Antibiotics are a powerful class of pharmaceutical drugs used specifically to treat and prevent bacterial infections. In the 2026 pharmaceutical landscape, they remain the most critical tools in modern medicine, though their production and use have shifted toward combating the rising global challenge of Antimicrobial Resistance (AMR).
1. Major Classifications of Antibiotics
Antibiotics are categorized based on their chemical structure and their specific mechanism for destroying bacteria:
| Category | Mechanism of Action | Common Examples |
| Beta-Lactams | Inhibits the formation of the bacterial cell wall, causing it to burst. | Amoxicillin, Ceftriaxone, Meropenem. |
| Macrolides | Stops bacteria from growing by interfering with protein synthesis. | Azithromycin, Erythromycin, Clarithromycin. |
| Fluoroquinolones | Directly blocks the bacteria’s ability to replicate its DNA. | Ciprofloxacin, Levofloxacin. |
| Tetracyclines | Broad-spectrum agents that inhibit protein production. | Doxycycline, Minocycline. |
| Aminoglycosides | Potent bactericidal agents often used in injectable formats. | Gentamicin, Amikacin. |
2. The 2026 Shift: Combatting Resistance
In 2026, the focus of antibiotic manufacturing in India has pivoted toward “Access, Watch, and Reserve” (AWaRe) categories defined by the WHO:
Novel Beta-Lactamase Inhibitors: Manufacturers are now producing advanced combinations (e.g., Ceftazidime-Avibactam) to treat “Superbugs” that have become resistant to standard carbapenems.
Narrow-Spectrum Focus: There is a strategic move toward “targeted” antibiotics that kill specific bacteria without destroying the patient’s healthy gut microbiome.
Stewardship Integration: 2026-era packaging in India now often includes “Smart QR” codes that provide doctors and patients with instant guidelines on correct dosage to prevent the further spread of resistance.
3. Significance of Antibiotic Manufacturing in India
As the “Pharmacy of the World,” India’s role in the global antibiotic supply chain is more significant than ever in 2026:
Vertical Integration: Many Indian firms have achieved Backward Integration, meaning they produce the Key Starting Materials (KSMs) and APIs (Active Pharmaceutical Ingredients) in-house, reducing global dependence on single-source supply chains.
Sterile Block Mastery: India leads in the production of sterile Cephalosporin and Penicillin injectables, using dedicated, isolated manufacturing blocks to ensure zero cross-contamination.
Global Tender Leadership: Indian facilities hold the highest number of WHO-Prequalifications, making them the primary suppliers for global health organizations like UNICEF and PAHO.
Pediatric Innovation: India is the top producer of Dispersible Antibiotic Tablets and taste-masked suspensions, ensuring that life-saving treatments are accessible and easy to administer to children worldwide.
4. Why Healthy Inc. is Your Strategic Sourcing Partner
Navigating the antibiotic market requires a partner who understands the technicality of stability and the urgency of supply. Healthy Inc. acts as your technical bridge:
Strategic Sourcing Hub: We are associated with multiple state-of-the-art, WHO-GMP compliant manufacturing units. Whether you need high-volume Amoxicillin capsules, sterile Ceftriaxone injections, or advanced carbapenems, we match you with the right facility.
Pharmacist-Led Technical Vetting: We provide “straight answers” on Dissolution and Purity profiles. Our team vets every batch to ensure the drug remains potent and effective against target pathogens.
Regulatory & Dossier Mastery: We simplify international registration by providing full CTD/ACTD dossiers, stability data for all climate zones, and bioequivalence reports required for global tenders.
Reliable Supply Chain: We understand that antibiotics are time-sensitive. Through our associated network, we ensure a consistent supply of world-class medications, backed by transparent technical documentation.
Showing all 179 results
Amikacin Sulphate Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Liquid in Glass Vials or Ampoules. The solution is clear, colorless to pale yellow, and requires no reconstitution (ready-to-use).
ant Active Ingredient Strength (Standard) Therapeutic Role Amikacin Sulphate USP/BP 100 mg / 2 ml Pediatric Dose Amikacin Sulphate USP/BP 250 mg / 2 ml Intermediate Dose Amikacin Sulphate USP/BP 500 mg / 2 ml Adult Standard Dose Excipients Q.S. Sodium Citrate / Sodium Metabisulfite Buffer / Antioxid *Pack Sizes: Tray of 10 Ampoules or Box of 1/10 Vials.
Amoxicillin & Potassium Clavulanate Dispersible Tablets
FreeProduct Composition & Strength
We supply this product in the globally standardized 7:1 ratio, packed in high-barrier Alu-Alu blister strips to ensure the stability of the moisture-sensitive Clavulanate.
Active Ingredient Strength Primary Clinical Function Amoxicillin (as Trihydrate) USP/BP 200 mg / 400 mg The Growth Inhibitor: Destroys the bacterial cell wall across a broad range of pathogens. Potassium Clavulanate USP/BP 28.5 mg / 57 mg The Resistance Shield: Neutralizes bacterial enzymes, allowing Amoxicillin to function. Excipients Sweetened Matrix Pediatric-Friendly System: Engineered with pleasant fruit flavors to ensure 100% child compliance. Amoxicillin Capsules
FreeAmoxicillin capsules are common penicillin-type antibiotics typically available in 250 mg and 500 mg strengths.
Amoxicillin Sodium Injection 250mg
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. It must be reconstituted with Sterile Water for Injection prior to use.
Active Ingredient Strength (Pediatric Dose) Therapeutic Role Amoxicillin Sodium USP/BP Equivalent to 250 mg Amoxicillin Broad Spectrum Antibiotic Excipients None (Pure Sterile Powder) – – *Pack Sizes: Tray of 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Amoxicillin Sodium Injection 500mg
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. It must be reconstituted with Sterile Water for Injection prior to use.
Active Ingredient Strength (Standard Adult Dose) Therapeutic Role Amoxicillin Sodium USP/BP Equivalent to 500 mg Amoxicillin Broad Spectrum Antibiotic Excipients None (Pure Sterile Powder) – – *Pack Sizes: Tray of 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Amoxicillin Trihydrate Dispersible Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Fruit-Flavored Tablet, packed in high-barrier Alu-Alu blister strips to ensure the stability of the moisture-sensitive API.
Active Ingredient Strength Primary Clinical Function Amoxicillin (as Trihydrate) USP/BP 125 mg The Pediatric Starter: Ideal for infants and toddlers requiring low-dose respiratory or ear infection therapy. Amoxicillin (as Trihydrate) USP/BP 250 mg The Junior Anchor: The standard dose for older children and adolescents for broad-spectrum anti-infective coverage. Excipients Sweetened Matrix Taste-Masking System: Engineered with pleasant flavors (Strawberry/Pineapple/Orange) to overcome the natural bitterness of the antibiotic. Amoxycillin , Cloxacillin And Lactic Acid Bacillus Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Film-Coated Tablet or Capsule, packed in high-barrier Alu-Alu blister strips to ensure the stability of the live probiotic spores.
Active Ingredient Strength Primary Clinical Function Amoxycillin (as Trihydrate) USP/BP 250 mg The Growth Inhibitor: Destroys the bacterial cell wall across a broad range of pathogens. Cloxacillin (as Sodium) USP/BP 250 mg The Defense Breaker: Specifically neutralizes bacterial enzymes, making the treatment effective against “Staph” infections. Lactic Acid Bacillus (LAB) 1.5 Billion Spores The Biological Shield: Replenishes healthy gut bacteria to prevent diarrhea and boost immunity. Amoxycillin & Cloxacillin Capsules
FreeAmoxycillin and cloxacillin capsules (commonly known as co-amoxiclox) are combination antibiotics used to treat infections where bacteria might produce enzymes that resist standard penicillin. They are typically available in a 500 mg total strength (250 mg / 250 mg)
Amoxycillin & Dicloxacillin Capsules
FreeAmoxycillin and dicloxacillin capsules are combination antibiotics designed to broaden the spectrum of treatment while protecting the medicine from bacterial resistance. They are typically available in a 500 mg total strength (often 250 mg / 250 mg)
Amoxycillin & Potassium Clavulanate Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The ratio is critically balanced (usually 5:1) for optimal pharmacokinetics in intravenous use.
Strength Variant Amoxicillin Content (as Sodium) Clavulanic Acid Content (as Potassium) Target Patient 1.2 g Vial 1000 mg (1 g) 200 mg Adults (Severe) 600 mg Vial 500 mg 100 mg Adults / Children 300 mg Vial 250 mg 50 mg Pediatric 150 mg Vial 125 mg 25 mg Infants *Pack Sizes: Tray of 1 Vial or Box of 1/10 Vials with Water for Injection (WFI).
Amoxycillin and Clavulanic Acid Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet, packed exclusively in highly secure, ultra-low moisture Alu-Alu blister strips (often containing integrated desiccants) to ensure the absolute stability of the incredibly hygroscopic Clavulanate salt.
