paracetamol BP 500 mg
Caffeine BP 30 mg
Phenylephrine Hydrochloride BP 2.5 mg
Chlorpheniramine Maleate BP 4 mgHecold Strong
Each Uncoated Tablet contains :
Paracetamol BP 500 mg
Caffeine BP 30 mg
Phenylephrine Hydrochloride BP 5 mg
Chlorpheniramine Maleate BP 4 mgParacetamol Diclo Sodium Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated or Enteric Coated Tablets (typically White, Orange, or Yellow). The coating protects the stomach from the direct irritation of Diclofenac.
Active Ingredient Strength (Standard) Therapeutic Role Paracetamol IP/USP 500 mg / 325 mg Central Analgesic / Antipyretic Diclofenac Sodium IP/USP 50 mg Peripheral Anti-Inflammatory Excipients Q.S. Maize Starch / Magnesium Stearate / Talc Binder / Lubricant Paracetamol Injection
FreeProduct Composition & Strength
We supply this product as a Sterile, Clear, Iso-osmotic Solution ready for direct intravenous infusion.
Active Ingredient Concentration Total Content & Format Paracetamol BP/USP 10 mg / ml 1000 mg (1 g) in 100 ml Glass Bottle / FFS Plastic Bottle Paracetamol BP/USP 150 mg / ml 300 mg in 2 ml Glass Ampoule (IM/IV use) Excipients Mannitol / Cysteine Hydrochloride Isotonic Agent / Oxygen Scavenger (Antioxidant) *Pack Sizes: 100 ml Single Bottle or Box of 10 Ampoules.
Paracetamol injection
FreeProduct Composition & Strength
We supply this product as a ready-to-use infusion in glass bottles or IV bags.
Active Ingredient Strength Total Content Paracetamol IP/USP 10 mg / ml 1000 mg / 100 ml *Supplied in 100ml Glass Bottles or FFS (Form-Fill-Seal) Plastic Bottles.
Paracetamol Tablet
Freeparacetamol tablets manufacturer & exporter supplier
Paracetamol Tablets IP
Each Uncoated tablet contains :
Paracetamol IP 500 mgParacetamol Tablets IP 650 mg
Each uncoated Tablet Contains: Paracetamol IP 650 mg
– Excipients – QSParafen forte
Paracetamol with Ibuprofen Tablets
Each Uncoated tablet contains :
Ibuprofen IP 400 mg
Paracetamol IP 325 mgwe have licenece for export also
Paracetamol Tablets
FreeParacetamol Tablets IP
Each Uncoated tablet contains :
Paracetamol IP 500 mgParacetamol Tablets IP 650 mg
Each uncoated Tablet Contains:
Paracetamol IP 650 mg
Excipients – QSParacetamol Tablets BP
Each Uncoated tablet contains :
Paractamol BP 100 mgParacetamol Tablets BP
Each Uncoated tablet contains :
Paracetamol BP 250 mgParafen te
Paracetamol with Ibuprofen Tablets
Each Uncoated tablet contains :
Ibuprofen IP 400 mg
Paracetamol IP 325 mgPARACETAMOL AND CAFFEINE TABLETS
Each uncoated Tablets Contains:
Paracetamol BP 325 mg
Caffeine anhydrous BP 30 mg
Excipients – QSParacetamol and Orphenadrine Citrate Tablets
Each uncoated tablet contains:
Orphenadrine citrate 35 mg
Paracetamol 450 mg
Excipients – QS
Each Uncoated Caplet contains :
Paracetamol BP 500 mg
Caffeine Anhydrous BP 30 mgPARACETAMOL AND CAFFEINE
Each uncoated Tablet contains:
Paracetamol BP 325 mg
Caffeine BP 30 mg
q.s. –PARACETAMOL,CAFFEINE,PHENYLEPHRINE HCL AND CHOLRPHENIRAMINE MALEATE TABLETS
Each Uncoated Tablet Contains:
Paracetamol BP 325 mg
Caffeine BP 30 mg
Chlorpheniramine maleate BP 2 mg
Phenylephrine Hcl BP 2.5 mg
Colour : Sunset Yellow –
Excipients – QSPARACETAMOL,CAFFEINE,PHENYLEPHRINE AND CHLORPHENIRAMINE TABLETS
Each uncoated Tablet contains:
Paracetamol BP 325 mg
Caffeine BP 15 mg
Phenylephrine BP 2.5 mg
Chlorpheniramine BP 2 mg
Tartrazine In House –Usage: – Treat aches and pain
Category: – Anti Analgesic Painkiller Drugs
Therapeutic category: – NSAID, Analgesic, Antipyretic, Anti Inflammatory
Paracetamol, Caffeine & Phenylephrine Tablets
FreeProduct Composition & Strength
We supply this product as Uncoated Tablets or Film Coated Tablets. The absence of an antihistamine makes this the ideal “Work-Safe” cold remedy.
