Showing 1001–1289 of 1289 results
Pramipexole Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Uncoated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the highly light-sensitive dihydrochloride monohydrate salt.
Active Ingredient Strength Primary Clinical Function Pramipexole (as Dihydrochloride Monohydrate) Ph.Eur./USP 0.125 mg Micro-Dose Standard: Mandatory initiation unit to prevent orthostatic shock, and the gold-standard maintenance dose for RLS. Pramipexole (as Dihydrochloride Monohydrate) Ph.Eur./USP 0.25 mg / 0.5 mg Titration Standard: Mid-level doses for the gradual, safe escalation of Parkinson’s therapy. Pramipexole (as Dihydrochloride Monohydrate) Ph.Eur./USP 1 mg Advanced PD Standard: High-dose maintenance therapy for severe, refractory Parkinson’s Disease. Excipients Mannitol / Maize Starch / Povidone K25 / Colloidal Silicon Dioxide / Magnesium Stearate Diluent / Binder / Glidant / Lubricant (Engineered for immediate gastric dissolution and rapid, highly predictable systemic absorption) *Pack Sizes: 10×10 Alu-Alu Blisters or 3×10 Unit-Dose Blisters (Optimized specifically for strict, high-compliance chronic neurology dispensing and complex titration regimens).
Prasugrel Tablet
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips containing dedicated desiccants to ensure the absolute chemical stability of the highly volatile thienopyridine molecule.
Active Ingredient Strength Primary Clinical Function Prasugrel HCl INN/Ph.Eur. 5 mg Dose-Adjusted Standard: Maintenance dose strictly for vulnerable patients weighing < 60 kg or aged ≥ 75 years to minimize fatal bleeding risk. Prasugrel HCl INN/Ph.Eur. 10 mg Global Clinical Standard: Standard adult maintenance dose following a 60 mg acute loading dose in the Cath Lab. Excipients Microcrystalline Cellulose / Mannitol / Croscarmellose Sodium / Hypromellose / Triacetin Diluent / Superdisintegrant / Moisture-Barrier Film-Coating (Engineered specifically to prevent hydrolytic degradation and ensure ultra-fast gastric release) *Pack Sizes: 10×10 Alu-Alu Blisters or 28/30-Tablet Calendar Blisters (Optimized specifically for strict 12-month post-PCI dual-antiplatelet dispensing regimens).
Praziquantel Tablets
FreePraziquantel Tablets USP 600 mg
Each Film Coated Tablet Contains:
Praziquantel USP 600 mgUsage: – Treat infections of certain parasites
Category: – Anthelmintic drugs
Therapeutic category: – Anthelmintic, Antiprotozoal, Antiparasitic
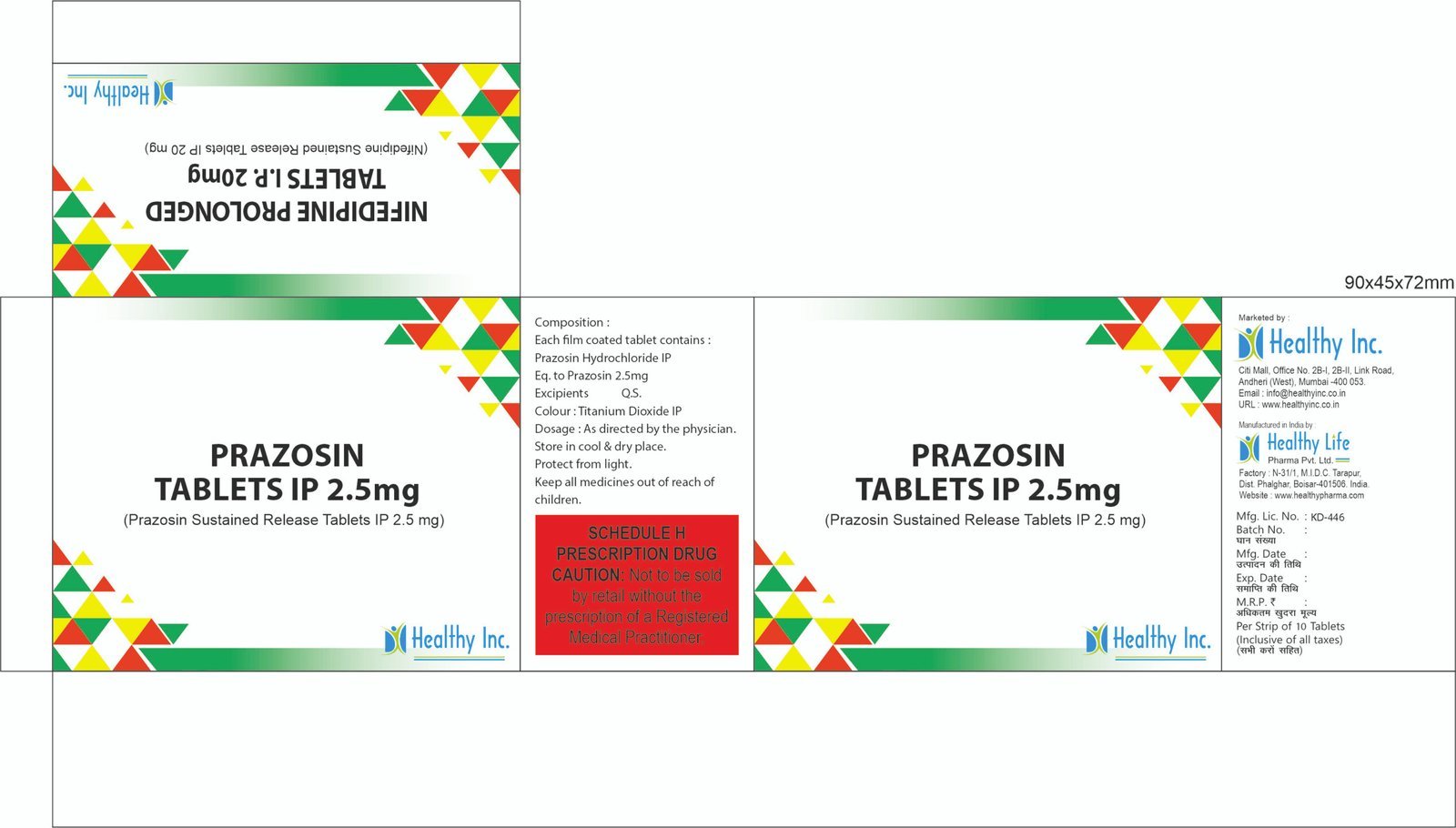
Prazosin Tablets
FreePrazosin Tablets IP 2 mg
Each filmcoated tablet contains:
Prazosin Hydrochloride IP
Eq to Prazosin 2 mgPRAZOSIN TABLETS IP 2.5 MG Each film coated Tablet Contains:
Prazosin hydrochloride IP equivalent to prazosin 2.5 mg Excipients q.s.PRAZOSIN TABLETS IP 5 MG Each film coated Tablet Contains:
Prazosin hydrochloride IP equivalent to prazosin 5 mg Excipients q.s.Usage: – Treat high blood pressure
Category: – Hypertensive / Cardiac Drugs
Therapeutic category: – Hypertensive
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Prednisolone Tablets
FreePrednisolone Tablets IP 5 mg
Each uncoated tablet contains:
Prednisolone IP 5 mgPrednisolone Tablets IP 10 mg
Each uncoated tablet contains:
Prednisolone IP 10 mgPrednisolone Tablets IP 20 mg
Each uncoated tablet contains:
Prednisolone IP 20 mg
Excipients q.sPrednisolone Tablets IP 10 mg
Each uncoated tablet contains:
Prednisolone IP 10 mgPregabalin & Methylcobalamin Capsules
FreePregabalin and methylcobalamin capsules are a common combination therapy used to manage neuropathic pain (nerve pain). They are most frequently available in strengths of 75 mg / 750 mcg or 75 mg / 1500 mcg.
Pregabalin & Methylcobalamin Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Polymeric Matrix Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the highly light-sensitive B12 coenzyme and the SR polymer network.
Active Ingredient Strength Primary Clinical Function Pregabalin INN/Ph.Eur. 75 mg (Sustained-Release) 24-hour controlled suppression of neuropathic pain and hyperalgesia. Methylcobalamin (Mecobalamin) JP/USP 1500 mcg Global Clinical Standard: High-dose active B12 for the anatomical regeneration of the myelin sheath. Excipients Hypromellose (HPMC) / Microcrystalline Cellulose / Colloidal Silicon Dioxide / Titanium Dioxide / Magnesium Stearate Hydrophilic SR Polymer Matrix / Diluent / Glidant / UV-Blocking Film-Coating (Engineered to control the exact 24-hour diffusion rate while shielding the API from light degradation) *Pack Sizes: 10×10 Alu-Alu Blisters (Optimized specifically for strict 30-day chronic diabetology and neurology dispensing regimens).
Pregabalin & Nortriptyline Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure absolute chemical stability and to mask the extremely bitter taste of the TCA component.
Active Ingredient Strength Primary Clinical Function Pregabalin INN/Ph.Eur. 50 mg / 75 mg Calcium channel neuromodulation for the direct suppression of hyperactive nerve firing. Nortriptyline Hydrochloride BP/USP 10 mg (Base Equivalent) Global Clinical Standard: SNRI/TCA action to amplify the brain’s natural descending pain-inhibiting pathways. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Povidone K30 / Magnesium Stearate / Titanium Dioxide Diluent / Superdisintegrant / Binder / Premium Film-Coating (Engineered specifically to ensure immediate, synchronized gastric dissolution while completely masking the bitter taste of Nortriptyline) *Pack Sizes: 10×10 Alu-Alu Blisters (Optimized specifically for strict 30-day chronic neurology dispensing regimens).
Pregabalin Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Hard Capsule or Extended-Release Matrix Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the active amino acid derivative and prevent illicit tampering.
Active Ingredient Strength Primary Clinical Function Pregabalin INN/Ph.Eur. 75 mg Initiation Standard: Base titration unit to safely acclimatize the brain and minimize initial dizziness. Pregabalin INN/Ph.Eur. 150 mg / 300 mg Global Clinical Standard: Advanced maintenance doses for severe diabetic neuropathy, fibromyalgia, and refractory epilepsy. Excipients (Capsules) Pregelatinized Starch / Talc / Hard Gelatin Shell Diluent / Glidant / Capsule Matrix (Engineered for immediate gastric dissolution and rapid systemic onset within 1 hour) *Pack Sizes: 10×10 Alu-Alu Blisters or 14-Capsule Titration Packs (Optimized specifically for strict chronic pain dispensing regimens and controlled-substance tracking).
Pregnyl
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder (White Plug/Cake) in glass vials, accompanied by a specific sterile diluent.
Active Ingredient Strength (International Units) Reconstitution Requirement Highly Purified hCG USP/BP 5,000 IU Requires 1 ml of 0.9% Sodium Chloride Highly Purified hCG USP/BP 10,000 IU Requires 1 ml of 0.9% Sodium Chloride Excipients Mannitol / Monobasic Sodium Phosphate Bulking Agent / pH Buffer *Pack Sizes: 1 Vial of Lyophilized Powder + 1 Ampoule of Diluent (1 ml) per box.