Active Ingredient Strength Primary Clinical Function Amoxicillin Trihydrate USP/Ph.Eur.
Potassium Clavulanate USP/Ph.Eur.500 mg
125 mg
(Total: 625 mg)Standard Adult Dose: Therapy for moderate RTIs, dental infections, and skin infections. Amoxicillin Trihydrate USP/Ph.Eur.
Potassium Clavulanate USP/Ph.Eur.875 mg
125 mg
(Total: 1000 mg)High-Dose Adult Therapy: Aggressive intervention for severe pneumonia, severe sinus infections, and bite wounds. Excipients Microcrystalline Cellulose / Sodium Starch Glycolate / Colloidal Silicon Dioxide / Magnesium Stearate Diluent / Superdisintegrant / Moisture Scavenger (Engineered for immediate gastric dissolution) *Pack Sizes: 1×6 or 1×10 Alu-Alu Blisters (Optimized specifically for strict 5-to-7 day acute antibiotic regimens).
Amoxycillin Capsules
FreeAmoxycillin capsules are widely used penicillin-type antibiotics. They are most frequently prescribed in 250 mg and 500 mg strengths.Amoxycillin Dispersible Kid Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Fruit-Flavored Tablet, packed in high-barrier Alu-Alu blister strips to ensure the stability of the moisture-sensitive API.
Active Ingredient Strength Primary Clinical Function Amoxycillin (as Trihydrate) USP/BP 125 mg The Pediatric Starter: Ideal for infants and toddlers requiring low-dose respiratory or ear infection therapy. Amoxycillin (as Trihydrate) USP/BP 250 mg The Junior Anchor: The standard dose for older children and adolescents for broad-spectrum anti-infective coverage. Excipients Sweetened Matrix Taste-Masking System: Engineered with pleasant flavors (Strawberry/Pineapple/Orange) to overcome the natural bitterness of the antibiotic. Ampicillin & Cloxacillin Capsules
FreeAmpicillin and cloxacillin capsules (often called co-ampiclox) are combination antibiotics used to treat infections where the bacteria may produce enzymes that destroy standard penicillin. They are most commonly available in a 500 mg total strength (250 mg / 250 mg)
Ampicillin & Cloxacillin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. It must be reconstituted with Sterile Water for Injection prior to use.
Active Ingredient Strength (Standard 1:1 Ratio) Therapeutic Role Ampicillin Sodium USP/BP 250 mg Broad Spectrum Coverage Cloxacillin Sodium USP/BP 250 mg Anti-Staphylococcal Shield Total Vial Strength 500 mg Pediatric / Mild Infection (Adult Variant) 500 mg Ampicillin + 500 mg Cloxacillin (1 g Total) Standard Adult Dose Excipients None (Pure Sterile Powder) – – *Pack Sizes: Tray of 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Ampicillin & Dicloxacillin Capsules
FreeAmpicillin and dicloxacillin capsules are combination antibiotics designed to provide broad-spectrum coverage while resisting degradation by certain bacterial enzymes. They are commonly available in a 500 mg total strength (typically 250 mg / 250 mg).
Ampicillin and Flucloxacillin Capsules
FreeAmpicillin and flucloxacillin capsules (often referred to by the co-drug name co-fluampicil) are combination antibiotics used to treat infections where the specific bacteria is unknown or suspected to be a penicillin-resistant strain. They are typically available in a 250 mg/250 mg strength.
Ampicillin Capsules
FreeAmpicillin capsules are a penicillin-type antibiotic used to treat various bacterial infections. They are most commonly available in 250 mg and 500 mg strengths.
Ampicillin Injection 500mg
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. It must be reconstituted with Sterile Water for Injection prior to use.
Active Ingredient Strength (Standard Pediatric/Mild Dose) Therapeutic Role Ampicillin Sodium USP/BP Equivalent to 500 mg Ampicillin Broad Spectrum Antibiotic Excipients None (Pure Sterile Powder) – – *Pack Sizes: Tray of 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Ampicillin Sulbactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The 2:1 ratio is clinically optimized for pharmacokinetics.
Active Ingredient Strength (Standard 1.5g Vial) Strength (High Dose 3g Vial) Ampicillin Sodium USP/BP 1000 mg (1 g) 2000 mg (2 g) Sulbactam Sodium USP/BP 500 mg (0.5 g) 1000 mg (1 g) Total Vial Content 1.5 g 3.0 g Excipients None (Pure Sterile Powder) None (Pure Sterile Powder) *Pack Sizes: Tray of 10 Vials, or Box of 1 Vial with Water for Injection (WFI).
Azithromycin Dispersible Tablet
FreeAzithromycin Dispersible Tablet 100mg
Each Disersible tablet cotain:
Azithromycin Dihydrate
Eq.To Anhydrous Azithromycin(100mg)
Std:IP(ACT)
Excipients(QS)(EXC)Azithromycin Tablets IP 250 mg
Each Filmcoated tablet contains:
Azithromycin Dihydrate IP
Eq. To Anhydrous Azithromycin 250 mg
Excipients q.sAzithromycin Tablets IP 500 mg
Each Filmcoated Tablet contains:
Azithromycin Dihydrate IP
Eq. To Anhydrous Azithromycin 500 mg
Excipients q.s
Colour : Ponceau 4RAzithromycin Tablets IP 500 mg
Each Filmcoated Tablet contains:
Azithromycin Dihydrate IP
Eq. To Anhydrous Azithromycin 500 mg
Excipients q.s
Colour : Ponceau 4RAzithromycin Tablets IP 1 gm
Each Film coated tablet contains:
Azithromycin Anhydrous IP 1 gm
As Azithromycin Dihydrate IPLAMITHROCIN / Azithromycin Tablets USP 500 Mg
Each film coated tablet contains:
– Azithromycin Dihydrate
eq. to Azithromycin USP (500 mg)
– Excipients (- QS)Usage: – chest infections
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic
Azithromycin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Infusion in glass vials. It must be reconstituted and further diluted before administration.
Active Ingredient Strength (Standard Adult Dose) Therapeutic Role Azithromycin (as Dihydrate/Citrate) Equivalent to 500 mg Azithromycin Broad Spectrum Macrolide Excipients Q.S. Citric Acid / Sodium Hydroxide pH Adjuster / Solubilizer *Pack Sizes: Tray of 1 Vial, 5 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Azithromycin Tablets
FreeAzithromycin Dispersible Tablet 100mg
Each Disersible tablet cotain:
Azithromycin Dihydrate
Eq.To Anhydrous Azithromycin(100mg)
Std:IP(ACT)
Excipients(QS)(EXC)Azithromycin Tablets IP 250 mg
Each Filmcoated tablet contains:
Azithromycin Dihydrate IP
Eq. To Anhydrous Azithromycin 250 mg
Excipients q.sAzithromycin Tablets IP 500 mg
Each Filmcoated Tablet contains:
Azithromycin Dihydrate IP
Eq. To Anhydrous Azithromycin 500 mg
Excipients q.s
Colour : Ponceau 4RAzithromycin Tablets IP 500 mg
Each Filmcoated Tablet contains:
Azithromycin Dihydrate IP
Eq. To Anhydrous Azithromycin 500 mg
Excipients q.s
Colour : Ponceau 4RAzithromycin Tablets IP 1 gm
Each Film coated tablet contains:
Azithromycin Anhydrous IP 1 gm
As Azithromycin Dihydrate IPLAMITHROCIN / Azithromycin Tablets USP 500 Mg
Each film coated tablet contains:
– Azithromycin Dihydrate
eq. to Azithromycin USP (500 mg)
– Excipients (- QS)Usage: – chest infections
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic
Benzathine Penicillin Injection 1.2 MIU
FreeProduct Composition & Strength
We supply this product as a Sterile Powder for Suspension in glass vials. It requires reconstitution with Sterile Water for Injection to form a milky white suspension.
Active Ingredient Strength (International Units) Therapeutic Role Benzathine Penicillin G USP/BP 1,200,000 IU (1.2 MIU) Standard Adult Prophylaxis Excipients Q.S. Lecithin / Sodium Citrate Suspending / Buffering Agents *Pack Sizes: Tray of 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Benzathine Penicillin Injection 2.4 MIU
FreeProduct Composition & Strength
We supply this product as a Sterile Powder for Suspension in glass vials. It requires reconstitution with Sterile Water for Injection to form a milky white suspension.
Active Ingredient Strength (International Units) Therapeutic Role Benzathine Penicillin G 1,200,000 IU (1.2 MIU) Pediatric / Standard Dose Benzathine Penicillin G 2,400,000 IU (2.4 MIU) Adult Syphilis / High Dose Benzathine Penicillin G 600,000 IU (0.6 MIU) Pediatric Prophylaxis Excipients Q.S. Lecithin / Sodium Citrate Suspending / Buffering Agents *Pack Sizes: Tray of 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Benzyl Penicillin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Buffered Powder for Injection in glass vials. The potency is measured in International Units (IU).