Active Ingredient Strength (Standard) Therapeutic Role Paracetamol IP/BP/USP 500 mg / 650 mg Fever & Pain Reducer Caffeine Anhydrous IP/BP/USP 30 mg Alertness / Analgesic Booster Phenylephrine HCl IP/BP/USP 5 mg / 10 mg Nasal Decongestant Excipients Q.S. Povidone / Starch / MCC Paracetamol, Caffeine, Phenylephrine HCl & CPM Tablets
FreeProduct Composition & Strength
We supply this product as Uncoated Tablets or Film Coated Tablets (often colored Red, Blue, or Bi-layered). The formulation is balanced for adult physiology.
Active Ingredient Strength (Standard) Therapeutic Role Paracetamol IP/BP/USP 500 mg Fever & Pain Reducer Caffeine Anhydrous IP/BP/USP 30 mg Analgesic Potentiator / Alertness Phenylephrine HCl IP/BP/USP 5 mg / 10 mg Nasal Decongestant Chlorpheniramine Maleate IP/BP/USP 2 mg / 4 mg Antiallergic / Antihistamine Excipients Q.S. Starch / MCC / Colorants Paracetamol, Diclofenac Potassium & Caffeine Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets (often capsule-shaped). The coating masks the bitter taste of Caffeine and the astringency of Diclofenac.
Active Ingredient Strength (Standard) Therapeutic Role Paracetamol IP/BP/USP 325 mg / 500 mg Central Analgesic / Antipyretic Diclofenac Potassium BP/USP 50 mg Rapid Anti-Inflammatory Caffeine Anhydrous BP/USP 30 mg Adjuvant / Vasoconstrictor Excipients Q.S. Microcrystalline Cellulose / HPMC Paracetamol, Diclofenac Sodium & Caffeine Tablets
FreeProduct Composition & Standard Strengths
We supply this product in standardized Fixed-Dose Combinations (FDCs) designed for maximum therapeutic synergy.
Pazopanib Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered, High-Density Film-Coated Tablet, packed exclusively in highly secure, child-resistant heavy-gauge HDPE bottles or specialized Alu-Alu blister strips to ensure the absolute chemical stability of the cytotoxic molecule and prevent accidental exposure.
Active Ingredient Strength Primary Clinical Function Pazopanib HCl INN/Ph.Eur. 200 mg (Base Equivalent) Dose-Modification Standard: Essential unit for precise dose reduction in patients experiencing severe hepatic toxicity or adverse drug reactions. Pazopanib HCl INN/Ph.Eur. 400 mg (Base Equivalent) Global Clinical Standard: Primary therapeutic unit designed to achieve the massive 800 mg daily protocol with minimal pill burden. Excipients Microcrystalline Cellulose / Sodium Starch Glycolate / Povidone K30 / Magnesium Stearate / Hypromellose / Titanium Dioxide Diluent / Superdisintegrant / Binder / Lubricant / Premium Cytotoxic-Barrier Film-Coating (Engineered specifically to seal the lethal HPAPI inside the tablet, completely protecting healthcare workers and caregivers from highly toxic dust exposure) *Pack Sizes: Bottles of 30 or 60 Tablets (Optimized specifically for strict 30-day chronic oncology dispensing regimens).
Pemetrexed Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder (White to Light-Yellow Cake) in glass vials.
Active Ingredient Strength Reconstitution Pemetrexed Disodium 100 mg Requires 0.9% Sodium Chloride Pemetrexed Disodium 500 mg Requires 0.9% Sodium Chloride Pemetrexed Disodium 1000 mg (1 g) High-Dose Protocols Excipients Mannitol / Hydrochloric Acid / Sodium Hydroxide Bulking Agent / pH Adjusters (pH 6.6 – 7.8) *Pack Sizes: Single Vial. (Must be handled with strict cytotoxic precautions).
Pentoxifylline Tablets
FreePentoxifylline Tablets 400 mg
Each Filmcoated tablet contains :
Pentoxifylline BP 400 mgUsage: – To reduce leg pain caused by poor blood circulation
Category: – Anti Cardiovascular drugs
Therapeutic category: – Cardiovascular
Phenazopyridine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet, packed in highly secure, moisture-resistant PVC/PVDC or Alu-PVC blister strips to ensure the stability of the active dye compound.
Active Ingredient Strength Primary Clinical Function Phenazopyridine Hydrochloride USP/Ph.Eur. 100 mg Standard OTC/Initiation dose for mild-to-moderate urinary tract discomfort. Phenazopyridine Hydrochloride USP/Ph.Eur. 200 mg Global Prescription Standard: Maximum adult dose for severe dysuria and post-operative catheter trauma. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Magnesium Stearate Diluent / Superdisintegrant (Engineered for rapid gastric dissolution and swift renal clearance) Phenazopyridine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant high-density PVC/PVDC or Alu-PVC blister strips to ensure absolute chemical stability and prevent dye leakage.