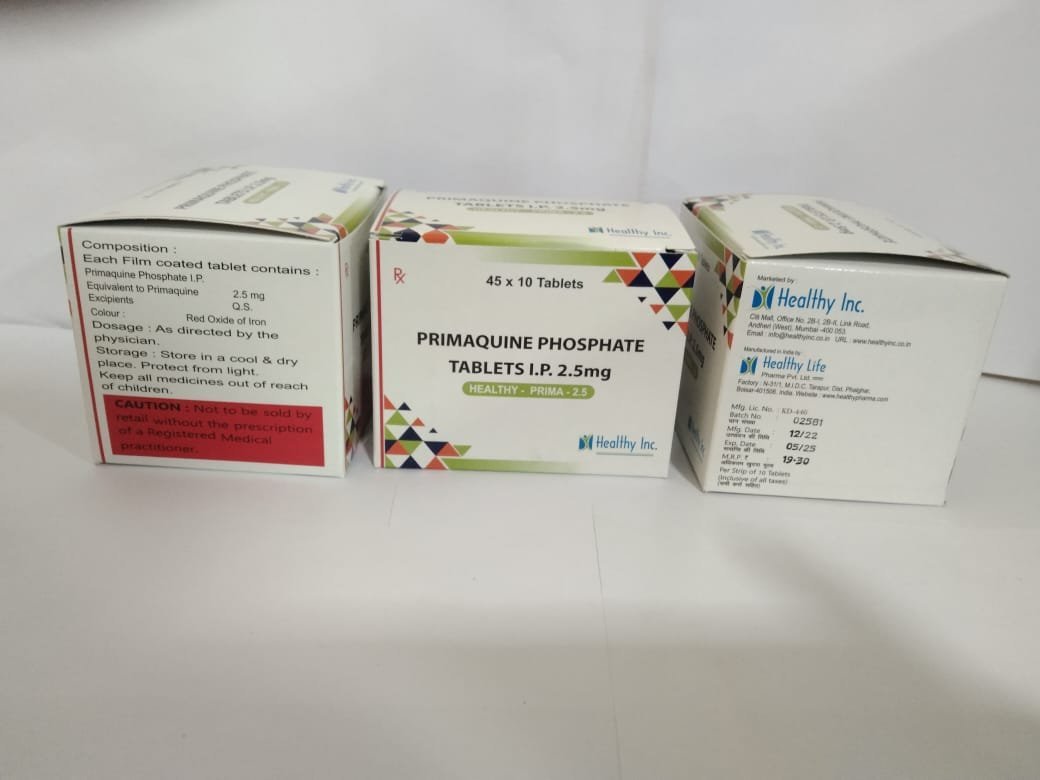
Primaquine Tablets
FreePrimaquine Phosphate Tablets IP 2.5 mg
Each Sugar Coated Tablet contains :
Primaquine Phosphate IP
Eq. To Primaquine 2.5 mgPrimaquine Phosphate tablets IP 2.5 mg
EACH FILM COATED TABLET CONTAINS:
Primaquine phosphate IP Equivalent to primaquine 2.5 mg
Primaquine Phosphate Tablets IP 7.5 mg
Each Sugar Coated Tablet contains :
Primaquine Phosphate IP
Eq. To Primaquine 7.5 mgPrimaquine Phosphate tablets IP 7.5 mg
EACH FILM COATED TABLET CONTAINS:
Primaquine phosphate IP , Equivalent to primaquine 7.5 mg
PRIMAQUINE PHOSPHATE TABLETS IP 15 MG
Each film coated Tablet Contains:
Primaquine Phosphate IP equivalent to primaquine 15 mgExcipients q.s.
Primaquine Phosphate Tablets 2.5 mg
Each Sugar coated Tablet contains:
Primaquine Phosphate BP
Eq. To Primaquine 2.5 mg
Excipients q.sPRIMAQUINE TABLETS 7.5 MG
Each film coated tablet contains:
Primaquine Phosphate BP – eq. to primaquine BP 7.5 mg
– Redoxide of iron –
– Excipients – QSUsage: – Treat or prevent malaria
Category: – Antimalarial drugs
Therapeutic category: – Antimalarial
Procaine Penicillin and Penicillin G Sodium Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Suspension in glass vials.
Active Ingredient Standard Dose (4 Lakh IU) Mega Dose (4 Mega IU) Procaine Penicillin G BP/USP 300,000 IU (Sustained Release) 3,000,000 IU (Sustained Release) Penicillin G Sodium BP/USP 100,000 IU (Immediate Peak) 1,000,000 IU (Immediate Peak) Excipients Lecithin / Sodium Citrate Suspending Agent / Buffer System *Pack Sizes: Single Vial or Tray of 50 Vials. (Requires reconstitution with Sterile Water for Injection to form a milky suspension).
Prochlorperazine Tablets
FreeProchlorperazine Maleate Tablets IP 5 mg
Each Uncoated Tablet contains :
Prochlorperazine Maleate IP 5 mgUsage: – control severe nausea and vomiting
Category: – Antipsychotic / CNS Drugs
Therapeutic category: – Antipsychotic
Procyclidine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Uncoated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu or high-density PVC/PVDC blister strips to ensure the absolute chemical stability of the hydrochloride salt.
Active Ingredient Strength Primary Clinical Function Procyclidine HCl BP/Ph.Eur. 5 mg Global Clinical Standard: Base therapeutic unit for the rapid reversal of EPS and parkinsonian rigidity. Excipients Lactose Monohydrate / Sodium Starch Glycolate / Povidone / Magnesium Stearate Diluent / Superdisintegrant / Binder / Lubricant (Engineered for rapid gastric dissolution and ultra-fast crossing of the blood-brain barrier) *Pack Sizes: 10×10 Blisters or Bottles of 100 (Optimized specifically for high-volume daily dispensing in acute psychiatric and neurology wards).
Progesterone Capsules
FreeProgesterone capsules are hormone replacement medications typically containing natural micronized progesterone, which is chemically identical to the hormone produced by the human body. They are most commonly available in 100 mg and 200 mg strengths.Progesterone Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Polymeric Matrix Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the micronized bio-identical hormone.
Active Ingredient Strength Primary Clinical Function Progesterone (Micronized) Ph.Eur./USP 200 mg (SR) HRT & Amenorrhea Standard: Base dose for secondary amenorrhea and endometrial protection during estrogen HRT. Progesterone (Micronized) Ph.Eur./USP 300 mg / 400 mg (SR) IVF & Obstetrics Standard: High-dose maintenance for Luteal Phase Support in ART/IVF and prevention of preterm birth. Excipients Hypromellose (HPMC) / Sodium Lauryl Sulfate / Microcrystalline Cellulose / Magnesium Stearate Hydrophilic Polymer Matrix / Wetting Agent / Diluent (Engineered specifically to maximize the dissolution of the highly insoluble micronized steroid and control its 24-hour release) *Pack Sizes: 10×10 Alu-Alu Blisters (Optimized specifically for strict, high-compliance obstetric protocols and IVF cycles).
Propanthaline Tablets
FreeXyproline Tablets
Propanthaline Tablets IP 15 mg
Each Coated tablet contains :
Propanthaline Bromide IP 15 mgUsage: – treat ulcers
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Propranolol Tablets
FreePropranolol Tablets BP 10 mg
Each Uncoated Tablet Contains:
Propranolol Hydrochloride BP 10 mgCIRPROL / TABLETS/PROPRANOLOL TABLETS BP 40 MG
Each uncoated tablet contains: Propranolol Hydrochloride BP 40 MGUsage: – High blood pressure. Treat illnesses
Category: – Hypertensive / Cardiac Drugs
Therapeutic category: – Hypertensive
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Pyrazinamide Tablets
FreePyrazinamide Tablet IP
Each Coated Tablets Contains:
Pyrazinamide IP 500 mgPyrazinamide tablet IP 750 mg
Each Coated Tablet Contains:
Pyrazinamide IP 750 mgPYRAZINAMIDE TABLETS BP 400 MG
Each filmcoated tablet contains:
Pyrazinamide BP 400 mgUsage: – Treat tuberculosis (TB)
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic, Antituberculosis
Pyridostigmine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips or heavy-density HDPE bottles with desiccants to ensure the absolute chemical stability of the highly hygroscopic bromide salt.
Active Ingredient Strength Primary Clinical Function Pyridostigmine Bromide USP/Ph.Eur. 60 mg Global Clinical Standard: Base maintenance dose for continuous 24-hour neuromuscular control in MG patients. Excipients Silicone Dioxide / Microcrystalline Cellulose / Pregelatinized Starch / Magnesium Stearate Glidant / Diluent / Binder / Lubricant (Engineered for immediate gastric dissolution and precise, rapid systemic onset within 30-45 minutes) *Pack Sizes: Bottles of 100 or 10×10 Alu-Alu Blisters (Optimized specifically for strict, high-volume daily neurology regimens where patients may consume 6 to 10 tablets daily).
Pyridoxine Hydrochloride Injection
FreeProduct Composition & Strength
We supply this product as a Sterile, Clear, Colorless to Pale Yellow Solution in glass ampoules or vials.
Active Ingredient Concentration Total Content & Format Pyridoxine Hydrochloride USP/BP 100 mg / ml 200 mg in 2 ml Glass Ampoule Pyridoxine Hydrochloride USP/BP 100 mg / ml 1000 mg (1 g) in 10 ml Multi-Dose Vial (Antidote Protocol) Excipients Water for Injection Preservative-Free (Ampoules) / Chlorobutanol (Vials) *Pack Sizes: Tray of 10 Ampoules or Box of 100 Ampoules.
Pyridoxine Tablets
FreePyridoxine Hydrochloride Tablets IP
Eac Uncoated tablet contains :
Pyridoxine Hydrochloride IP 25 mgPyridoxine Tablets IP 50 mg
Each Uncoated tablet contains:
Pyridoxine Hydrochoride IP 50 mgPyridoxine Tablets IP 100 mg
Each Uncoated tablet contains:
Pyridoxine Hydrochoride IP 100 mgPyridoxine Tablets BP 25 mg
Eac Uncoated tablet contains :
Pyridoxine Hydrochloride BP 25 mgUsage: – Prevent or treat a certain nerve disorder
Category: – Vitamin supplement
Therapeutic category: – Vitamin supplement
Quinapyramine Sulphate And Quinapyramine Chloride Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection in glass vials, requiring reconstitution with Sterile Water for Injection to form an aqueous suspension.
Active Ingredient Standard 2.5 g Vial Function Quinapyramine Sulphate BP (Vet) 1.5 g Curative (Immediate Release) Quinapyramine Chloride BP (Vet) 1.0 g Prophylactic (Slow Release / Depot) Total Pro-salt Content 2.5 g *Reconstitute with 15 ml Sterile Water *Pack Sizes: Single 2.5g or 3g Vial, often packed with a Sterile Water diluent ampoule.
Quinine Injection
FreeProduct Composition & Strength
We supply this product as a Sterile, Clear, Pale Yellow Aqueous Solution in glass ampoules.
Active Ingredient Concentration Total Content & Format Quinine Dihydrochloride BP 300 mg / ml 600 mg in 2 ml Glass Ampoule Excipients Water for Injection Preservative-Free Solution *Pack Sizes: Tray of 10 Ampoules or Box of 100 Ampoules.
Quinine Sulphate Tablets
FreeGlaquine
Quinine Sulphate Tablets BP 300 mg
Eac Sugarcoated tablet contains :
Quinine Sulphate BP 300 mgUsage: – treat malaria caused by mosquito bites
Category: – Antimalarial drugs
Therapeutic category: – Antimalarial
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Rabeprazole injection
FreeComposition
Each vial contains : Rabeprazole sodium 20 mg,
Reconstitute with 5ml of sterile water for injection
provided in this packUsage: –Use a syringe to withdraw the specified amount of sterile water for injection.
Category: – Rabeprazole injection
Therapeutic category: – gastroesophageal reflux
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Rabeprazole Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder (White to Pale Yellow Cake) in clear glass vials.
Active Ingredient Strength Reconstitution Requirement Rabeprazole Sodium USP/BP 20 mg Requires 5 ml of Sterile Water for Injection Excipients Mannitol / Sodium Hydroxide Bulking Agent / Alkalizing Stabilizer *Pack Sizes: Single Vial or Tray of 10 Vials. (Often packed with a 5 ml diluent ampoule).
Rabeprazole Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Enteric-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the highly acid-labile Rabeprazole sodium salt.
Active Ingredient Strength Primary Clinical Function Rabeprazole Sodium INN/Ph.Eur. 10 mg Maintenance Standard: Long-term maintenance of healed erosive esophagitis and symptomatic GERD. Rabeprazole Sodium INN/Ph.Eur. 20 mg Global Clinical Standard: Acute healing of erosive esophagitis, active duodenal ulcers, and H. Pylori eradication. Excipients Light Magnesium Oxide / Mannitol / Methacrylic Acid Copolymer / Triethyl Citrate / Titanium Dioxide Alkaline Core Stabilizer / Diluent / Enteric Coating Polymer / Plasticizer (Engineered to absolutely block stomach acid and dissolve only in the alkaline duodenum) *Pack Sizes: 10×10 Alu-Alu Blisters (Optimized specifically for strict 14-to-28 day acute gastroenterology dispensing regimens).
Racecadotril Capsules
FreeRacecadotril capsules are non-antibiotic, anti-diarrheal medications used primarily for the treatment of acute diarrhea. They are most commonly available in a 100 mg strength for adults.
Racecadotril Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet or Hard Gelatin Capsule, packed exclusively in highly secure, moisture-resistant Alu-Alu or high-density Alu-PVC blister strips to ensure the absolute chemical stability of the prodrug formulation.
Active Ingredient Strength Primary Clinical Function Racecadotril INN/Ph.Eur. 100 mg Global Clinical Standard: Adult dose for the rapid, anti-secretory resolution of acute diarrhea. Excipients Microcrystalline Cellulose / Pregelatinized Starch / Croscarmellose Sodium / Colloidal Silicon Dioxide / Magnesium Stearate Diluent / Binder / Superdisintegrant / Glidant (Engineered for immediate gastric dissolution and rapid conversion to the active metabolite, Thiorphan) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict 3-to-7 day acute gastrointestinal dispensing regimens).