Active Ingredient Strength (IU) Mass Equivalent Therapeutic Role Benzyl Penicillin Sodium 1,000,000 IU (1 MU) ~600 mg Standard Dose Benzyl Penicillin Sodium 5,000,000 IU (5 MU) ~3000 mg High Dose (Meningitis) Benzyl Penicillin Sodium 10,000,000 IU (10 MU) ~6000 mg Severe Sepsis Buffering Agent Sodium Citrate – pH Stabilizer *Pack Sizes: Tray of 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefadroxil 500 mg Tablets
FreeA Cefadroxil 500 mg tablet (or capsule) is a first-generation cephalosporin antibiotic used to treat various bacterial infections, including those of the throat, urinary tract, and skin.
Cefepime & Sulbactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 2:1.
Strength Variant Cefepime Content Sulbactam Content Ratio 1.5 g Vial 1000 mg (1 g) 500 mg (Sodium) 2:1 (Standard) Excipients L-Arginine Buffer / Stabilizer – *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefepime & Tazobactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 8:1.
Strength Variant Cefepime Content Tazobactam Content Ratio 1.125 g Vial 1000 mg (1 g) 125 mg (Sodium) 8:1 (Standard) Excipients L-Arginine Buffer / Stabilizer – *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefepime Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. It contains L-Arginine as a buffering agent (instead of sodium salts).
Active Ingredient Strength Therapeutic Role Cefepime Hydrochloride USP/BP Equivalent to 1000 mg (1 g) Cefepime Severe Infection / Sepsis Cefepime Hydrochloride USP/BP Equivalent to 500 mg Cefepime Pediatric / Moderate Infection Buffer L-Arginine pH Stabilizer (Approx 725 mg per g) *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefixime Capsules 400mg
FreeCefixime 400mg capsules are third-generation cephalosporin antibiotics used for a variety of bacterial infections. While 200mg is a common dose, the 400mg strength is often used for once-daily dosing.
Cefoperazone & Sulbactam 2Gm Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in 20ml glass vials.
Active Ingredient Strength Function Cefoperazone Sodium USP/BP 1000 mg (1 g) Anti-Pseudomonal Cephalosporin Sulbactam Sodium USP/BP 1000 mg (1 g) Beta-Lactamase Inhibitor Ratio 1 : 1 High Sulbactam Load *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with 10ml Water for Injection (WFI).
Cefoperazone & Sulbactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 1:1.
Strength Variant Cefoperazone Content Sulbactam Content Ratio 1.5 g Vial 500 mg (Sodium) 500 mg (Sodium) 1:1 (Standard) 3.0 g Vial 1000 mg (1 g) 1000 mg (1 g) 1:1 (High Dose) Excipients None (Sterile Blend) – – *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefoperazone & Tazobactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 8:1.
Strength Variant Cefoperazone Content Tazobactam Content Ratio 1.125 g Vial 1000 mg (1 g) 125 mg (Sodium) 8:1 (Standard) Excipients None (Sterile Blend) – – *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
1.125 g Vial 1000 mg (1 g) 125 mg (Sodium) 8:1 (Standard) Excipients None (Sterile Blend) – – Cefoperazone Sodium Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. It contains approximately 34 mg (1.5 mEq) of sodium per gram.
Active Ingredient Strength (Standard) Therapeutic Role Cefoperazone Sodium USP/BP Equivalent to 1000 mg (1 g) Cefoperazone Adult Surgical / Biliary Dose Excipients None (Pure Sterile Powder) – *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefotaxime Sodium Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The sodium content is lower than many other cephalosporins (approx 2.2 mmol/g).
Active Ingredient Strength Primary Patient Cefotaxime Sodium USP/BP Equivalent to 1000 mg (1 g) Adults (Meningitis/Sepsis) Cefotaxime Sodium USP/BP Equivalent to 500 mg Pediatrics Cefotaxime Sodium USP/BP Equivalent to 250 mg Neonates / Infants Excipients None (Pure Sterile Powder) – *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefpirome & Sulbactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 2:1.
Strength Variant Cefpirome Content (as Sulfate) Sulbactam Content (as Sodium) Ratio 1.5 g Vial 1000 mg (1 g) 500 mg 2:1 (Standard) Excipients Sodium Carbonate Buffer – *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefpirome Sulfate Injection
FreeCefpirome sulfate injection is a fourth-generation cephalosporin antibiotic typically supplied as a sterile dry powder for intravenous (IV) or intramuscular (IM) administration
Ceftazidime & Sulbactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is either 2:1 or 1:1 depending on the market requirement.
Strength Variant Ceftazidime Content Sulbactam Content Ratio 1.5 g Vial 1000 mg (1 g) 500 mg (Sodium) 2:1 (Standard) 2.25 g Vial 1125 mg 1125 mg 1:1 (High Potency) Excipients Sodium Carbonate Buffer – *Pack Sizes: Tray of 1 Vial or Box of 1/10 Vials with Water for Injection (WFI).
Ceftazidime & Tazobactam Injection
FreeCeftazidime and tazobactam injection is a combination antibiotic used primarily in hospital settings for serious, multi-drug resistant bacterial infections. It is typically available as a sterile dry powder for reconstitution.
Product Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 8:1.
Strength Variant Ceftazidime Content Tazobactam Content Target Patient 1.125 g Vial 1000 mg (1 g) 125 mg (Sodium) Standard Adult Dose 281.25 mg Vial 250 mg 31.25 mg Pediatric Dose Excipients Sodium Carbonate Buffer Solubilizer Ceftazidime Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. It contains Sodium Carbonate as a solubilizer.
Active Ingredient Strength Primary Patient Ceftazidime Pentahydrate USP/BP Equivalent to 1000 mg (1 g) Adults (Standard Dose) Ceftazidime Pentahydrate USP/BP Equivalent to 250 mg Pediatrics / Neonates Excipients Sodium Carbonate Gas-Releasing Buffer *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Ceftriaxone & Sulbactam injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 2:1.
Strength Variant Ceftriaxone Content Sulbactam Content Target Patient 1.5 g Vial 1000 mg (1 g) 500 mg (Sodium) Adult Standard Dose 750 mg Vial 500 mg 250 mg Pediatric / Moderate Dose 375 mg Vial 250 mg 125 mg Infant / Neonate Dose Ratio 2 : 1 – Optimal Synergy *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Ceftriaxone & Tazobactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 8:1.
Strength Variant Ceftriaxone Content Tazobactam Content Ratio 1.125 g Vial 1000 mg (1 g) 125 mg (Sodium) 8:1 (Standard) 562.5 mg Vial 500 mg 62.5 mg Pediatric Excipients None (Sterile Blend) – – *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Ceftriaxone 1gm Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials.
Active Ingredient Strength Standard Use Ceftriaxone Sodium USP/BP Equivalent to 1000 mg (1 g) Ceftriaxone Adult Standard Dose Sodium Content Approx 83 mg (3.6 mEq) Per 1g Vial *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Ceftriaxone 500mg Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials.
Active Ingredient Strength Target Use Ceftriaxone Sodium USP/BP Equivalent to 500 mg Ceftriaxone Pediatrics / IM Gonorrhea Sodium Content Approx 41.5 mg (1.8 mEq) Low Sodium Load *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefuroxime & Sulbactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. The standard ratio is 2:1.
Strength Variant Cefuroxime Content Sulbactam Content Primary Use 1.5 g Vial 1000 mg (1 g) 500 mg (Sodium) Adult Therapy / Surgery 1.125 g Vial 750 mg 375 mg Moderate Infection 375 mg Vial 250 mg 125 mg Pediatrics *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cefuroxime Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials.
Active Ingredient Strength Primary Use Cefuroxime Sodium USP/BP Equivalent to 750 mg Cefuroxime Standard Adult Dose Cefuroxime Sodium USP/BP Equivalent to 1500 mg (1.5 g) Severe Infections / Surgery Excipients None (Pure Sterile Powder) – *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Cephalexin Capsules
FreeCephalexin capsules are a first-generation cephalosporin antibiotic used to treat various bacterial infections. They are typically available in strengths of 250 mg, 500 mg, and sometimes 333 mg or 750 mg.
Cephalothin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials. It contains approximately 2.8 mEq (64 mg) of Sodium per gram.
Active Ingredient Strength Therapeutic Role Cephalothin Sodium USP/BP Equivalent to 1000 mg (1 g) Cephalothin Surgical Prophylaxis / Skin Infection Excipients Sodium Bicarbonate Buffer / pH Adjuster *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Chloramphenicol Sodium Succinate Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in glass vials. It must be reconstituted before use.