Active Ingredient Strength Primary Clinical Function Phenazopyridine HCl USP/Ph.Eur. 100 mg OTC Standard: Base therapeutic unit for mild to moderate UTI symptom relief. Phenazopyridine HCl USP/Ph.Eur. 200 mg Prescription Standard: Maximum adult dose for severe, acute dysuria and post-surgical trauma. Excipients Microcrystalline Cellulose / Sodium Starch Glycolate / Povidone / Magnesium Stearate / Heavy Carnauba Wax Coating Diluent / Superdisintegrant / Binder / Lubricant / Premium Film-Coating (Engineered specifically to seal the intense red dye inside the tablet, preventing it from staining the patient’s hands or mouth upon swallowing) *Pack Sizes: 10×10 Blisters (Optimized specifically for high-turnover acute dispensing and short-term 2-day urology protocols).
Pheniramine Maleate Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Clear Solution in glass ampoules.
Active Ingredient Strength Total Content (2 ml) Pheniramine Maleate BP/USP 22.75 mg / ml 45.5 mg (per 2 ml Ampoule) Excipients Water for Injection Isotonic Solution *Pack Sizes: Tray of 10 Ampoules, 50 Ampoules, or 100 Ampoules.
PHENIRAMINE MALEATE INJECTION
FreeComposition
Each ml. contains: Pheniramine Maleate I.P. 22.75 mg.
Methyl Paraben I.P. 0.135 % w/v (As Preserva9ve)
Propyl Paraben I.P. 0.015% w/v (As Preserva9ve)
Water for Injec9on I.P. Q.SUsage: – It is used to relieve pruritus (itching) associated with various dermatological conditions.
Category: –Pheniramine Maleate injection
Therapeutic category: – Pheniramine Maleate injection
Pcd pharma franchise:-
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Pheniramine Maleate Tablets
FreePheniramine Maleate Tablets IP 25 mg
Each Uncoated Tablet Contains :
Pheniramine Maleate IP 25 mgPheniramine Maleate veterinary injection
FreeComposition
Pheniramine Maleate VET INJ. 30 ML
Usage: – It is used to relieve pruritus (itching) associated with various dermatological conditions.
Category: –Pheniramine Maleate injection
Therapeutic category: – Pheniramine Maleate injection
Pcd pharma franchise:-
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Phenyl Butazone & Sodium Salicylate veterinary injection
FreeComposition
Phenyl Butazone & Sodium Salicylate VET INJ. 30 ML
Phenyl Butazone & Sodium Salicylate VET INJ. 100 ML
Usage: – Usage in Veterinary Medicine: Phenylbutazone is frequently employed in horses, dogs, and other large animals.
Category: – veterinary injection
Therapeutic category: – veterinarian’s assessment
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Phenytoin Tablets
FreePhenytoin Sodium tablets IP
Each Sugarcoated tablet contains :
Phenytoin Sodium IP 100 mgPhenytoin Sodium Tablets IP 100 mg
Each Filmcoated Tablets Contains:
Phenytoin Sodium IP 100 mgPhenytoin Tablets BP
Each Sugar Coated tablet contains :
Phenytoin Sodium 25 mgPhenytoin Sodium Tablets BP
Each Sugar Coated tablet contains :
Phenytoin Sodium BP 100 mgUsage: – Treat epilepsy
Category: – Anti Hypertensive / Cardiac Drugs
Therapeutic category: – Anticonvulsant, Antiepileptic
Phytomenadione Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Aqueous Colloidal Solution or Mixed-Micelle Emulsion in glass ampoules.
Active Ingredient Strength Primary Indication Phytomenadione BP/USP 1 mg / 0.5 ml Pediatric: Prophylaxis of HDN Phytomenadione BP/USP 10 mg / 1 ml Adult: Anticoagulant Overdose / Bleeding Excipients Lecithin / Glycocholic Acid (or Cremophor) Solubilizer (Mixed-Micelle system) *Pack Sizes: Tray of 10 Ampoules or 100 Ampoules.
Phytomenadione Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Opaque Coated Tablet, packed exclusively in highly secure, moisture-resistant, and entirely light-blocking Alu-Alu or Opaque PVC/PVDC blister strips to ensure the absolute chemical stability of the photosensitive vitamin.