Ramipril & Hydrochlorothiazide Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet or Capsule, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the highly degradable Ramipril molecule.
Active Ingredient Strength Primary Clinical Function Ramipril Ph.Eur./USP 5 mg (or 2.5 mg / 10 mg) ACE inhibition for vasodilation, RAAS blockade, and critical potassium retention. Hydrochlorothiazide Ph.Eur./USP 12.5 mg / 25 mg Global Clinical Standard: Thiazide diuretic for sodium excretion and sustained blood volume reduction. Excipients Microcrystalline Cellulose / Sodium Starch Glycolate / Sodium Bicarbonate / Hypromellose / Magnesium Stearate Diluent / Superdisintegrant / Alkaline Stabilizing Agent (Engineered specifically to protect Ramipril from acidic degradation and moisture-induced cyclization) *Pack Sizes: 10×10 Alu-Alu Blisters or 3×10 Unit-Dose Blisters (Optimized specifically for strict, high-compliance 30-day chronic cardiology dispensing regimens).
Ramipril and Amlodipine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Capsule or Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the highly degradable Ramipril molecule.
Active Ingredient Strength Primary Clinical Function Ramipril Ph.Eur./USP 5 mg / 10 mg ACE inhibition for vasodilation, neurohormonal blockade, and venous pressure equalization. Amlodipine (as Besilate) Ph.Eur./USP 5 mg / 10 mg Global Clinical Standard: Sustained CCB for profound, 24-hour arterial vasodilation. Excipients Microcrystalline Cellulose / Crospovidone / Sodium Stearyl Fumarate / Hypromellose / Iron Oxide Red Diluent / Superdisintegrant / Alkaline Stabilizing Lubricant (Engineered to protect Ramipril from acidic degradation and moisture) *Pack Sizes: 10×10 Alu-Alu Blisters or 3×10 Unit-Dose Blisters (Optimized specifically for strict, high-compliance 30-day chronic cardiology dispensing regimens).
Ramipril Tablets
FreeRamipril Tablets IP 2.5mg
Each Film coated tablet contains:
Ramipril IP 2.5 mgRamipril tablets IP 5 mg
Each Filmcoated tablet contains :
Ramipril IP 5 mgUsage: – high blood pressure and heart failure
Category: – Anti Cardiovascular drugs
Therapeutic category: – Cardiovascular Agent, Antihypertensive
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
RANITIDINE INJECTION
FreeComposition
Each ml. contains:Rani9dine Hydrochloride I.P. 27.9 mg.
Eq. to Rani9dine 25 mg.
Phenol I.P. 0.5 % w/v(As Preserva9ve)
Water for Injection I.P 2 mlUsage: – Indigestion, heartburn and acid reflux, gastro-esophageal reflux disease
Category: – Antacid & Anti ulcer injection
Therapeutic category: – Antiulcer, Antacid
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Ranitidine Injection
FreeProduct Composition & Strength
We supply this product as a Sterile, Clear, Aqueous Solution in glass ampoules.
Active Ingredient Concentration Total Content & Format Ranitidine Hydrochloride USP/BP 25 mg / ml 50 mg in 2 ml Glass Ampoule Excipients Phenol / Potassium Di-hydrogen Phosphate Preservative / pH Buffer System (pH 6.7 to 7.3) *Pack Sizes: Tray of 10 Ampoules, 50 Ampoules, or 100 Ampoules.
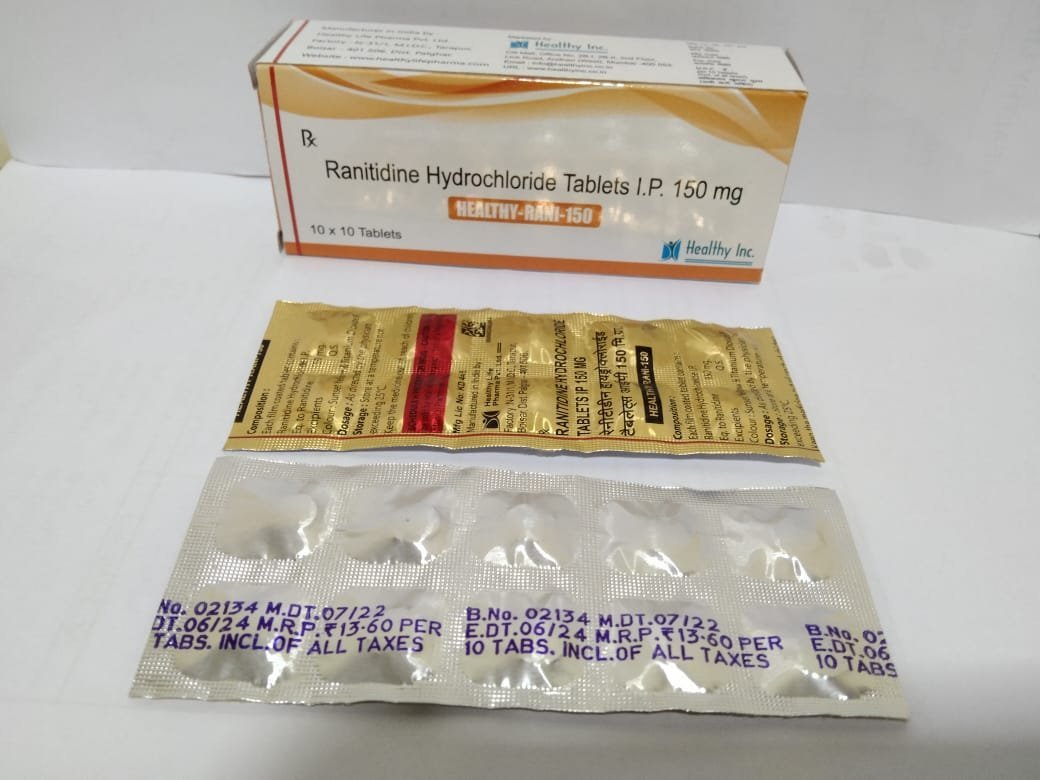
Ranitidine Tablets
FreeRanitidine Hydrochloride Tablets IP
Each Filmcoated tablet contains :
Ranitidine Hydrochloride IP
Eq. To Ranitidine 150 mgRanitidine Hydrochloride Tablets IP
Each Filmcoated tablet contains :
Ranitidine Hydrochloride IP
Eq. To Ranitidine 300 mgRanitidine Hydrochloride Tablets USP
Each Filmcoated tablet contains :
Ranitidine Hydrochloride USP
Eq. To Ranitidine 300 mgUsage: – Indigestion, heartburn and acid reflux, gastro-esophageal reflux disease
Category: – Antacid & Anti ulcer drugs
Therapeutic category: – Antiulcer, Antacid
Ranolazine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Polymeric Matrix Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips or heavy-gauge HDPE bottles to ensure the absolute stability of the modified-release polymers.
Active Ingredient Strength Primary Clinical Function Ranolazine INN/Ph.Eur. 500 mg (Extended-Release) Global Clinical Standard: Base initiation dose for chronic angina management (taken twice daily). Ranolazine INN/Ph.Eur. 1000 mg (Extended-Release) Advanced maintenance dose for severe, refractory angina requiring maximum cellular relaxation. Excipients Hypromellose (HPMC) / Methacrylic Acid Copolymer / Microcrystalline Cellulose / Magnesium Stearate Hydrophilic Polymer Matrix / Enteric Matrix / Diluent (Engineered to form a viscous gel layer that controls the strict 12-hour drug diffusion rate) *Pack Sizes: 10×10 Alu-Alu Blisters or Bottles of 60 (Optimized specifically for strict 30-day chronic cardiology dispensing regimens).
Rasagiline Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Uncoated or Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the highly sensitive mesylate salt.
Active Ingredient Strength Primary Clinical Function Rasagiline Mesylate INN/Ph.Eur. 0.5 mg (Base Equivalent) Adjunct Standard: Initiating dose when used in combination with Levodopa. Rasagiline Mesylate INN/Ph.Eur. 1 mg (Base Equivalent) Global Clinical Standard: Maximum daily dose for monotherapy or advanced adjunct therapy. Excipients Mannitol / Maize Starch / Pregelatinized Starch / Colloidal Silicon Dioxide / Stearic Acid Diluent / Binder / Superdisintegrant / Glidant (Engineered for immediate gastric dissolution and rapid crossing of the blood-brain barrier) *Pack Sizes: 10×10 Alu-Alu Blisters or 3×10 Unit-Dose Blisters (Optimized specifically for strict high-compliance 30-day chronic neurology dispensing regimens).
Rebamipide Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu or high-density Alu-PVC blister strips to ensure the absolute chemical stability of the active mucosal agent.
Active Ingredient Strength Primary Clinical Function Rebamipide INN/Ph.Eur. 100 mg Global Clinical Standard: Mucosal protective agent for active ulcer healing and gastritis management. Excipients Microcrystalline Cellulose / Hydroxypropyl Methylcellulose (HPMC) / Croscarmellose Sodium / Magnesium Stearate Diluent / Premium Film-Coating / Superdisintegrant (Engineered for optimal gastric dispersion directly onto the mucosal lining) *Pack Sizes: 10×10 Blisters (Optimized specifically for strict 30-day chronic gastroenterology dispensing regimens).
Repaglinide & Metformin Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu or heavy-density PVC/PVDC blister strips to ensure the absolute chemical stability of the combined formulation.
Active Ingredient Strength Primary Clinical Function Repaglinide USP/Ph.Eur. 1 mg / 2 mg Short-acting meglitinide for rapid post-prandial (after-meal) insulin stimulation. Metformin Hydrochloride USP/Ph.Eur. 500 mg Global Clinical Standard: Biguanide for hepatic glucose suppression and insulin sensitization. Excipients Microcrystalline Cellulose / Povidone K30 / Croscarmellose Sodium / Poloxamer / Magnesium Stearate Diluent / Binder / Superdisintegrant / Surfactant (Engineered for immediate gastric dissolution and synchronized dual-absorption) *Pack Sizes: 10×10 or 3×10 Blisters (Optimized specifically for high-compliance 30-day chronic metabolic dispensing regimens tied directly to meal times).
Repaglinide Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu or heavy-density PVC/PVDC blister strips to ensure the absolute chemical stability of the highly sensitive meglitinide API.
Active Ingredient Strength Primary Clinical Function Repaglinide USP/Ph.Eur. 0.5 mg Base titration unit for treatment-naive patients or those with severe renal/hepatic impairment. Repaglinide USP/Ph.Eur. 1 mg / 2 mg Global Clinical Standard: Advanced maintenance doses for rapid post-prandial insulin stimulation. Excipients Microcrystalline Cellulose / Calcium Hydrogen Phosphate / Meglumine / Poloxamer / Magnesium Stearate Diluent / Stabilizer / Surfactant / Lubricant (Engineered for immediate gastric dissolution and ultra-fast systemic absorption) *Pack Sizes: 10×10 or 3×10 Blisters (Optimized specifically for high-compliance 30-day chronic metabolic dispensing regimens tied directly to meal times).
Rhoclone Injection
FreeProduct Composition & Strength
We supply this product as a Sterile, Ready-to-Use Liquid Solution or Lyophilized Powder in pre-filled syringes (PFS) or glass vials.
Active Ingredient Strength (mcg & IU) Standard Indication Anti-D (Rho) Immunoglobulin 150 mcg (750 IU) Micro-hemorrhage / 1st Trimester Miscarriage Anti-D (Rho) Immunoglobulin 300 mcg (1500 IU) Standard Postpartum Dose / Antenatal Prophylaxis Excipients Glycine / Polysorbate 80 Stabilizer / Surfactant (Preservative-Free) *Pack Sizes: Single Pre-Filled Syringe (PFS) or Single Vial.
Riboflavin Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips or opaque, high-density HDPE bottles to protect the violently light-sensitive vitamin.
Active Ingredient Strength Primary Clinical Function Riboflavin USP/Ph.Eur. 100 mg Nutritional Standard: Treatment of severe Ariboflavinosis, stomatitis, and malnutrition. Riboflavin USP/Ph.Eur. 400 mg Neurology Standard: High-dose daily prophylaxis for chronic migraine headaches. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Hypromellose / Titanium Dioxide / Magnesium Stearate Diluent / Superdisintegrant / UV-Blocking Film Coating (Engineered to protect the API from light degradation and ensure immediate gastric release) *Pack Sizes: 10×10 Alu-Alu Blisters or Bottles of 100 (Optimized specifically for chronic daily neurology regimens and massive NGO bulk dispensing).
Rifampicin & Isoniazid Tablets
FreeTechnical & Manufacturing Specifications
Formulated for chemical stability and precise bioavailability of both active ingredients.