Active Ingredient Strength Therapeutic Role Chloramphenicol Sodium Succinate USP/BP Equivalent to 1000 mg (1 g) Chloramphenicol Broad Spectrum Antibiotic Excipients None (Pure Sterile Powder) – *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Ciprofloxacin Tablets
FreeCiprofloxacin Dispersible Tablets
Each Uncoated Dispersible tablet contains:
Ciprofloxacin Hydrochloride IP
Eq. Ciprofloxacin 100 mg
Approved colour & FlavourCiprofloxacin Tablets IP 250 mg
Each Film coated tablet contains:
Ciprofloxacin Hydrochloride IP
Eq.To Ciprofloxacin Anhydrous 250 mg
Color : Titanium Dioxide IPCiprofloxacin Tablets IP 500 mg
Each Filmcoated tablet contains :
Ciprofloxacin Hydrochloride IP
Eq.To Ciprofloxacin Anhydrous 500 mg
Excipients q.sCiproctan – 250 Tablets
Ciprofloxacin Tablets BP 250 mg
Each Filmcoated tablet contains :
Ciprofloxacin Hydrochloride BP
Eq.To Ciprofloxacin Anhydrous 250 mgCiproctan
Ciprofloxacin Tablets BP 500 mg
Each Filmcoated tablet contains :
Ciprofloxacin Hydrochloride BP
Eq.To Ciprofloxacin Anhydrous 500 mgUsage: – Treat or prevent certain infections caused by bacteria such as pneumonia
Category: – Antibiotic Drugs
Therapeutic category: – Antibiotic
Ciprofloxacin Tinidazole Tablets
FreeOctum – TZ
Each Filmcoated tablet contains :
Ciprofloxacin HCl BP
Eq.To Ciprofloxacin Anhydrous 500 mg
Tinidazole BP 600 mgUsage: – Bacterial & Parasitic infections
Category: – Antibiotic Drugs
Therapeutic category: – Antibiotic
Clarithromycin Tablets
FreeClarithromycin Tablets USP 500 mg
Each Filmcoated tablet contains :
Clarithromycin USP 500 mgUsage: – Chest infections
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic
Clindamycin Capsules
FreeClindamycin capsules are lincosamide antibiotics primarily used to treat serious bacterial infections, including those of the skin, lungs, and internal organs. They are most commonly formulated as Clindamycin Hydrochloride.
Cloxacillin Capsules
FreeCloxacillin capsules are penicillin-type antibiotics used specifically to treat infections caused by staphylococci (staph infections) that produce an enzyme called penicillinase.
Cloxacillin 1gm Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials.
Active Ingredient Strength Primary Use Cloxacillin Sodium USP/BP Equivalent to 1000 mg (1 g) Cloxacillin Severe Infection / Surgery Sodium Content Approx 2.3 mEq (53 mg) per gram Monitor in Heart Failure *Pack Sizes: Tray of 1 Vial, 10 Vials, 50 Vials, or Box of 1/10 Vials with Water for Injection (WFI).
Co -Trimoxazole Tablets Tablet
FreeCo- Trimoxazole Tablets IP
Trimethoprime & Sulphamethoxazole Tablets IP
Each Uncoated tablet contains :
Trimethoprime IP 20 mg
Sulphamethoxazole IP 100 mgTrimethoprim & sulphamethoxazole tablets IP Co -trimoxazole Tablets IP
Each film coated tablets : Excipients q.s
Trimethoprim IP 80 mg
Sulphamethoxazole IP 400 mgTrimethoprim & sulphamethoxazole tablets IP
Each film coated tablets :
Trimethoprim IP 160 mg
Sulphamethoxazole IP 800 mgCo – Trimoxazole Tablets BP
Trimethoprim & Sulphamethoxazole Tablets BP
Each Uncoated Tablet contains :
Trimethoprim BP 80 mg
Sulfamethoxazole BP 400 mgTACOT – DS
Trimethoprim & Sulphamethoxazole Tablets BP
Each Uncoated tablet contains :
Trimethoprim BP 160 mg
Sulphamethoxazole BP 800 mgUsage: – Traveler’s diarrhea
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic
Doripenem Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in glass vials.
Active Ingredient Strength Primary Use Doripenem Monohydrate USP/BP Equivalent to 500 mg Doripenem Standard Adult Dose Doripenem Monohydrate USP/BP Equivalent to 250 mg Renal Impairment Excipients None (Pure Sterile Powder) – *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials.
Doxycycline 100mg Capsules
FreeDoxycycline 100mg capsules are broad-spectrum tetracycline antibiotics used for bacterial infections, malaria prophylaxis, and severe acne.
Doxycycline Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in amber glass vials.
Active Ingredient Strength Primary Use Doxycycline Hyclate USP/BP Equivalent to 100 mg Doxycycline Standard Adult Dose Excipients Ascorbic Acid / Mannitol Stabilizer / Bulking Agent *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials.
Doxycycline Tablets
FreeDoxycycline Tablets 100 mg
Each Film Coated Tablet Contains:
Doxycycline Hydrochloride IP
eq. to Doxycycline 100 mgDispersible Doxycycline Tablets BP 100 mg
Each Uncoated Tablet Contains:
Doxycycline Monohydrate BP
eq. To Anhydrous Doxycycline 100 mgUsage: – Infections
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic
Erythromycin Capsules
FreeErythromycin capsules are macrolide antibiotics used to treat various bacterial infections. Because erythromycin base is easily destroyed by stomach acid, these capsules are typically formulated as delayed-release systems containing enteric-coated pellets
Erythromycin Tablets
FreeErythromycin Stearate Tablets IP 250 mg
Each Film coated tablet contains:
Erythromycin Stearate IP
Eq. To Erythromycin 250 mg
Color: ErythrosineErythromycin Stearate Tablets IP 500 mg
Each Film coated tablet contains:
Erythromycin Stearate IP
Eq. To Erythromycin 500 mg
Excipients q.sElthro – 250
Erythromycin Stearate Tablets IP 250 mg
Each Filmcoated tablet contains:
Erythromycin Stearate IP
Eq. To Erythromycin 250 mgERYTHROMYCIN STEARATE TABLETS IP 500 MG
Each Film Coated Tablet Contains:
Erythromycin Stearate IP (- )
Eq. to Erythromycin (500 mg)
Excipients (- QS)Eritro C
(Erythromycin Stearate Tablets BP)
Each Filmcoated tablet contains :
Erythromycin Stearate BP
Eq. To Erythromycin 500 mgYALLETHRO BP 500 (ERYTHROMYCIN STEAATE TABLETS BP 500 MG) Each film coated tablets contains: Erythromycin stearate eq. To Erythromycin BP 500 mg.
Usage: – Chest infections
Category: – Antibiotic Drugs
Therapeutic category: – Antibiotic
Ethambutol Tablets
FreeEthambutol Hydrochloride Tablets IP 200 mg
Each Film Coated Tablet contains:
Ethambutol Hydrochloride IP 200 mgEthambutol tablets IP 400 mg
Each film coated tablet contains :
Ethambutol hydrochloride IP 400 mgEthambutol Hydrochloride Tablets IP 800 mg
Each Film Coated Tablet contains:
Ethambutol Hydrochloride IP 800 mgEthambutol Tablets BP 200 mg
Each Film Coated Tablet Contains :
Ethambutol Hydrochloride BP 200 mgEthambutol Tablets BP 400 mg
Each Film Coated Tablet Contains :
Ethambutol Hydrochloride BP 400 mgEthambutol Tablets BP 800 mg
Each Film Coated Tablet Contains :
Ethambutol Hydrochloride BP 800 mgUsage: – Treat Tuberculosis
Category: – Antibiotic Drugs
Therapeutic category: – Antibiotic, Anti tuberculosis
Ethambutol Tablets
FreeEthambutol Hydrochloride Tablets IP 200 mg
Each Film Coated Tablet contains:
Ethambutol Hydrochloride IP 200 mgEthambutol tablets IP 400 mg
Each film coated tablet contains :
Ethambutol hydrochloride IP 400 mgEthambutol Hydrochloride Tablets IP 800 mg
Each Film Coated Tablet contains:
Ethambutol Hydrochloride IP 800 mgEthambutol Tablets BP 200 mg
Each Film Coated Tablet Contains :
Ethambutol Hydrochloride BP 200 mgEthambutol Tablets BP 400 mg
Each Film Coated Tablet Contains :
Ethambutol Hydrochloride BP 400 mgEthambutol Tablets BP 800 mg
Each Film Coated Tablet Contains :
Ethambutol Hydrochloride BP 800 mgUsage: – Treat Tuberculosis
Category: – Antibiotic Drugs
Therapeutic category: – Antibiotic, Anti tuberculosis
Ethionamide Tablets
FreeEthionamide Tablets IP 125 mg
Each Film Coated Tablet contains:
Ethionamide IP 125 mgEthionamide Tablets IP 250 mg
Each Film Coated Tablet contains:
Ethionamide IP 250 mgETHIONAMIDE TABLETS 250 MG
Each filmcoated tablet contains:
Ethionamide BP 250 mgUsage: – Treat Tuberculosis
Category: – Antibiotic Drugs
Therapeutic category: – Antibiotic, Ant tuberculosis
Flucloxacillin and Amoxicillin Capsules
FreeFlucloxacillin and amoxicillin combination capsules (often referred to by trade names like Flamox (cosmos-pharm.com) or Amoxam) contain two distinct penicillin-type antibiotics (hra.nhs.uk) that work together to treat a broader range of bacterial infections.