Active Ingredient Strength Primary Clinical Function Phytomenadione (Vitamin K1) BP/USP 10 mg Global Clinical Standard: Base therapeutic unit for the predictable reduction of elevated INR and treatment of hypoprothrombinemia. Excipients Microcrystalline Cellulose / Lactose Monohydrate / Colloidal Silicon Dioxide / Magnesium Stearate / Titanium Dioxide Diluent / Binder / Glidant / Lubricant / Heavy Opaque Film-Coating (Engineered specifically to form an impenetrable barrier against ultraviolet and visible light degradation) *Pack Sizes: 10×10 Blisters or Bottles of 100 (Optimized specifically for high-turnover pharmacy dispensing and cardiology ward stockpiles).
Piperacillin Sodium injection
FreeComposition
Piperacillin Sodium USP equivalent to
Piperacillin 4g + Tazobactam sodium equivalent to
Tazobactam 0.5mg injection
Pcd pharma franchise:-
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Piperacillin Tazobactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder buffered with Sodium Citrate/EDTA.
Strength (Ratio 8:1) Piperacillin Content Tazobactam Content 4.5 g (Standard Adult) 4000 mg (4 g) 500 mg (0.5 g) 2.25 g (Pediatric/Renal) 2000 mg (2 g) 250 mg (0.25 g) Sodium Content ~2.35 mEq per gram of Piperacillin Monitor in Heart Failure *Pack Sizes: Single Vial or Tray of 10 Vials.
Piracetam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Clear Solution in glass ampoules or large volume infusion bottles.
Active Ingredient Strength Volume Piracetam BP/USP 200 mg / ml 5 ml Ampoule (1 g Total) Piracetam BP/USP 200 mg / ml 15 ml Ampoule (3 g Total) Piracetam BP/USP 200 mg / ml 60 ml Infusion Bottle (12 g Total) Excipients Sodium Acetate / Acetic Acid Buffer (pH 5.0 – 7.0) *Pack Sizes: Tray of 5/10 Ampoules or Single Infusion Bottle.
Piracetam Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu or heavy-gauge PVC/PVDC blister strips to ensure the absolute chemical stability of the highly hygroscopic API.
Active Ingredient Strength Primary Clinical Function Piracetam Ph.Eur./USP 400 mg Initiation / Pediatric Standard: Base unit for pediatric dyslexia and mild vertigo management. Piracetam Ph.Eur./USP 800 mg Global Cognitive Standard: Standard adult maintenance dose for age-related cognitive decline and memory impairment. Piracetam Ph.Eur./USP 1200 mg Acute Neurology Standard: Massive-dose unit designed specifically for Cortical Myoclonus and intensive post-stroke rehabilitation protocols to minimize pill burden. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Macrogol (PEG) / Colloidal Silicon Dioxide / Magnesium Stearate High-Density Diluent / Superdisintegrant / Premium Film-Coating (Engineered specifically to compress the massive API load into a smooth, swallowable, moisture-resistant oblong tablet) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict, high-volume chronic neurology dispensing where patients may consume 2.4g to 4.8g daily).
Piroxicam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile, Clear, Pale Yellow to Yellow Aqueous-Solvent Solution in glass ampoules.
Active Ingredient Concentration Total Content & Format Piroxicam USP/BP 20 mg / ml 20 mg in 1 ml Glass Ampoule Piroxicam USP/BP 20 mg / ml 40 mg in 2 ml Glass Ampoule Excipients Propylene Glycol / Benzyl Alcohol Solvent System / Local Anesthetic (Preservative) *Pack Sizes: Tray of 10 Ampoules, 50 Ampoules, or 100 Ampoules.
Piroxicam Injection
FreeComposition
Each mL contains: Piroxicam BP : 20 mg
Benzyl Alcohol BP (as preserva9ve) : 2 % v/v
Water For Injec9on BP : q.s 1mlUsage: –used to manage postoperative pain after certain surgical procedures.
Category: – piroxicam injection
Therapeutic category: – piroxicam injection
Pcd pharma franchise:-
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Piroxicam Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Tablet or Hard Gelatin Capsule, packed exclusively in highly secure, moisture-resistant Alu-Alu or high-density PVC/PVDC blister strips to ensure the absolute chemical stability of the highly insoluble active ingredient.
Active Ingredient Strength Primary Clinical Function Piroxicam USP/Ph.Eur. 10 mg Initiation / Elderly Standard: Reduced dose for elderly patients or those with mild renal impairment to mitigate gastrointestinal risks. Piroxicam USP/Ph.Eur. 20 mg Global Clinical Standard: Maximum adult daily dose for the once-daily, continuous suppression of severe rheumatoid and osteoarthritis. Excipients Microcrystalline Cellulose / Lactose Monohydrate / Sodium Starch Glycolate / Sodium Lauryl Sulfate / Magnesium Stearate Diluent / Superdisintegrant / Wetting Agent / Lubricant (Engineered specifically with high-grade surfactants to force the rapid gastric dissolution of the highly water-insoluble Piroxicam API) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict 30-day chronic rheumatology and orthopedic dispensing regimens).