Technical Metric Specification Standard Active Composition Rifampicin (150mg/300mg) + Isoniazid (75mg/150mg) Dosage Form Film-Coated Tablet HS Code 3004.20.19 (Anti-TB Medicaments) Packaging Alu-Alu Blister (Crucial for Rifampicin stability) Shelf Life 24 to 36 Months (Subject to stability data) Rifampicin & Isoniazid Tablets
FreeProduct Composition & Strength (RH 300/150)
We supply this product in standardized Fixed-Dose Combinations (FDCs) as per international pharmacopoeial standards.
Active Ingredient Strength (per Tablet) Primary Clinical Function Rifampicin IP/BP/USP 300 mg The RNA Blocker: Sterilizing agent against TB bacilli. Isoniazid IP/BP/USP 150 mg The Wall Breaker: Potent bactericidal agent. Excipients Pharma Grade The Matrix: Ensures stability of the FDC. Rifampicin Capsules
FreeRifampicin capsules (also known as rifampin) are potent broad-spectrum antibiotics primarily used for treating tuberculosis and leprosy. They are typically available in strengths of 150 mg and 300 mg.
Rifampicin Isoniazid Tablets
FreeRifampicin And Isoniazid Tablets Int Ph
Each Film Coated Tablet Contains:
Rifampicin BP/Int.Ph. 150 mg
Isoniazid BP/Int.Ph. 150 mgISORIF TABLETS
(Rifampicin And Isoniazid Tablets Int. Ph.)
Each Film Coated Tablet Contains:
Rifampicin BP/Int. Ph. 300 mg
Isoniazid BP/Int. Ph. 150 mgRifampicin And Isoniazid Tablets Int. Ph.
Each Film Coated Tablet Contains:
Rifampicin BP/Int. Ph. 150 mg
Isoniazid BP/Int. Ph. 75 mgRIFAMPICIN IP 150 MG + ISONIAZIDE IP 300 MG TABLETS Each film coated tablet contains: Rifampicin IP 150 mg isoniazide IP 300 mg Excipients Q.S.9
Usage: – treat tuberculosis (TB) infection
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic,
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Rifampicin Isoniazide Pyrizinamide Tablets
FreeRifampicin, Isoniazide and Pyrizinamide Tab (Combi Pack)
(Rifampicin Tablets IP) + (Isoniazide Tablets IP) + (Pyrizinamide Tablets IP)
Each film Coated Tablet Contains :
Rifampicin IP 100 mg
Each coated Tablet Contains :
Isonoazide IP 50 mg
Each coated Tablet contains :
Pyrizinamide IP 250 mgRIP TABLETS
(Rifampicin, Isoniazid And Pyrazinamide Tablets Int.Ph.)
Each Filmcoated Tablet Contains:
Rifampicin BP/Int.Ph. 150 mg
Isoniazid BP/Int.Ph. 75 mg
Pyrazinamide BP/Int.Ph. 400 mgUsage: – treat tuberculosis (TB) infection
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic, Antituberculosis
Rifampicin Tablet
FreeRifampicin Tablets IP 100 mg
Each film Coated Tablet Contains:
Rifampicin IP 100 mgRifampicin Tablets Int. Ph. 300 mg
Each Film Coated Tablets Contains:
Rifampicin BP/Int.Ph. 300 mgUsage: – treat or prevent tuberculosis (TB)
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic,
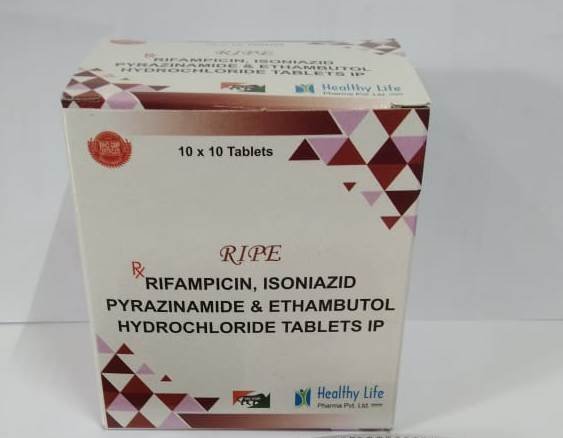
Rifampicin, Isoniazid & Pyrazinamide Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets (Red/Brown). Stability is paramount as Rifampicin can degrade in the presence of Isoniazid if not properly formulated.
Active Ingredient Strength (Standard Adult) Strength (Pediatric) Therapeutic Role Rifampicin IP/BP/USP 150 mg 75 mg / 60 mg Broad Spectrum Sterilizer Isoniazid IP/BP/USP 75 mg 50 mg / 30 mg Cell Wall Inhibitor Pyrazinamide IP/BP/USP 400 mg 150 mg Intracellular Sterilizer Excipients Q.S. Ascorbic Acid (Stabilizer) Bioavailability Enhancer Rifampicin, Isoniazid & Pyrazinamide Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets (Red/Brown). Stability is paramount as Rifampicin can degrade in the presence of Isoniazid if not properly formulated.
Active Ingredient Strength (Standard Adult) Strength (Pediatric) Therapeutic Role Rifampicin IP/BP/USP 150 mg 75 mg / 60 mg Broad Spectrum Sterilizer Isoniazid IP/BP/USP 75 mg 50 mg / 30 mg Cell Wall Inhibitor Pyrazinamide IP/BP/USP 400 mg 150 mg Intracellular Sterilizer Excipients Q.S. Ascorbic Acid (Stabilizer) Bioavailability Enhancer Rifampicin, Isoniazid, Pyrazinamide & Ethambutol Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets (usually Brown or Reddish-Brown). We offer both the “Daily Regimen” and “Intermittent Regimen” strengths.
Active Ingredient Strength (WHO Daily FDC) Therapeutic Role Rifampicin IP/BP/USP 150 mg Sterilizing / Broad Spectrum Isoniazid IP/BP/USP 75 mg Early Bactericidal Pyrazinamide IP/BP/USP 400 mg Intracellular Sterilizing Ethambutol HCl IP/BP/USP 275 mg Resistance Prevention Rifaximin Tablets
FreeRifaximin Tablets 400 mg Each Film Coated Tablet Contains : Rifaximin In house 400 mg
Usage: – prevent episodes of hepatic encephalopathy
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic
Rifaximin Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute stability of the crystalline polymorph structure.
Active Ingredient Strength Primary Clinical Function Rifaximin Ph.Eur. 200 mg / 400 mg GI Infection Dose: Acute eradication of non-invasive E. coli (Traveler’s Diarrhea). Rifaximin Ph.Eur. 550 mg Hepatology & IBS Standard: High-dose maintenance therapy for Hepatic Encephalopathy and IBS-D. Excipients Microcrystalline Cellulose / Sodium Starch Glycolate / Colloidal Silicon Dioxide / Propylene Glycol Diluent / Superdisintegrant / Glidant / Film-Coating Plasticizer (Engineered for optimal dispersion throughout the intestinal lumen) *Pack Sizes: 10×10 Alu-Alu Blisters or 14-Tablet Blisters (Optimized specifically for standard acute infection or chronic hepatology dispensing regimens).
Risperidone Tablets
FreeRisperidone Tablets 1 mg
Each film coated tablet contains:
Risperidone IP 1 mgRisperidone Tablets 2 mg
Each film coated tablet contains:
Risperidone IP 2 mgRisperidone Tablets 3 mg
Each film coated tablet contains:
Risperidone IP 3 mgRisperidone Tablets 4 mg
Each film coated tablet contains:
Risperidone IP 4 mgRisperidone Tablets 1 mg
Each film coated tablet contains:
Risperidone BP 1 mgRisperidone Tablets 2 mg
Each film coated tablet contains:
Risperidone BP 2 mgRisperidone Tablets 3 mg
Each film coated tablet contains:
Risperidone BP 3 mgRisperidone Tablets 4 mg
Each film coated tablet contains:
Risperidone BP 4 mgUsage: – treat certain mental/mood disorders
Category: – Anti Anxiety drugs
Therapeutic category: – Antipsychotic, Antidepressant, CNS Drugs, Anti Anxiety
Rivaroxaban Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute stability of the potent DOAC molecule.
Active Ingredient Strength Primary Clinical Function Rivaroxaban INN/Ph.Eur. 2.5 mg Vascular Dose: Co-administered with Aspirin to prevent major cardiovascular events in CAD/PAD. Rivaroxaban INN/Ph.Eur. 10 mg Surgical Prophylaxis: Prevention of DVT following knee or hip replacement surgery. Rivaroxaban INN/Ph.Eur. 15 mg / 20 mg Global Clinical Standard: Treatment of acute DVT/PE and stroke prevention in Atrial Fibrillation. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Hypromellose / Sodium Lauryl Sulfate / Magnesium Stearate Diluent / Superdisintegrant / Surfactant (Engineered for optimal solubilization and synchronized gastrointestinal absorption) *Pack Sizes: 10×10 Alu-Alu Blisters or 14/28/30-Tablet Blisters (Optimized specifically for strict chronic cardiology and acute post-op dispensing regimens).
Ropinirole Extended-Release
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Polymeric Matrix Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips or heavy-gauge HDPE bottles to ensure the absolute stability of the modified-release polymers.
Active Ingredient Strength Primary Clinical Function Ropinirole HCl USP/Ph.Eur. 2 mg / 4 mg Base titration units for the slow, safe initiation of dopaminergic therapy. Ropinirole HCl USP/Ph.Eur. 8 mg Global Clinical Standard: Advanced maintenance dose for 24-hour continuous symptom control. (Higher total daily doses may be required). Excipients Hypromellose (HPMC) / Carmellose Sodium / Lactose Monohydrate / Magnesium Stearate Hydrophilic Polymer Matrix / Diluent / Lubricant (Engineered to form a viscous gel layer that controls the strict 24-hour drug diffusion rate) *Pack Sizes: 10×10 Alu-Alu Blisters or Bottles of 28/30 (Optimized specifically for strict 30-day chronic neurology dispensing regimens).
Rosuvastatin & Clopidogrel Capsules
FreeRosuvastatin and clopidogrel capsules are fixed-dose combination (FDC) medications used for the secondary prevention of cardiovascular events like heart attacks and strokes. They are typically available in strengths such as 10 mg/75 mg or 20 mg/75 mg.
Rosuvastatin Clopidogrel and Aspirin Capsules
FreeRosuvastatin, clopidogrel, and aspirin capsules are fixed-dose combination (FDC) medications used for the prevention of serious cardiovascular events such as heart attacks and strokes. This “triple therapy” combines a cholesterol-lowering statin with two antiplatelet agents
Rosuvastatin Tablets
FreeRucol
Rosuvastatin Tablets 10 mg
Each Film Coated Tablet Contains:
Rosuvastatin Calcium
eq. To Rosuvastatin 10 mgUsage: – treat certain mental/mood disorders
Category: – Anti Cardiovascular drugs
Therapeutic category: – Antilipidemic, Cardiovascular Agent
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Roxithromycin Tablets
FreeRoxiron-150
(Roxithromycin tablets 150 mg)
Each Film coated tablets contains:
Roxithromycin BP 150 mgUsage: – treats bacterial infections
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic
Sacubitril & Valsartan Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the sensitive dual-molecule complex.
Active Ingredient Strength (Total Dose) Primary Clinical Function Sacubitril / Valsartan INN/Ph.Eur. 24 mg / 26 mg (50 mg Total) Starting dose for patients with severe renal impairment, hepatic impairment, or those not currently taking an ACEi/ARB. Sacubitril / Valsartan INN/Ph.Eur. 49 mg / 51 mg (100 mg Total) Standard Initiation Dose: For patients switching from standard doses of ACE inhibitors or ARBs. Sacubitril / Valsartan INN/Ph.Eur. 97 mg / 103 mg (200 mg Total) Target Maintenance Dose: Maximum adult therapy for optimal mortality reduction in heart failure. Excipients Microcrystalline Cellulose / Crospovidone / Colloidal Silicon Dioxide / Magnesium Stearate Diluent / Superdisintegrant / Glidant (Engineered for immediate gastric dissolution and synchronized dual-absorption) *Pack Sizes: 14×2 or 28-Tablet Alu-Alu Blisters (Optimized specifically for strict twice-daily chronic cardiology dispensing regimens).
Salbutamol & Theophylline Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of both active pharmaceutical ingredients.
Active Ingredient Strength Primary Clinical Function Salbutamol Sulphate BP/Ph.Eur. 2 mg (Equivalent to Salbutamol base) Rapid-onset beta-2 agonism for immediate smooth muscle relaxation. Theophylline (Anhydrous) BP/Ph.Eur. 100 mg Global Clinical Standard: PDE inhibition for sustained bronchodilation and mild respiratory anti-inflammation. Excipients Microcrystalline Cellulose / Maize Starch / Purified Talc / Magnesium Stearate Diluent / Binder / Glidant / Lubricant (Engineered for strict, synchronized gastric dissolution) *Pack Sizes: 10×10 Blisters (Optimized specifically for high-compliance 30-day chronic pulmonology dispensing regimens).