Flucloxacillin Capsules
FreeFlucloxacillin capsules contain flucloxacillin sodium as the active ingredient, along with specific excipients that vary slightly by manufacturer but typically include a lubricant, a glidant, and sodium.
Flucloxacillin Injection
FreeFlucloxacillin injection is a narrow-spectrum penicillin antibiotic used to treat infections caused by staphylococci. It is typically supplied as a white to off-white sterile powder for reconstitution.
Fortified Procaine Peniciilin Injection with Streptomycin
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials.
Active Ingredient Strength Role Procaine Penicillin G 300,000 IU Sustained Release (24 Hours) Benzyl Penicillin Sodium 100,000 IU Rapid Onset (Peak Levels) Streptomycin Sulfate Equivalent to 0.5 g (500 mg) Base Gram-Negative Coverage Total Formulation 400,000 IU + 0.5 g Standard Adult Dose *Pack Sizes: Tray of 50 Vials or 100 Vials.
Fortified Procaine Penicillin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials.
Active Ingredient Proportion Role Procaine Penicillin G 300,000 IU (75%) Long Acting (Depot) Benzyl Penicillin Sodium 100,000 IU (25%) Rapid Acting (Peak) Total Strength 400,000 IU (4 Lac) Standard Adult Dose Total Strength 4,000,000 IU (40 Lac) High Dose / Veterinary *Pack Sizes: Tray of 50 Vials or 100 Vials.
Gentamicin Injection
FreeGentamicin injection (brand name Garamycin) is a sterile, clear, colourless to slightly yellow aqueous solution of an aminoglycoside antibiotic. It is used to treat serious gram-negative bacterial infections.
Imipenem & Cilastatin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Buffered Lyophilized Powder for Injection in glass vials.
Active Ingredient Strength Role Imipenem USP/BP 500 mg Antibiotic (Carbapenem) Cilastatin Sodium USP/BP Equivalent to 500 mg Cilastatin DHP-I Inhibitor Buffer Sodium Bicarbonate pH Stabilizer *Pack Sizes: Tray of 1 Vial, 10 Vials, or Box of 1/10 Vials.
Ivermectin Tablets
FreeIvermectin Tablets 3 mg
Each tablet contains :
Ivermectin BP 3 mgIvermectin Tablets 6 mg
Each Uncoated tablet contains :
Ivermectin IP 6 mgUsage: – Parasitic infections
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic, Antiparasitic
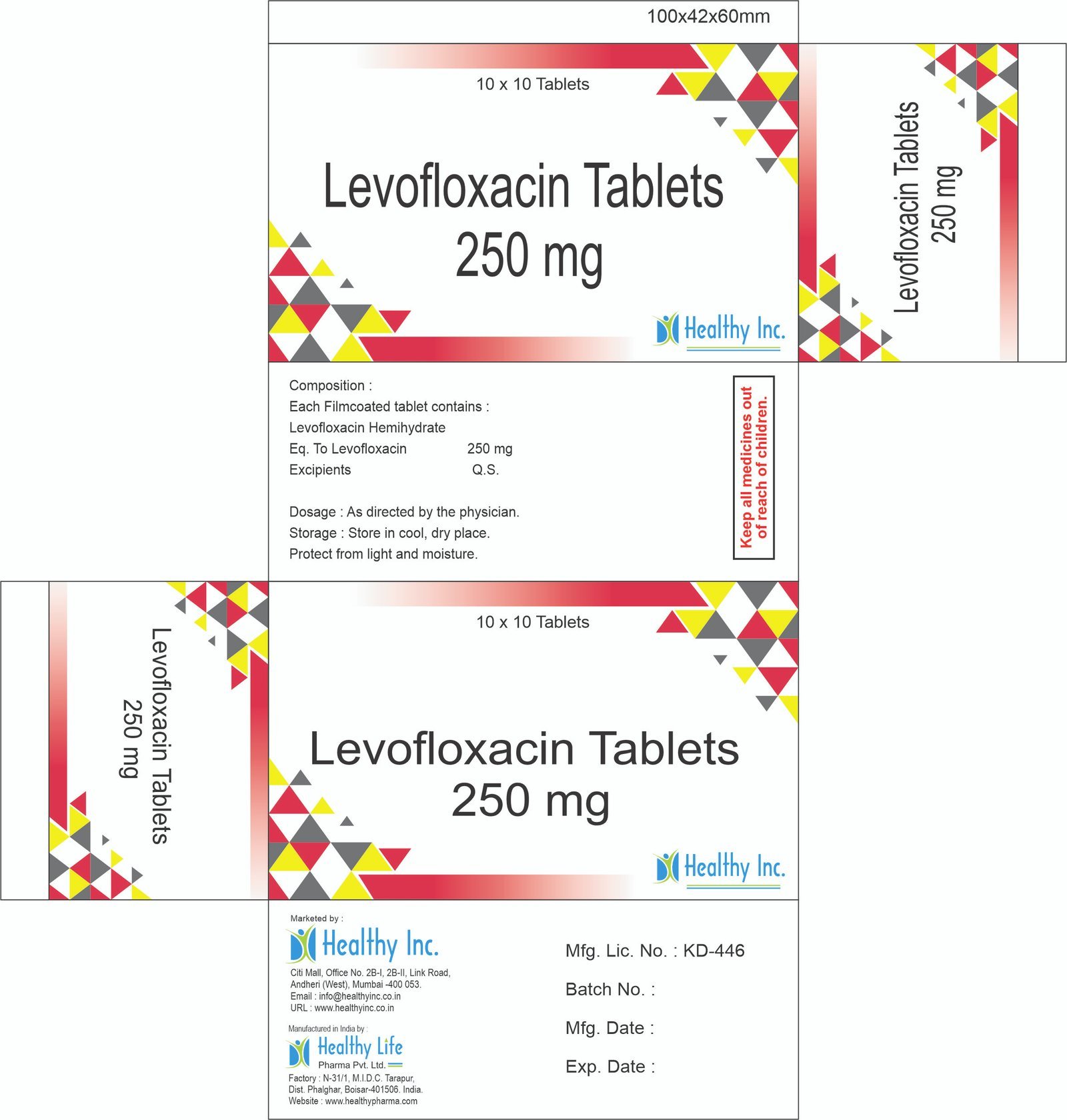
Levofloxacin Tablets
FreeLevofloxacin tablets IP 250 mg
Each Filmcoated tablet contains :
Levofloxacin Hemihydrate IP
Eq. To Levofloxacin 250 mgLevofloxacin tablets IP 500 mg
Each Filmcoated tablet contains :
Levofloxacin Hemihydrate IP
Eq. To Levofloxacin 500 mgLevofloxacin Tablets 750 mg
Each Film Coated Tablet Contains :
Levofloxacin Hemihydrate
Eq To Levofloxacin 750 mgLevofloxacin tablets 250 mg
Each Filmcoated tablet contains :
Levofloxacin Hemihydrate
Eq. To Levofloxacin 250 mgLevofloxacin tablets 500 mg
Each Filmcoated tablet contains :
Levofloxacin Hemihydrate
Eq. To Levofloxacin 500 mgUsage: – Variety of bacterial infections
Category: – Antibiotic Drugs
Therapeutic category: – Antibiotic
Lincomycin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Solution in Glass Ampoules or Vials.
Active Ingredient Strength Volume Lincomycin Hydrochloride USP/BP 600 mg 2 ml Ampoule (300 mg/ml) – Standard Lincomycin Hydrochloride USP/BP 300 mg 1 ml Ampoule (300 mg/ml) – Pediatric Preservative Benzyl Alcohol 0.945% v/v *Pack Sizes: Tray of 10 Ampoules, 50 Ampoules, or 100 Ampoules.
Linezolide Tablets
FreeLinezolide Tablets
Each Film Coated Tablet Contains
Linezolide 600 mgUsage: – Treat different types of bacterial infections
Category: – Antibiotic drugs
Therapeutic category: – Antibiotics
Meropenem & Sulbactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder for Injection in glass vials.
Active Ingredient Strength Role Meropenem USP/BP 1000 mg (1 g) Broad-Spectrum Carbapenem Sulbactam Sodium USP/BP Equivalent to 500 mg Beta-Lactamase Inhibitor Total Strength 1.5 g (1500 mg) Per Single Dose Vial *Pack Sizes: Single Vial with or without Sterile Water for Injection.