Salbutamol Inhaler
FreeSalbutamol Inhaler Metered-Dose Inhaler
Product Composition & Strength
We supply this product in standardized Metered-Dose Inhaler (MDI) formats, optimized for direct pulmonary delivery.
Component Strength (per actuation) Primary Clinical Function Salbutamol (as Sulfate) IP/BP 100 mcg The Rescue Agent: Instantly opens airways. Propellant HFA-134a (CFC-Free) The Carrier: Ensures ozone-friendly delivery. Formulation Pressurized Aerosol The Standard: 200 metered doses per canister. Salbutamol Tablets
FreeSalbutamol Tablets IP 2 mg Each Uncoated Tablet contains: Salbutamol Sulphate IP – Equivalent to Salbutamol 2 mg
Excipients – QSSalbutamol Tablets IP 4 mg Each uncoated tablets contains: Salbutamol Sulphate IP –
Equivalent to Salbutamol 4 mg
Excipients – QSSalbutamol Tablets BP
Each Uncoated tablet contains:
Salbutamol Sulphate BP
Eq. to Salbutamol 4 mgUsage: – treat wheezing and shortness of breath caused by breathing problems
Category: – Antacid & Anti ulcer drugs
Therapeutic category: – Antiasthmatic
Saxagliptin Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the sensitive active pharmaceutical ingredient.
Active Ingredient Strength Primary Clinical Function Saxagliptin (as Hydrochloride) INN/Ph.Eur. 2.5 mg Target dose for patients with moderate/severe renal impairment or those taking strong CYP3A4 inhibitors. Saxagliptin (as Hydrochloride) INN/Ph.Eur. 5 mg Global Clinical Standard: Maximum adult daily dose for the maintenance of Type 2 Diabetes. Excipients Lactose Monohydrate / Microcrystalline Cellulose / Croscarmellose Sodium / Magnesium Stearate Diluent / Binder / Superdisintegrant (Engineered for immediate gastric dissolution and optimal bioavailability) *Pack Sizes: 10×10 Alu-Alu Blisters or 3×10 Unit-Dose Blisters (Optimized specifically for high-compliance 30-day chronic metabolic dispensing regimens).
Secnidazole Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets (typically White or Off-White). The high-dose tablet is designed for ease of swallowing.
Active Ingredient Strength (Standard) Therapeutic Role Secnidazole USP/BP 500 mg Pediatric / Divided Dose Secnidazole USP/BP 1000 mg (1g) Standard Adult Dose (2 tablets = 2g) Excipients Q.S. Microcrystalline Cellulose / Sodium Starch Glycolate Binder / Disintegrant Secnidazole Tablets
FreeSecnidazole Tablets IP 500 mg
Each Filmcoated tablet contains :
Secnidazole IP 500 mgSecnidazole Tablets I.P.
Each Filmcoated tablet contains :
Secnidazole IP 1 gmSeczole -2 Secnidazole Tablets IP 2 gm
Each film coated tablet contains:
Secnidazole IP 2 gmUsage: – treat vaginal infection (vaginosis) caused by bacteria
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Senna Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated or Uncoated Tablet, packed in highly secure, moisture-resistant PVC/PVDC or Alu-PVC blister strips to ensure the absolute chemical stability of the botanical extract.
Active Ingredient Strength Primary Clinical Function Sennosides (Standardized Extract) USP/Ph.Eur. 12 mg (Calculated as Sennoside B) Global Retail & Clinical Standard: Targeted adult dose for rapid, overnight relief of severe constipation. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Calcium Phosphate / Magnesium Stearate Diluent / Superdisintegrant / Binder (Engineered for immediate gastric transit to the lower intestine) *Pack Sizes: 10×10 Blisters or Bottles of 100 (Optimized specifically for high-turnover retail pharmacy shelves and chronic geriatric/oncology hospital dispensing).
Serratiopeptidase Tablets
FreeSERRATIOPTIDASE TABLETS 5 MG
Enteric Coated Tablet:
Serratiopeptidase 10000 Serratiopeptidase unit 5 MG
Usage: – back pain, osteoarthritis, rheumatoid arthritis, osteoporosis, fibromyalgia, carpel tunnel syndrome, migraine headache, and tension headache
Category: – Anti-inflammatory drug (NSAIDs)
Therapeutic category: – Anti-inflammatory
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Sertaconazole Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Compressed Vaginal Tablet (Pessary), packed exclusively in highly secure, moisture-resistant Alu-PVC blister strips. Each tablet is often packaged with a hygienic, medical-grade disposable applicator to ensure proper and comfortable insertion.
Active Ingredient Strength Primary Clinical Function Sertaconazole Nitrate BP/Ph.Eur. 500 mg Global Clinical Standard: Single-dose acute therapy for the complete eradication of vaginal Candida infections. Excipients Lactose Monohydrate / Microcrystalline Cellulose / Adipic Acid / Sodium Bicarbonate / Magnesium Stearate Diluent / Effervescent Base (Engineered to rapidly dissolve in the low-moisture vaginal environment and disperse evenly across the mucosal walls) *Pack Sizes: 1 Tablet per Blister Box + 1 Disposable Applicator (Optimized specifically for premium, high-turnover OTC pharmacy shelves and acute gynecology dispensing).
Sertraline Tablets
FreeSertraline Tablet IP 50 mg
Each Filmcoated Tablet Contains:
Sertraline Hydrochloride
eq. To Sertraline 50 mgUsage: – Treat depression
Category: – Anti Anxiety drugs
Therapeutic category: – Antipsychotic, Antidepressant, CNS Drugs, Anti Anxiety
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Sevelamer Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Compressed Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant heavy-gauge HDPE bottles to prevent the highly hygroscopic polymer from absorbing ambient humidity and expanding.
Active Ingredient Strength Primary Clinical Function Sevelamer Carbonate INN/Ph.Eur. 800 mg Global Standard: High-dose adult therapy for the aggressive management of hyperphosphatemia. Sevelamer Carbonate INN/Ph.Eur. 400 mg Base titration unit for specific dietary adjustments and milder phosphate elevations. Excipients Microcrystalline Cellulose / Hypromellose / Diacetylated Monoglycerides / Colloidal Silicon Dioxide / Zinc Stearate Diluent / Premium Film-Coating / Glidant / Lubricant (Engineered for structural integrity and smooth swallowing) *Pack Sizes: Bottles of 30, 90, or 270 Tablets (Optimized specifically for strict, massive-volume daily dialysis regimens where patients consume multiple grams with every meal).
Sildenafil Citrate Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets (typically Blue, Diamond-Shaped). The coating is essential to mask the extremely bitter taste of the citrate salt.
Active Ingredient Strength (Standard) Therapeutic Role Sildenafil Citrate USP/BP 25 mg Initiation / Renal Impairment Sildenafil Citrate USP/BP 50 mg Standard Adult Dose Sildenafil Citrate USP/BP 100 mg Maximum Efficacy Dose Excipients Q.S. Microcrystalline Cellulose / Indigo Carmine Binder / Blue Colorant Sildenafil Citrate oral jelly
FreeProduct Composition & Strength
We supply this product as a Single-Dose Liquid Jelly Sachet, optimized for portability and discreet use.
Active Ingredient Strength (per 5g sachet) Primary Clinical Function Sildenafil Citrate IP/BP 100 mg The PDE5 Inhibitor: Restores erectile function. Excipients Glycerin / Sodium CMC The Vehicle: Ensures smooth jelly texture and stability. Flavoring Orange / Strawberry / Pineapple / Butterscotch The Palatable Base: Improves patient compliance. Sildenafil Tablets
FreeVegatone – 25
Sildenfil Citrate Tablets
Each Film Coated Tablet Contains:
Sildenfil Citrate eq. to Sildenfil 25 mgVegatone – 50
Sildenafil Citrate tablets
Each Filmcoated tablet contains :
Sildenafil Citrate Eq. To Sildenafil 50 mgVegatone – 100
Sildenafil Citrate tablets
Each Filmcoated tablet contains :
Sildenafil Citrate Eq. To Sildenafil 100 mgVegatone – 100 C
Sildenafil Citrate Chewable Tablets 100 mg
Each Uncoated Chewable Tablet Contains:
Sildenafil Citrate eq. To Sildenafil 100 mgSildenafil Citrate Tablets 100 Mg Each Film coated Tablet contains: Sildenafil Citrate – mg – eq to sildenafil In House 100 mg
Metalic Red oxide –
Excipients – QSSildenafil Citrate Tablets 100 Mg Each Film coated tablet Contains: Slidenafil Citrate eq to sildenafil In House 100 mg
colour metalic redoxide – QS
Excipient In House – QSUsage: – improve the ability to exercise in adults with pulmonary arterial hypertension
Category: – Generic Drugs & Medicine
Therapeutic category: – Generic Drugs, Erectile Dysfunction
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Silodosin Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the active pharmaceutical ingredient.
Active Ingredient Strength Primary Clinical Function Silodosin INN/Ph.Eur. 8 mg Global Clinical Standard: Maximum adult daily dose for the rapid relief of lower urinary tract symptoms (LUTS) associated with BPH. Excipients Mannitol / Pregelatinized Starch / Sodium Stearyl Fumarate / Hypromellose / Titanium Dioxide Diluent / Binder / Lubricant / Film-Coating (Engineered for highly synchronized gastric dissolution and optimal bioavailability) *Pack Sizes: 10×10 Alu-Alu Blisters (Optimized specifically for high-compliance 30-day urology dispensing regimens).
Simvastatin Tablets
FreeSimvastatin tablets USP
Each Filmcoated tablet contains :
Simvastatin USP 20 mgSimvastatin tablets USP
Each Filmcoated tablet contains :
Simvastatin USP 40 mgUsage: – lower cholesterol
Category: – Anti Cardiovascular Drugs
Therapeutic category: – Antilipidemic, Cardiovascular Agent
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Sirolimus Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Nano-crystal Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute kinetic stability of the sensitive macrolide API.
Active Ingredient Strength Primary Clinical Function Sirolimus USP/Ph.Eur. 1 mg Global Standard: Base titration unit for precise blood-level adjustments in renal transplant recipients. Sirolimus USP/Ph.Eur. 2 mg Standard daily maintenance dose (following initial loading protocols). Excipients Macrogol / Microcrystalline Cellulose / Sucrose / Povidone / Magnesium Stearate Solid Dispersion Carrier / Diluent / Binder (Engineered to drastically enhance the systemic absorption of the highly insoluble API) *Pack Sizes: 10×10 Alu-Alu Blisters or Bottles of 100 (Optimized specifically for strict, life-long transplant adherence regimens).
Sitagliptin Tablet
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets in Alu-Alu or PVC-PVDC blisters.
Active Ingredient Strength (Standard) Role Sitagliptin Phosphate IP/BP/USP 25 mg Renal Adjustment Dose Sitagliptin Phosphate IP/BP/USP 50 mg Intermediate Dose Sitagliptin Phosphate IP/BP/USP 100 mg Standard Adult Dose Excipients Q.S. Core & Coating Sodium Aminosalicylate Granule
FreeSodium Aminosalicylate Granules 80% Each Gram of Enteric Coate Granules Contains : Sodium Aminosaligylate IP (800mg) Quinoline Yellow Lake And Titanium Dioxide IP(-) Excipients(-QS)
Sodium Bicarbonate Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Uncoated Tablet, packed in highly secure, moisture-resistant Alu-Alu blister strips, Alu-PVC, or heavy-gauge HDPE bottles to prevent the highly reactive API from absorbing ambient humidity and effervescing prematurely.
Active Ingredient Strength Primary Clinical Function Sodium Bicarbonate USP/Ph.Eur. 500 mg Standard OTC antacid dose for rapid heartburn and dyspepsia relief. Sodium Bicarbonate USP/Ph.Eur. 650 mg (approx. 10 grains) Global Retail Standard: High-strength OTC dose for severe indigestion and mild urinary alkalinization. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Stearic Acid Diluent / Superdisintegrant / Lubricant (Engineered for immediate gastric dissolution and rapid neutralization) Sodium Bicarbonate, Sodium Citrate, Citric Acid & Tartaric Acid Granules
FreeProduct Composition & Features
We supply this product as a Dry, Free-Flowing Granular Mix, optimized for rapid solubility and a palatable taste.
Active Ingredients Primary Clinical Function Sodium Bicarbonate IP/BP The Alkalizer: Directly provides bicarbonate ions. Sodium Citrate IP/BP The Metabolic Buffer: Slowly converted to bicarbonate in the liver. Citric Acid (Anhydrous) IP/BP The Effervescent Catalyst: Reacts to release $CO_{2}$ and aids solubility. Tartaric Acid IP/BP The Stabilizer: Maintains the granular structure and pH balance. Sodium Chloride Infusions
FreeTechnical & Logistics Specifications
HS Code: 3004.90.99 (Medicaments acting on the circulatory system / Others)
CAS Number: 7647-14-5 (Sodium Chloride)
Dosage Form: Intravenous Infusion (IV)
Packaging: 100 ml / 250 ml / 500 ml / 1000 ml Self-Collapsible LDPE Bags (FFS Technology) or Glass Bottles. Our specialized packaging ensures 36-month stability in Zone IVb tropical climates.
Sodium Picosulfate tablet
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Uncoated or Film-Coated Tablet, packed in highly secure, moisture-resistant PVC/PVDC or Alu-PVC blister strips to ensure the stability of the active pharmaceutical ingredient.
Active Ingredient Strength Primary Clinical Function Sodium Picosulfate BP/Ph.Eur. 10 mg Global Standard: Maximum adult dose for the rapid, overnight relief of severe constipation. Excipients Lactose Monohydrate / Maize Starch / Colloidal Silicon Dioxide / Magnesium Stearate Diluent / Binder / Glidant (Engineered for immediate gastric transit to the lower intestine) Sodium Valproate & Valproic Acid Tablets
FreeProduct Composition & Strength
We supply this product as Controlled Release (CR) / Chrono Tablets to ensure stable blood levels over 24 hours.
Product Strength Sodium Valproate Valproic Acid Total Valproate 200 mg CR 133 mg 58 mg ~200 mg 300 mg CR 200 mg 87 mg ~300 mg 500 mg CR 333 mg 145 mg ~500 mg Enteric Coated 200/500 mg (Sodium Valproate Only) Standard Release Sodium Valproate Tablets
FreeGastro – Resistant Sodium Valproate tablets IP 200 mg
Each enteric coated tablets contains :
Sodium Valproate IP 200 mg
Excipents. QS
Sodium Valproate Gastro – Resistant Tablets IP 300 mg
Each enteric coated tablets contains :
Sodium Valproate IP 300 mg
Excipents. QS
Sodium Valproate Tablets IP 500 mg
Each Enteric- Coated Tablet Contains:
Sodium Valproate IP 500 mgUsage: – treat epilepsy and bipolar disorder
Category: – Hypertensive / Cardiac Drugs
Therapeutic category: – Antiepileptic, Anticonvulsant
Solifenacin Succinate Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu or High-Density Alu-PVC blister strips to ensure the absolute stability of the low-dose API.
Active Ingredient Strength Primary Clinical Function Solifenacin Succinate USP/Ph.Eur. 5 mg Global Standard: Starting and standard maintenance dose for Overactive Bladder; required maximum dose for patients with hepatic/renal impairment. Solifenacin Succinate USP/Ph.Eur. 10 mg High-Dose Therapy: Maximum adult dose for severe, refractory urinary urgency and frequency. Excipients Lactose Monohydrate / Maize Starch / Hypromellose / Magnesium Stearate / Macrogol Diluent / Binder / Premium Film-Coating (Engineered for immediate gastric dissolution and light protection) Sorafenib Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet, packed in highly secure, child-resistant Alu-Alu blister strips or heavy-gauge HDPE bottles to ensure the absolute stability of the tosylate salt formulation.
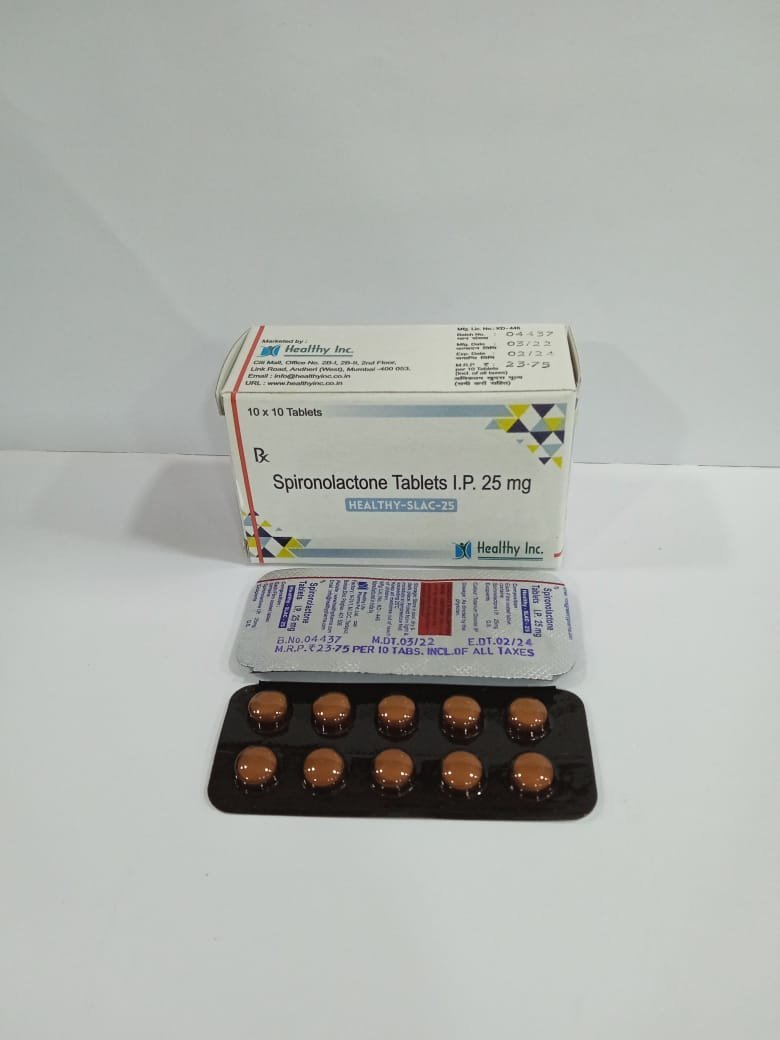
Active Ingredient Strength Primary Clinical Function Sorafenib Tosylate INN/Ph.Eur. 200 mg (Equivalent to Sorafenib free base) Global Standard: Base titration unit for the standard 800 mg daily oncology regimen. Excipients Croscarmellose Sodium / Microcrystalline Cellulose / Hypromellose / Magnesium Stearate Superdisintegrant / Diluent / Film-Coating (Engineered for strict gastric dissolution and reliable systemic bioavailability) Spironolactone Tablets
FreeSpironolactone Tablets IP 25 mg
Each Filmcoated tablet contains :
Spironolactone IP 25 mgSpironolactone Tablets IP
Each Film Coated Contanis:
Spironolactone IP 50 mgUsage: – high blood pressure and heart failure
Category: – Diuretic Drugs
Therapeutic category: – Diuretic
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Stanozolol Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Uncoated or Film-Coated Tablet, packed in highly secure, moisture-resistant and child-resistant Alu-PVC or Alu-Alu blister strips to ensure the stability of the active hormonal ingredient.
Active Ingredient Strength Primary Clinical Function Stanozolol USP/Ph.Eur. 2 mg Clinical Standard: Targeted oral anabolic therapy for the prophylaxis of Hereditary Angioedema (HAE). Excipients Lactose Monohydrate / Microcrystalline Cellulose / Magnesium Stearate / Starch Diluent / Binder / Lubricant (Engineered for immediate gastric dispersion and reliable systemic absorption) Sterile Sulbactam & Cefoperazone veterinary injection
FreeComposition
Sterile Sulbactam & Cefoperazone INJ. 3 GM
Sterile Sulbactam & Cefoperazone INJ. 4.5 GM
Usage: – veterinary injection is commonly used to treat bacterial infections in animals
Category: – veterinary injection
Therapeutic category: – veterinary medicine
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Sterile Water injection
FreeComposition
Contains : Sterile Water For Injection
. 2 ml, 3 ml, 5 ml, 10 ml, 15 ml, 30 ml, 50 ml
Usage: – commonly used to treat bacterial infections
Category: – Sterile Water for Injection
Therapeutic category: – Sterile Water for Injection
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Streptomycin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Dry Powder for Injection (White to Off-White) in glass vials, requiring reconstitution with a provided sterile diluent.
Active Ingredient Base Strength Reconstitution Requirement Streptomycin Sulfate USP/BP 1000 mg (1 g) Requires 3 ml to 4.5 ml of Sterile Water for Injection or 0.9% Normal Saline Excipients None Preservative-Free Pure API *Pack Sizes: Single Vial or Tray of 50/100 Vials. (Often procured in massive bulk for government TB programs).
Sulfasalazine Tablet
FreeProduct Composition & Strength
We supply this product as Enteric Coated (Gastro-Resistant) Tablets to prevent gastric irritation and ensure colonic delivery.
Active Ingredient Strength (Standard) Form Sulfasalazine USP/BP 500 mg Enteric Coated Tablet Excipients Q.S. Cellulose Acetate Phthalate (Coating) Sumatriptan & Naproxen Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Bilayer or Matrix Film-Coated Tablet, packed exclusively in highly secure, child-resistant Alu-Alu or high-density Alu-PVC blister strips to ensure the absolute kinetic stability of both active pharmaceutical ingredients.
Active Ingredient Strength Primary Clinical Function Sumatriptan Succinate USP/Ph.Eur. 85 mg (Equivalent to Sumatriptan base) Rapid-onset 5-HT1 agonism for the immediate abortion of the migraine attack. Naproxen Sodium USP/Ph.Eur. 500 mg Long-acting COX inhibition for sustained neuro-inflammatory blockade and rebound prevention. Excipients Croscarmellose Sodium / Microcrystalline Cellulose / Povidone / Magnesium Stearate Superdisintegrant / Diluent / Binder (Engineered for immediate gastric dissolution and synchronized absorption) Sumatriptan Tablets
FreeA Sumatriptan tablet (often known by the brand name Imitrex) is a prescription medication used to treat acute migraine attacks. It belongs to the triptan class of drugs, which are selective serotonin (5-HT1B/1D) receptor agonists.
Surgical Spirit
FreeProduct Composition & Features
We supply this product as a Clear, Colorless (or Pink/Blue Tinted) Liquid, optimized for clinical efficacy and surface safety.
Component Type Specification Primary Clinical Benefit Active Agent Isopropyl Alcohol (IPA) or Ethanol The Germicide: 70% to 95% v/v concentration. Denaturants Methyl Salicylate / Diethyl Phthalate The Safety: Prevents oral consumption/misuse. Water Content Purified Water q.s. The Facilitator: Water is physically required for protein entry. Action Time 15 to 30 Seconds The Speed: Rapid microbial kill rate. Tacrolimus Tablets
FreeTacrolimus Tablets 1 MG film coated tablet: Anhydrous Tacrolimus 1 MG
Usage: – to prevent rejection of a kidney, heart, or liver transplant
Category: – Generic drugs & Medicine
Therapeutic category: – Generic
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Tadalafil & Dapoxetine Oral Jelly
FreeProduct Composition & Strength
We supply this product as a Single-Dose Liquid Jelly Sachet, optimized for high bioavailability and patient discretion.
Active Ingredient Strength (per 5g sachet) Primary Clinical Function Tadalafil IP/BP 20 mg The Sustainer: Restores erectile function for 36 hours. Dapoxetine IP/BP 60 mg The Controller: Extends time and prevents PE. Excipients Sodium CMC / Glycerin The Vehicle: Ensures stable, non-gritty jelly texture. Flavoring Mixed Fruit / Butterscotch The Palatable Base: Masks chemical bitterness. Tadalafil & Dapoxetine Tablets
FreeA tablet containing Tadalafil 10mg + Dapoxetine 30mg is a combination medication designed to treat two common male sexual health issues: erectile dysfunction (ED) and premature ejaculation (PE).
Tadalafil Oral jelly
FreeProduct Composition & Strength
We supply this product as a Single-Dose Liquid Jelly Sachet, optimized for high bioavailability and portability.
Active Ingredient Strength (per 5g sachet) Primary Clinical Function Tadalafil IP/BP/USP 20 mg The Sustainer: Restores erectile function for 36 hours. Excipients Glycerin / Sodium CMC The Vehicle: Ensures smooth jelly texture and stability. Flavoring Cherry / Mango / Mint / Chocolate The Palatable Base: Masks medicinal bitterness. Tadalafil Tablets
FreeKwiklis -10 Tablets
Tadalafil Tablets 10 mg
Each Filmcoated tablet contains :
Tadalafil 10 mgTHADAAL / TADALAFIL TABLETS BP 20 MG
Each Film coated Tablet Contains:
Tadalafil BP 20 mg
q.s. –Usage: – treat erection problems
Category: – Generic drugs & Medicine
Therapeutic category: – Generic Drugs, Erectile Dysfunction
Tadalista Softgel
FreeProduct Composition & Strength
We supply this product as a Liquid-Filled Soft Gelatin Capsule, optimized for stability and ease of swallowing.