Meropenem Injection
FreeMeropenem injection (brand name Keppra) is a broad-spectrum carbapenem antibiotic supplied as a sterile white to pale yellow powder for reconstitution. It is used to treat severe bacterial infections
Metronidazole Capsule 500mg
FreeMetronidazole 500 mg capsules are an antibiotic and antiprotozoal medication used to treat a variety of bacterial and parasitic infections. The capsule contains a specific amount of the active drug and various inactive ingredients that help with its delivery and stability.
Metronidazole Tablets
FreeMetronidazole Tablets IP 200 mg
Each Uncoated tablet contains :
Metronidazole IP 200 mgMetronidazole Tablets IP 400 mg
Each Uncoated tablet contains :
Metronidazole IP 400 mgMetronidazole Tablets IP 200 mg
Each Filmcoated Tablets contains :
Metronidazole IP 200 mg
Excipients q.sMetronidazole Tablets IP 400 mg
Each film coated tablet contains :
Metronidazole IP 400 mg Excipients Q.S.Metro V – 250
Metronidazole Vaginal Tablets
Each Uncoated tablets
Metronidazole BP 250 mgMetronidazole tablets BP
Each Sugarcoated tablet contains :
Metronidazole BP 250 mgMetronidazole Tablets USP
Each Uncoated tablet contains :
Metronidazole USP 200 mgMetronidazole Tablets USP
Each Uncoated tablet contains :
Metronidazole USP 400 mgMetronidazole Tablets USP
Each Uncoated tablet contains :
Metronidazole USP 500 mgMetronidazole tab BP 250 mg Metronidazole BP 250 mg Titanium dioxide bp Excipent
Cirogyl – 400
Metronidazole Tablets BP 400 mg
Each Film coated Tablets contains :
Metronidazole BP 400 mgMetronidazole Tab BP 500 mg Metronidazole BP 500 mg Brilliant blue Excipents
Usage: – Skin infections, rosacea and mouth infections
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic
Metronidazole Vaginal Tablets
FreeMetronidazole Tablets IP 200 mg
Each Uncoated tablet contains :
Metronidazole IP 200 mgMetronidazole Tablets IP 400 mg
Each Uncoated tablet contains :
Metronidazole IP 400 mgMetronidazole Tablets IP 200 mg
Each Filmcoated Tablets contains :
Metronidazole IP 200 mg
Excipients q.sMetronidazole Tablets IP 400 mg
Each film coated tablet contains :
Metronidazole IP 400 mgExcipients Q.S.
Metro V – 250
Metronidazole Vaginal Tablets
Each Uncoated tablets
Metronidazole BP 250 mgMetronidazole tablets BP
Each Sugarcoated tablet contains :
Metronidazole BP 250 mgMetronidazole Tablets USP
Each Uncoated tablet contains :
Metronidazole USP 200 mgMetronidazole Tablets USP
Each Uncoated tablet contains :
Metronidazole USP 400 mgMetronidazole Tablets USP
Each Uncoated tablet contains :
Metronidazole USP 500 mgMetronidazole tab BP 250 mg
Metronidazole BP 250 mg
Titanium dioxide bp Excipent
Cirogyl – 400
Metronidazole Tablets BP 400 mg
Each Film coated Tablets contains :
Metronidazole BP 400 mgMetronidazole Tab BP 500 mg
Metronidazole BP 500 mg
Nalidixic Acid Tablets
FreeNalidixic Acid Tablets IP
Each Tablet contains :
Nalidixic Acid IP 500 mgUsage: – Treatment of bacterial infections
Category: – Antibiotic Drugs
Therapeutic category: – Antibiotic
Nitrofurantoin Tablets
FreeA Nitrofurantoin tablet is an antibiotic specifically used to treat and prevent urinary tract infections (UTIs), such as cystitis. It works by damaging the DNA of bacteria in the urinary tract.
Norfloxacin Tablets
FreeNorfloxacin Tablets IP 400 mg
Each Film Coated Tablet Contains:
Norfloxacin IP 400 mgFlonor Tablets
Norfloxacin Tablets BP 400 mg
Each Filmcoated tablet contains :
Norfloxacin BP 400 mgUsage: – Treat a variety of bacterial infections
Category: – Antibiotic Drugs
Therapeutic category: – Antibiotic
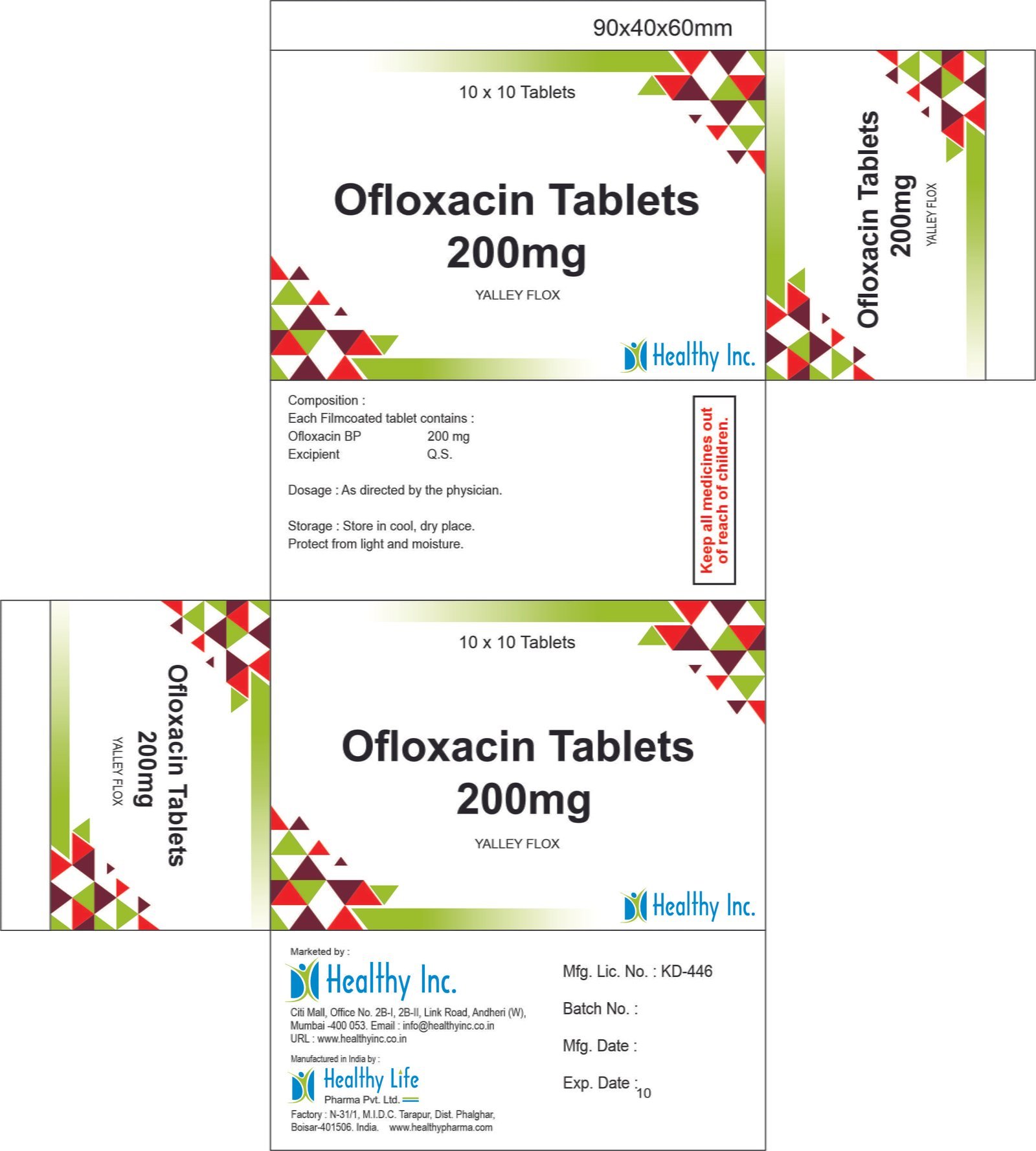
Ofloxacin Tablets
FreeOfloxacin Tablets 200 mg
Each Filmcoated tablet contains :
Ofloxacin IP 200 mg
Excipients q.sYALLEY FLOX
Ofloxacin Tablets 200mg Each Film coated tablet contains :
Ofloxacin BP 200 mg
Excipients q.sOfnis -200
Ofloxacin Tablets
Each Filmcoated tablet contains :
Ofloxacin BP 200 mgUsage: – Treat bacterial infections
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic
Oxacillin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Crystalline Powder buffered for stability in glass vials.
Active Ingredient Strength Sodium Content Oxacillin Sodium USP/BP 500 mg Approx. 1.4 mEq (32 mg) Oxacillin Sodium USP/BP 1000 mg (1 g) Approx. 2.8 mEq (64 mg) Oxacillin Sodium USP/BP 2000 mg (2 g) High-Dose Protocols Buffer Dibasic Sodium Phosphate Maintains pH upon reconstitution *Pack Sizes: Single Vial or Tray of 10/50 Vials. (Requires reconstitution with Sterile Water for Injection).
Piperacillin Tazobactam Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder buffered with Sodium Citrate/EDTA.
Strength (Ratio 8:1) Piperacillin Content Tazobactam Content 4.5 g (Standard Adult) 4000 mg (4 g) 500 mg (0.5 g) 2.25 g (Pediatric/Renal) 2000 mg (2 g) 250 mg (0.25 g) Sodium Content ~2.35 mEq per gram of Piperacillin Monitor in Heart Failure *Pack Sizes: Single Vial or Tray of 10 Vials.
Procaine Penicillin and Penicillin G Sodium Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Suspension in glass vials.
Active Ingredient Standard Dose (4 Lakh IU) Mega Dose (4 Mega IU) Procaine Penicillin G BP/USP 300,000 IU (Sustained Release) 3,000,000 IU (Sustained Release) Penicillin G Sodium BP/USP 100,000 IU (Immediate Peak) 1,000,000 IU (Immediate Peak) Excipients Lecithin / Sodium Citrate Suspending Agent / Buffer System *Pack Sizes: Single Vial or Tray of 50 Vials. (Requires reconstitution with Sterile Water for Injection to form a milky suspension).
Pyrazinamide Tablets
FreePyrazinamide Tablet IP
Each Coated Tablets Contains:
Pyrazinamide IP 500 mgPyrazinamide tablet IP 750 mg
Each Coated Tablet Contains:
Pyrazinamide IP 750 mgPYRAZINAMIDE TABLETS BP 400 MG
Each filmcoated tablet contains:
Pyrazinamide BP 400 mgUsage: – Treat tuberculosis (TB)
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic, Antituberculosis
Rifampicin 150 Isoniazid 75 pyrazinamide 400 Tablet
FreeRifampicin And Isoniazid Tablets Int Ph
Each Film Coated Tablet Contains:
Rifampicin BP/Int.Ph. 150 mg
Isoniazid BP/Int.Ph. 150 mgISORIF TABLETS
(Rifampicin And Isoniazid Tablets Int. Ph.)
Each Film Coated Tablet Contains:
Rifampicin BP/Int. Ph. 300 mg
Isoniazid BP/Int. Ph. 150 mgRifampicin And Isoniazid Tablets Int. Ph.
Each Film Coated Tablet Contains:
Rifampicin BP/Int. Ph. 150 mg
Isoniazid BP/Int. Ph. 75 mgRIFAMPICIN IP 150 MG + ISONIAZIDE IP 300 MG TABLETS Each film coated tablet contains: Rifampicin IP 150 mg isoniazide IP 300 mg Excipients Q.S.9
Usage: – treat tuberculosis (TB) infection
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic,
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Rifampicin Isoniazid pyrazinamide Tablet manufacturer supplier exporter
FreeRifampicin And Isoniazid Tablets Int Ph
Each Film Coated Tablet Contains:
Rifampicin BP/Int.Ph. 150 mg
Isoniazid BP/Int.Ph. 150 mgISORIF TABLETS
(Rifampicin And Isoniazid Tablets Int. Ph.)
Each Film Coated Tablet Contains:
Rifampicin BP/Int. Ph. 300 mg
Isoniazid BP/Int. Ph. 150 mgRifampicin And Isoniazid Tablets Int. Ph.
Each Film Coated Tablet Contains:
Rifampicin BP/Int. Ph. 150 mg
Isoniazid BP/Int. Ph. 75 mgRIFAMPICIN IP 150 MG + ISONIAZIDE IP 300 MG TABLETS
Each film coated tablet contains:
Rifampicin IP 150 mg isoniazide IP 300 mg
Excipients Q.S.
Usage: – treat tuberculosis (TB) infection
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic,
Rifampicin Isoniazide Pyrizinamide ethambutol Tablets
FreeRifampicin, Isoniazide and Pyrizinamide Tab (Combi Pack)
(Rifampicin Tablets IP) + (Isoniazide Tablets IP) + (Pyrizinamide Tablets IP)
Each film Coated Tablet Contains :
Rifampicin IP 100 mg
Each coated Tablet Contains :
Isonoazide IP 50 mg
Each coated Tablet contains :
Pyrizinamide IP 250 mgRIP TABLETS
(Rifampicin, Isoniazid And Pyrazinamide Tablets Int.Ph.)
Each Filmcoated Tablet Contains:
Rifampicin BP/Int.Ph. 150 mg
Isoniazid BP/Int.Ph. 75 mg
Pyrazinamide BP/Int.Ph. 400 mgUsage: – treat tuberculosis (TB) infection
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic, Antituberculosis
Rifampicin Isoniazide Pyrizinamide Tablets
FreeRifampicin, Isoniazide and Pyrizinamide Tab (Combi Pack)
(Rifampicin Tablets IP) + (Isoniazide Tablets IP) + (Pyrizinamide Tablets IP)
Each film Coated Tablet Contains :
Rifampicin IP 100 mg
Each coated Tablet Contains :
Isonoazide IP 50 mg
Each coated Tablet contains :
Pyrizinamide IP 250 mgRIP TABLETS