Active Ingredient Strength (per Softgel) Primary Clinical Function Tadalafil IP/BP/USP 20 mg The Sustainer: Restores erectile function for 36 hours. Gelatin Base Pharmaceutical Grade The Shell: Rapidly dissolves in gastric fluid. Solubilizers Vegetable Oil / PEG The Vehicle: Ensures maximum drug bioavailability. Tadarise Oral Jelly
FreeProduct Composition & Strength
We supply this product as a Single-Dose Liquid Jelly Sachet, optimized for high bioavailability and portability.
Active Ingredient Strength (per 5g sachet) Primary Clinical Function Tadalafil IP/BP/USP 20 mg The Sustainer: Restores erectile function for 36 hours. Excipients Glycerin / Sodium CMC The Vehicle: Ensures smooth jelly texture and stability. Flavoring Orange / Strawberry / Pineapple The Palatable Base: Improves patient compliance. Tamoxifen Citrate Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet, packed in highly secure, moisture-resistant Alu-PVC or opaque Alu-Alu blister strips to preserve the integrity of the highly sensitive hormonal API.
Active Ingredient Strength Primary Clinical Use Tamoxifen Citrate USP/Ph.Eur. 10 mg (Equivalent to Tamoxifen Base) Standard divided-dose therapy and specific prophylactic risk-reduction protocols. Tamoxifen Citrate USP/Ph.Eur. 20 mg (Equivalent to Tamoxifen Base) Global Standard: Target once-daily adult maintenance dose for ER+ breast cancer. Excipients Mannitol / Maize Starch / Magnesium Stearate / Croscarmellose Sodium Diluent / Superdisintegrant (Engineered for immediate gastric dispersion and reliable absorption) Tamoxifen Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Uncoated or Film-Coated Tablet, packed in highly secure, moisture-resistant Alu-PVC or opaque Alu-Alu blister strips to preserve the integrity of the highly sensitive hormonal API.
Active Ingredient Strength Primary Clinical Use Tamoxifen Citrate USP/Ph.Eur. 10 mg Standard divided-dose therapy and specific risk-reduction protocols. Tamoxifen Citrate USP/Ph.Eur. 20 mg Global Standard: Target once-daily adult maintenance dose for ER+ breast cancer. Excipients Mannitol / Maize Starch / Magnesium Stearate / Croscarmellose Sodium Diluent / Superdisintegrant (Engineered for immediate gastric dispersion and reliable absorption) Tamsulosin & Dutasteride Capsules
FreeTamsulosin and dutasteride capsules are a combination therapy used to treat moderate to severe symptoms of benign prostatic hyperplasia (BPH). They are most commonly available in a fixed-dose strength of 0.4 mg tamsulosin hydrochloride and 0.5 mg dutasteride.
Tamsulosin & Dutasteride Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Bilayer Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant and child-resistant Alu-Alu blister strips to ensure the absolute stability of both the kinetic polymer matrix and the sensitive hormonal API.
Active Ingredient Strength Primary Clinical Function Tamsulosin Hydrochloride USP/Ph.Eur. 0.4 mg (Modified-Release) Continuous 24-hour uro-selective alpha-1A blockade for immediate symptom relief. Dutasteride USP/Ph.Eur. 0.5 mg Dual 5-alpha-reductase inhibition for profound DHT suppression and prostate volume reduction. Excipients Hypromellose (HPMC) / Butylated Hydroxytoluene (BHT) / Microcrystalline Cellulose / Magnesium Stearate SR Polymer matrix for Tamsulosin / Antioxidant stabilizer for solid-state Dutasteride (Engineered for strict segregation) Tamsulosin & Finasteride Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Bilayer Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant and child-resistant Alu-Alu blister strips to ensure the absolute stability of both the kinetic polymer matrix and the sensitive hormonal API.
Active Ingredient Strength Primary Clinical Function Tamsulosin Hydrochloride USP/Ph.Eur. 0.4 mg (Modified-Release) Continuous 24-hour uro-selective alpha-1A blockade for immediate symptom relief. Finasteride USP/Ph.Eur. 5 mg Targeted Type II 5-alpha-reductase inhibition for profound DHT suppression and prostate shrinkage. Excipients Hypromellose (HPMC) / Microcrystalline Cellulose / Sodium Starch Glycolate / Magnesium Stearate SR Polymer matrix for Tamsulosin / Diluent for Finasteride (Engineered for strict kinetic segregation) Tamsulosin Capsules
FreeTamsulosin capsules are selective alpha-blocker medications used to treat symptoms of an enlarged prostate (Benign Prostatic Hyperplasia). They are most commonly available in a 0.4 mg strength as prolonged-release or sustained-release capsules
Tamsulosin Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Gel-Matrix Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute stability of the hygroscopic polymers.
Active Ingredient Strength Primary Clinical Use Tamsulosin Hydrochloride USP/Ph.Eur. 0.4 mg (Modified-Release Tablet) Premium Global Standard: Target once-daily, food-independent adult dose for BPH. Excipients Macrogol (Polyethylene Glycol) / Magnesium Stearate / Hypromellose / Iron Oxide Advanced Non-Ionic Gel Matrix / Lubricant / Film Coat (Engineered for strict 24-hour continuous diffusion) Teicoplanin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder (Spongy, Ivory to Pale Yellow Cake) in glass vials, accompanied by a specific sterile diluent.
Active Ingredient Strength Reconstitution Requirement Teicoplanin USP/BP/Ph.Eur. 200 mg Requires 3 ml of Sterile Water for Injection Teicoplanin USP/BP/Ph.Eur. 400 mg Requires 3 ml of Sterile Water for Injection Excipients Sodium Chloride / Sodium Hydroxide Isotonicity Agent / pH Adjuster (pH 7.2 to 7.8) *Pack Sizes: 1 Vial of Lyophilized Powder + 1 Ampoule of Diluent (3 ml) per box
Telmisartan & Chlorthalidone Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Bilayer or Matrix Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute stability of both the hygroscopic ARB and the micro-dosed diuretic.
Active Ingredient Strength Primary Clinical Function Telmisartan USP/Ph.Eur. 40 mg Standard ARB maintenance dose for continuous 24-hour peripheral vasodilation and RAAS blockade. Chlorthalidone USP/Ph.Eur. 6.25 mg Highly precise, long-acting thiazide-like diuresis optimized for maximum safety and efficacy. Excipients Meglumine / Sodium Hydroxide / Microcrystalline Cellulose / Sodium Starch Glycolate Alkalizer for Telmisartan / Diluent / Superdisintegrant (Engineered for kinetic segregation and immediate dissolution) Telmisartan & Metoprolol Succinate Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Bilayer Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute stability of both the hygroscopic ARB and the kinetic polymer matrix.
Active Ingredient Strength Primary Clinical Function Telmisartan USP/Ph.Eur. 50 mg Custom-titrated ARB dose for continuous 24-hour peripheral vasodilation and RAAS blockade. Metoprolol Succinate USP/Ph.Eur. 40 mg (Enteric-Coated / ER) Cardioselective beta-1 blockade for continuous heart rate control and anti-anginal defense. Excipients Meglumine / Methacrylic Acid Copolymer (Enteric Coat) / Microcrystalline Cellulose Alkalizer for Telmisartan / Gastric-resistant polymer for Metoprolol (Engineered for strict kinetic segregation) Telmisartan & Metoprolol Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Bilayer Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute stability of both the hygroscopic ARB and the kinetic polymer matrix.
Active Ingredient Strength Primary Clinical Function Telmisartan USP/Ph.Eur. 40 mg Standard ARB maintenance dose for continuous 24-hour peripheral vasodilation. Metoprolol Succinate USP/Ph.Eur. 25 mg (Extended-Release) Low-dose cardioselective beta-1 blockade for gentle heart rate control and initiation therapy. Excipients Meglumine / Hypromellose (HPMC) / Microcrystalline Cellulose / Sodium Hydroxide Alkalizer for Telmisartan / SR Polymer matrix for Metoprolol (Engineered for kinetic segregation) Telmisartan, Amlodipine & Hydrochlorothiazide Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered Multi-Layer or Segregated Matrix Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute stability of the hygroscopic ARB and the sensitive CCB.
Active Ingredient Strength Primary Clinical Function Telmisartan USP/Ph.Eur. 40 mg Continuous 24-hour peripheral vasodilation and potassium-sparing RAAS blockade. Amlodipine Besylate USP/Ph.Eur. 5 mg (Equivalent to Amlodipine base) Calcium channel blockade for direct arterial relaxation and anti-anginal defense. Hydrochlorothiazide (HCTZ) USP/Ph.Eur. 12.5 mg Thiazide diuresis for blood volume reduction and mitigation of peripheral edema. Excipients Meglumine / Microcrystalline Cellulose / Sodium Starch Glycolate / Magnesium Stearate Alkalizer for Telmisartan / Diluent / Superdisintegrant (Engineered for kinetic segregation) *Pack Sizes: 10×10 or 10×15 Alu-Alu Blisters (Optimized specifically for strict 30-day chronic cardiology adherence regimens).
Temazepam Tablets
FreeProduct Composition & Strength
We supply this product as Tablets. (Note: In some markets, capsules are common, but we specialize in the stable tablet dosage form).
Active Ingredient Strength (Standard) Therapeutic Role Temazepam BP/USP 10 mg Elderly / Initiation Dose Temazepam BP/USP 20 mg Standard Adult Dose Temazepam BP/USP 30 mg Severe Insomnia (Short-term) Excipients Q.S. Lactose / Magnesium Stearate Teneligliptin & Metformin Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Bilayer or Matrix Sustained-Release Film-Coated Tablet, packed in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute stability of the highly hygroscopic Metformin component.
Active Ingredient Strength Primary Clinical Function Teneligliptin Hydrobromide Hydrate INN 20 mg Immediate-release DPP-4 inhibition for rapid post-prandial (after-meal) glucose control. Metformin Hydrochloride BP/Ph.Eur. 500 mg (Sustained-Release) Continuous 24-hour hepatic glucose suppression and peripheral insulin sensitization. Excipients Hypromellose (HPMC) / Microcrystalline Cellulose / Magnesium Stearate SR Polymer Matrix / Diluent / Lubricant (Engineered for strict 24-hour kinetic release of Metformin) Teneligliptin Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet, packed in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute stability of the active pharmaceutical ingredient across diverse tropical and high-humidity climates.
Active Ingredient Strength Primary Clinical Use Teneligliptin Hydrobromide Hydrate INN 20 mg Standard Global Dose: Once-daily therapy for glycemic control in Type 2 Diabetes. Excipients Microcrystalline Cellulose / Mannitol / Croscarmellose Sodium / Magnesium Stearate Diluent / Superdisintegrant (Engineered for immediate gastric dissolution and rapid absorption) Terbinafine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Scored Uncoated Tablet, packed in highly secure, moisture-resistant Alu-PVC or opaque Alu-Alu blister strips, optimized for long-term chronic dermatology dispensing regimens.
Active Ingredient Strength Primary Clinical Use Terbinafine Hydrochloride USP/Ph.Eur. 250 mg (Equivalent to Terbinafine Base) Global Standard: Target adult daily dose for Onychomycosis and Tinea Capitis. Excipients Microcrystalline Cellulose / Sodium Starch Glycolate / Colloidal Silicon Dioxide / Magnesium Stearate Diluent / Superdisintegrant / Glidant (Engineered for immediate gastric dissolution and lipophilic API absorption) Terlipressin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder (White to Off-White Cake) in glass vials, requiring reconstitution with a provided sterile diluent.
Active Ingredient Strength Reconstitution Requirement Terlipressin (as Acetate) USP/BP 1 mg Requires 5 ml of 0.9% Sodium Chloride Diluent Excipients Mannitol / Glacial Acetic Acid Bulking Agent / pH Adjustment *Pack Sizes: 1 Vial of Lyophilized Powder + 1 Ampoule of Diluent (5 ml) per box.
Tetrabenazine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Scored Tablet, packed in highly secure, moisture-resistant Alu-PVC or opaque Alu-Alu blister strips, optimized for strict neurological dose titration and chronic dispensing.