(Rifampicin, Isoniazid And Pyrazinamide Tablets Int.Ph.)
Each Filmcoated Tablet Contains:
Rifampicin BP/Int.Ph. 150 mg
Isoniazid BP/Int.Ph. 75 mg
Pyrazinamide BP/Int.Ph. 400 mgUsage: – treat tuberculosis (TB) infection
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic, Antituberculosis
Rifampicin Tablet
FreeRifampicin Tablets IP 100 mg
Each film Coated Tablet Contains:
Rifampicin IP 100 mgRifampicin Tablets Int. Ph. 300 mg
Each Film Coated Tablets Contains:
Rifampicin BP/Int.Ph. 300 mgUsage: – treat or prevent tuberculosis (TB)
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic,
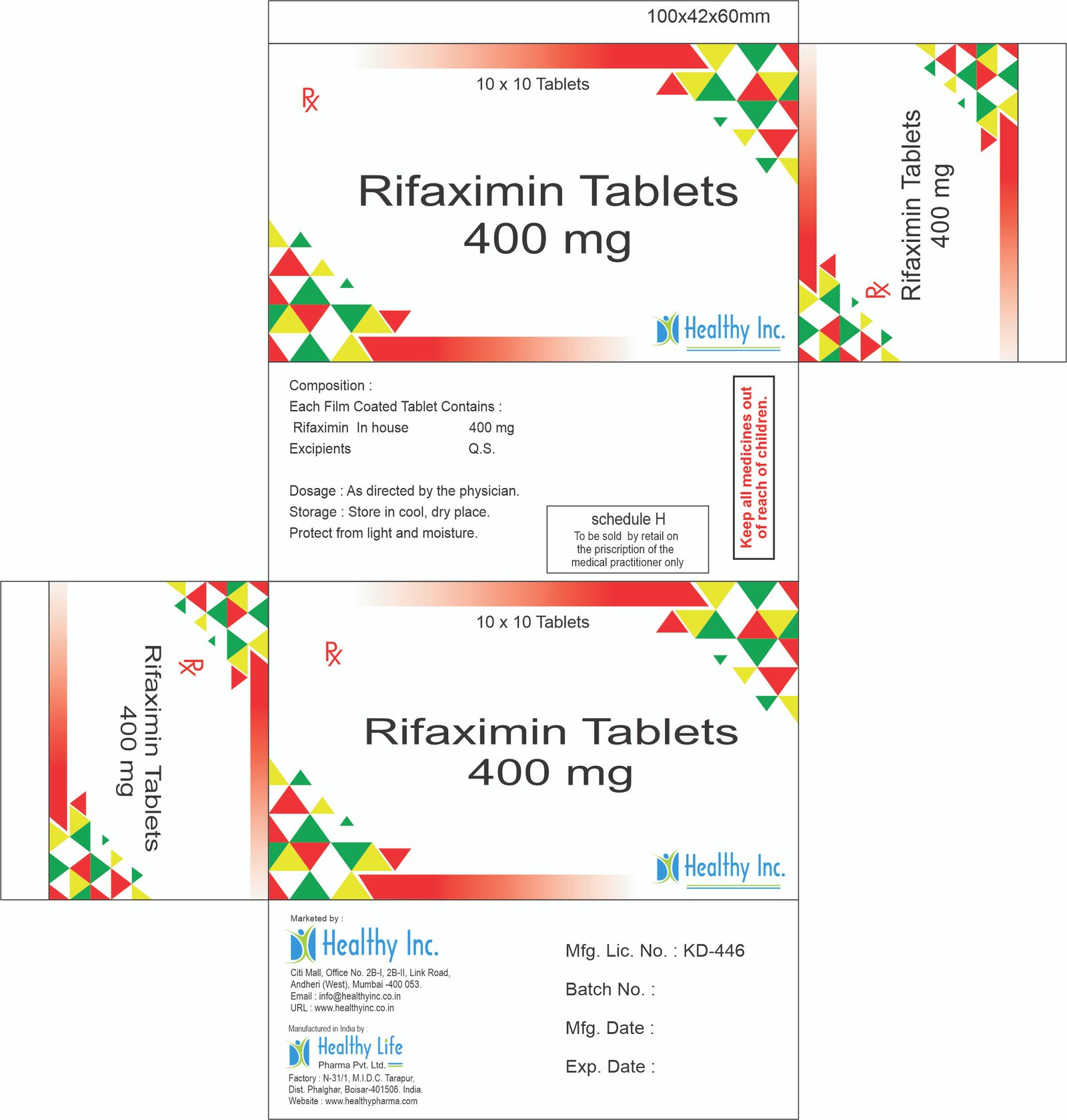
Rifaximin Tablets
FreeRifaximin Tablets 400 mg Each Film Coated Tablet Contains : Rifaximin In house 400 mg
Usage: – prevent episodes of hepatic encephalopathy
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic
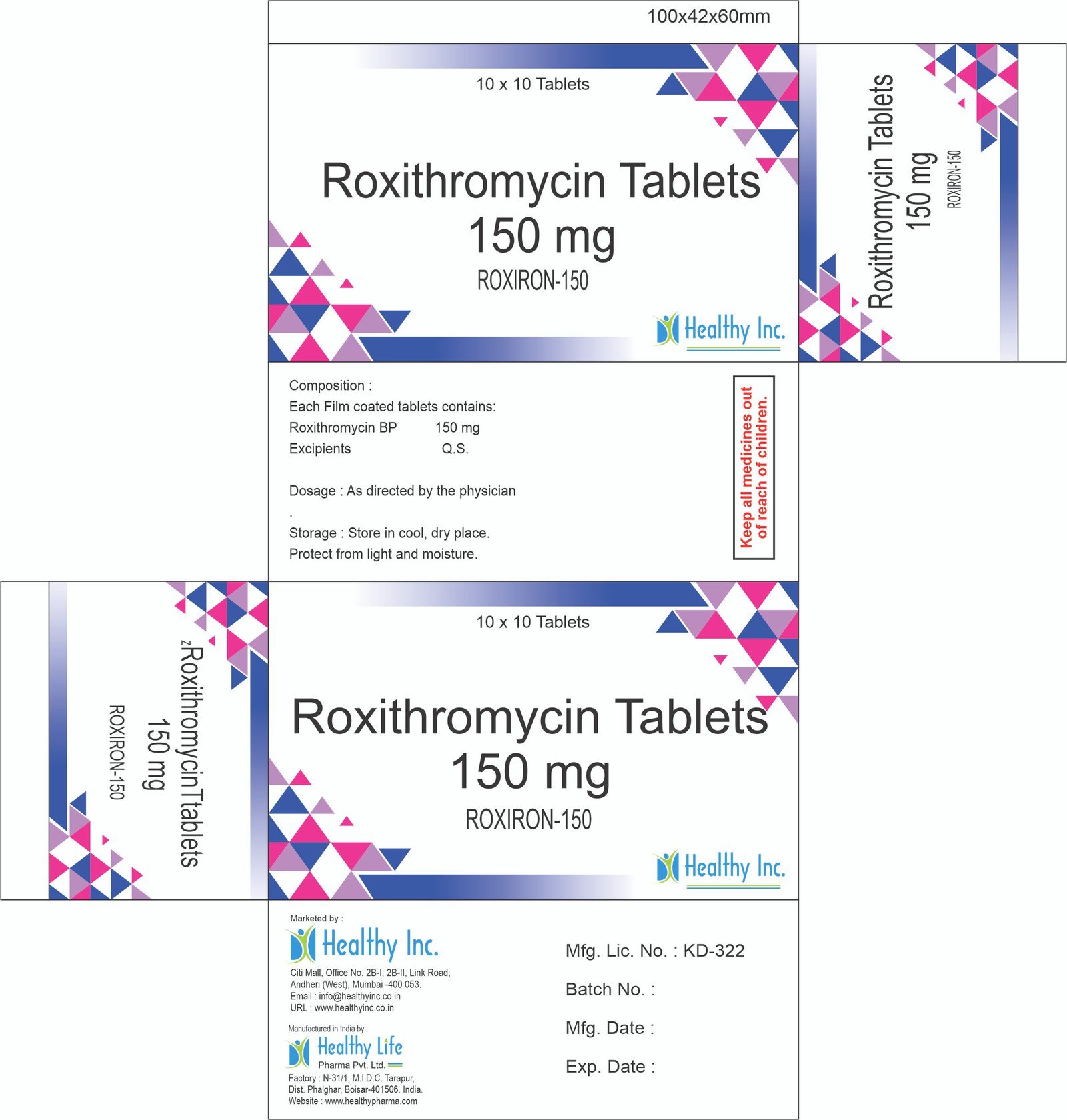
Roxithromycin Tablets
FreeRoxiron-150
(Roxithromycin tablets 150 mg)
Each Film coated tablets contains:
Roxithromycin BP 150 mgUsage: – treats bacterial infections
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic
Sodium Aminosalicylate Granule
FreeSodium Aminosalicylate Granules 80% Each Gram of Enteric Coate Granules Contains : Sodium Aminosaligylate IP (800mg) Quinoline Yellow Lake And Titanium Dioxide IP(-) Excipients(-QS)
Streptomycin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection (White to Off-White) in glass vials, requiring reconstitution with a provided sterile diluent.
Active Ingredient Base Strength Reconstitution Requirement Streptomycin Sulfate USP/BP 1000 mg (1 g) Requires 3 ml to 4.5 ml of Sterile Water for Injection or 0.9% Normal Saline Excipients None Preservative-Free Pure API *Pack Sizes: Single Vial or Tray of 50/100 Vials. (Often procured in massive bulk for government TB programs).
Teicoplanin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder (Spongy, Ivory to Pale Yellow Cake) in glass vials, accompanied by a specific sterile diluent.
Active Ingredient Strength Reconstitution Requirement Teicoplanin USP/BP/Ph.Eur. 200 mg Requires 3 ml of Sterile Water for Injection Teicoplanin USP/BP/Ph.Eur. 400 mg Requires 3 ml of Sterile Water for Injection Excipients Sodium Chloride / Sodium Hydroxide Isotonicity Agent / pH Adjuster (pH 7.2 to 7.8) *Pack Sizes: 1 Vial of Lyophilized Powder + 1 Ampoule of Diluent (3 ml) per box
Tetracycline Capsules
FreeTetracycline capsules are broad-spectrum antibiotics used to treat various bacterial infections and severe acne. They typically contain Tetracycline Hydrochloride as the active ingredient in strengths of 250 mg or 500 mg.
Tigecycline Injection
FreeTigecycline injection is a sterile, lyophilized (freeze-dried) powder containing a first-in-class glycylcycline antibiotic. It is primarily used for complicated skin and intra-abdominal infections, as well as community-acquired bacterial pneumonia.
Tinidazole Tablet
FreeTinidazole Tablets
Each Filmcoated tablet contains :
Tinidazole BP 500 mgUsage: – treat infections caused by protozoa
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic, Anthelmintic, Antiprotozoal, Antiparasitic
Tobramycin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile, Clear, Colorless to Pale Yellow Aqueous Solution in glass vials or ampoules.
Active Ingredient Concentration Total Content & Format Tobramycin (as Sulfate) USP/BP 40 mg / ml 80 mg in 2 ml Glass Vial/Ampoule Tobramycin (as Sulfate) USP/BP 10 mg / ml 20 mg in 2 ml Glass Vial (Pediatric Dose) Excipients Sodium Metabisulfite / Edetate Disodium Antioxidant / Chelating Agent (Preservative System) *Pack Sizes: Tray of 5/10 Vials or Box of 50 Ampoules.
Vancomycin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder (White to Off-White Plug/Cake) in glass vials, requiring two-step reconstitution and dilution prior to intravenous administration.
Active Ingredient Strength Initial Reconstitution Requirement Vancomycin Hydrochloride USP/BP 500 mg (0.5 g) Requires 10 ml of Sterile Water for Injection Vancomycin Hydrochloride USP/BP 1000 mg (1.0 g) Requires 20 ml of Sterile Water for Injection Further Dilution (CRITICAL) IV Infusion Matrix Must be further diluted in at least 100 ml to 200 ml of 0.9% Normal Saline or 5% Dextrose before infusing. *Pack Sizes: Single Vial or Tray of 10 Vials.