Active Ingredient Strength Primary Clinical Use Tetrabenazine INN/Ph.Eur. 25 mg Standard Titration & Maintenance: Scored to allow for precise 12.5 mg micro-dosing during the critical initiation phase. Excipients Lactose / Maize Starch / Magnesium Stearate / Iron Oxide Yellow Diluent / Glidant / Colorant (Engineered for immediate gastric dispersion and precise tablet splitting) Tetracycline Capsules
FreeTetracycline capsules are broad-spectrum antibiotics used to treat various bacterial infections and severe acne. They typically contain Tetracycline Hydrochloride as the active ingredient in strengths of 250 mg or 500 mg.
Theophyline Tablet
FreeTheophyline Tablets 200 mg
Each Uncoated tablet contains :
Theophyline IP 200 mUsage: – Prevent and treat wheezing, shortness of breath, and chest tightness caused by asthma
Category: – Anti Allergic Drugs
Therapeutic category: – Antiasthmatic, Antitussive, Bronchodilator
Thiamine Hydrochloride Tablets
FreeProduct Composition & Strength
We supply this product as Tablets (Uncoated or Film Coated). High-dose variants are essential for therapeutic correction of deficiencies.
Active Ingredient Strength (Standard) Strength (Therapeutic) Primary Role Thiamine HCl USP/BP 10 mg – Dietary Supplement Thiamine HCl USP/BP 50 mg 100 mg Deficiency Treatment Thiamine HCl USP/BP – 250 mg / 300 mg Alcoholism / Neuropathy Excipients Q.S. Lactose / Magnesium Stearate Standard Release Thiamine Injection
FreeComposition
Each mL contains: Thiamine Hydrochloride BP : 100 mg
Water For Injec9on BP : q.s. 1ml
Usage: – used for preventive purposes in certain high-risk groups, such as individuals with chronic illnesses
Category: –Thiamine Injection
Therapeutic category: – Thiamine Injection
Pcd pharma franchise:-
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Thiamine, Pyridoxine & Cyanocobalamin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile, Clear, Red Aqueous Solution in glass ampoules.
Active Ingredient Strength (per 3 ml Ampoule) Primary Function Thiamine Hydrochloride (Vitamin B1) 100 mg Nerve Conduction & Energy Pyridoxine Hydrochloride (Vitamin B6) 100 mg Neurotransmitter Synthesis Cyanocobalamin (Vitamin B12) 1000 mcg (1 mg) Myelin Sheath Repair Excipients Benzyl Alcohol / Water for Injection Preservative / Solvent Base *Pack Sizes: Tray of 5 or 10 Ampoules. (Often presented in larger bulk boxes for clinic use).
Thiocolchicoside Capsules
FreeThiocolchicoside capsules are centrally acting muscle relaxants used primarily to treat painful muscle spasms associated with spinal or musculoskeletal disorders. They are semi-synthetic derivatives of the naturally occurring compound colchicoside.
Thiocolchicoside Injection
FreeProduct Composition & Strength
We supply this product as a Sterile, Clear, Bright Yellow Aqueous Solution in glass ampoules.
Active Ingredient Concentration Total Content & Format Thiocolchicoside BP/Ph.Eur. 2 mg / ml 4 mg in 2 ml Glass Ampoule (Standard) Thiocolchicoside BP/Ph.Eur. 4 mg / ml 8 mg in 2 ml Glass Ampoule (High Dose) Excipients Sodium Chloride / Water for Injection Isotonic Agent / Solvent *Pack Sizes: Tray of 5 or 10 Ampoules.
Thiocolchicoside Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet or Hard Gelatin Capsule, packed in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute stability of the semi-synthetic active ingredient.
Active Ingredient Strength Primary Clinical Use Thiocolchicoside INN/Ph.Eur. 4 mg Initiation / Mild Spasm: Standard adult dose for moderate acute back pain and torticollis (wry neck). Thiocolchicoside INN/Ph.Eur. 8 mg Severe Trauma: Maximum single-dose therapy for severe, immobilizing orthopedic spasms. Excipients Microcrystalline Cellulose / Lactose Monohydrate / Magnesium Stearate Diluent / Lubricant (Engineered for immediate gastric dissolution and rapid onset of relief) Thyroxine Sodium tablets
FreeProduct Composition & Strength
We supply this product as Uncoated Tablets (typically Color-Coded by strength). The wide range of strengths is critical for precise dose titration.
Active Ingredient Strength (Micrograms) Therapeutic Role Thyroxine Sodium USP/BP 12.5 mcg / 25 mcg Pediatric / Elderly Start Thyroxine Sodium USP/BP 50 mcg / 75 mcg / 88 mcg Standard Initiation Dose Thyroxine Sodium USP/BP 100 mcg / 125 mcg / 150 mcg Full Replacement Dose Thyroxine Sodium USP/BP 200 mcg Suppressive Therapy (Cancer) Excipients Q.S. Lactose / Acacia / Magnesium Stearate Diluent / Binder Ticagrelor Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet, packed in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute stability of the highly sensitive active pharmaceutical ingredient across all global climate zones.
Active Ingredient Strength Primary Clinical Use Ticagrelor INN/Ph.Eur. 90 mg Acute & Post-ACS: Standard twice-daily dose for the first 12 months following a heart attack or stent placement. Ticagrelor INN/Ph.Eur. 60 mg Extended Maintenance: Long-term prophylaxis for high-risk patients continuing therapy beyond the initial 12-month period. Excipients Mannitol / Dibasic Calcium Phosphate / Sodium Starch Glycolate / Hypromellose Diluent / Superdisintegrant / Film Coating (Engineered for immediate, highly predictable gastric dissolution) Tigecycline Injection
FreeTigecycline injection is a sterile, lyophilized (freeze-dried) powder containing a first-in-class glycylcycline antibiotic. It is primarily used for complicated skin and intra-abdominal infections, as well as community-acquired bacterial pneumonia.
Tinidazole Tablet
FreeTinidazole Tablets
Each Filmcoated tablet contains :
Tinidazole BP 500 mgUsage: – treat infections caused by protozoa
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic, Anthelmintic, Antiprotozoal, Antiparasitic
Tizanidine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Uncoated Tablet, packed in highly secure, moisture-resistant Alu-PVC or opaque Alu-Alu blister strips, optimized with a deep score-line to allow for exact 1 mg micro-dosing if required by pediatric or highly sensitive geriatric patients.
Active Ingredient Strength Primary Clinical Use Tizanidine Hydrochloride USP/Ph.Eur. 2 mg (Equivalent to Tizanidine base) MANDATORY INITIATION: Dose titration and mild-to-moderate spasm relief. Excipients Anhydrous Lactose / Microcrystalline Cellulose / Colloidal Silicon Dioxide / Stearic Acid Diluent / Glidant / Lubricant (Engineered for immediate gastric dispersion and precise splitting) Tobramycin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile, Clear, Colorless to Pale Yellow Aqueous Solution in glass vials or ampoules.
Active Ingredient Concentration Total Content & Format Tobramycin (as Sulfate) USP/BP 40 mg / ml 80 mg in 2 ml Glass Vial/Ampoule Tobramycin (as Sulfate) USP/BP 10 mg / ml 20 mg in 2 ml Glass Vial (Pediatric Dose) Excipients Sodium Metabisulfite / Edetate Disodium Antioxidant / Chelating Agent (Preservative System) *Pack Sizes: Tray of 5/10 Vials or Box of 50 Ampoules.
Tolperisone Tablets
FreeProduct Composition & Strength
We supply this product as a Film-Coated Tablet, packed in highly secure, moisture-resistant Alu-PVC or opaque Alu-Alu blister strips, optimized for both acute 7-day dispensing and chronic rehabilitative regimens.
Active Ingredient Strength Primary Clinical Use Tolperisone Hydrochloride INN/Ph.Eur. 150 mg Global Standard: Target adult dose for acute spasms and post-stroke spasticity. Excipients Microcrystalline Cellulose / Citric Acid Monohydrate / Stearic Acid / Hypromellose Diluent / Stabilizer / Film Coating (Engineered for immediate gastric dispersion) Tolvaptan Tablets
FreeTolvaptan tablets primarily contain Tolvaptan as the active pharmaceutical ingredient. It is a selective vasopressin V2 receptor antagonist used to treat hyponatremia (low blood sodium levels) and to slow kidney function decline in patients with autosomal dominant polycystic kidney disease (ADPKD)
Topiramate Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet, packed in highly secure, moisture-resistant Alu-Alu blister strips to guarantee absolute stability of the active pharmaceutical ingredient across diverse climate zones.
Active Ingredient Strength Primary Clinical Use Topiramate USP/Ph.Eur. 25 mg Titration & Migraine: Mandatory low-dose initiation to prevent cognitive side effects. Topiramate USP/Ph.Eur. 50 mg Standard Adult Maintenance Dose for chronic migraine prophylaxis. Topiramate USP/Ph.Eur. 100 mg / 200 mg High-dose therapy for refractory epilepsy and generalized tonic-clonic seizures. Excipients Lactose Monohydrate / Pregelatinized Starch / Sodium Starch Glycolate Diluent / Superdisintegrant (Engineered for immediate, uniform gastric dispersion) Torsemide Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Uncoated Tablet, packed in highly secure, moisture-resistant Alu-PVC or opaque Alu-Alu blister strips, providing dosage flexibility for both mild hypertension and severe renal failure.
Active Ingredient Strength Primary Clinical Use Torsemide USP/Ph.Eur. 5 mg / 10 mg Standard maintenance therapy for mild-to-moderate congestive heart failure and essential hypertension. Torsemide USP/Ph.Eur. 20 mg Acute diuresis for severe hepatic cirrhosis and advanced heart failure exacerbations. Torsemide USP/Ph.Eur. 100 mg High-dose therapy specifically reserved for patients with severe chronic renal failure (CRF) and massive fluid overload. Excipients Lactose Monohydrate / Crospovidone / Magnesium Stearate Diluent / Superdisintegrant / Lubricant (Engineered for immediate, predictable gastric dissolution) Tranexamic Acid 250mg Injection
FreeProduct Composition & Strength
We supply this product as a Sterile, Clear, Colorless Aqueous Solution in glass ampoules.
Active Ingredient Concentration Total Content & Format Tranexamic Acid BP/USP 50 mg / ml 250 mg in 5 ml Glass Ampoule Excipients Water for Injection Preservative-Free Formulation *Pack Sizes: Tray of 5 or 10 Ampoules.
Tranexamic Injection
FreeProduct Composition & Strength
We supply this product as a Sterile, Clear, Colorless Aqueous Solution in glass ampoules.
Active Ingredient Concentration Total Content & Format Tranexamic Acid BP/USP 100 mg / ml 500 mg in 5 ml Glass Ampoule Tranexamic Acid BP/USP 100 mg / ml 1000 mg (1 g) in 10 ml Glass Ampoule Excipients Water for Injection Preservative-Free Formulation *Pack Sizes: Tray of 5 or 10 Ampoules.
Tranexamic Tablet
FreeProduct Composition & Strength
We supply this product in film-coated tablets for oral administration.
Active Ingredient Strength Form Tranexamic Acid BP/USP 250 mg Film Coated Tablet Tranexamic Acid BP/USP 500 mg Film Coated Tablet Triamcinolone Acetonide injection
FreeComposition
Triamcinolone Acetonide 40mg/ml ( 1 ML VIAL )
Pcd pharma franchise:-
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Triamcinolone Acetonide Injection
FreeProduct Composition & Strength
We supply this product as a Sterile, White to Off-White Aqueous Suspension in glass vials or ampoules.
Active Ingredient Concentration Total Content & Format Triamcinolone Acetonide USP/BP 10 mg / ml 50 mg in 5 ml Multi-Dose Vial (Intralesional use) Triamcinolone Acetonide USP/BP 40 mg / ml 40 mg in 1 ml Ampoule / Vial (Intra-articular/IM use) Excipients Polysorbate 80 / Carmellose Sodium / Benzyl Alcohol Surfactant / Suspending Agent / Preservative *Pack Sizes: Single Vial, Tray of 5 Vials, or Tray of 10 Ampoules.
Triamcinolone Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Uncoated Tablet, packed in highly secure, moisture-resistant Alu-PVC or opaque Alu-Alu blister strips to preserve the integrity of the steroidal API across high-humidity tropical climates.
Active Ingredient Strength Primary Clinical Use Triamcinolone IP/USP/BP 4 mg Standard Clinical Dose: Maintenance and flare-reduction therapy for severe inflammatory, allergic, and autoimmune disorders. Excipients Lactose Monohydrate / Magnesium Stearate / Microcrystalline Cellulose Diluent / Lubricant (Engineered for optimal compressibility and rapid gastric dispersion) Trifluoperazine Tablet
FreeTrifluoperazine Tablets IP 5 mg
Each Film coated tablet contains:
Trifluoperazine Hydrochloride IP
Eq. To Trifluoperazine 5 mg
Excipients q.sUsage: – treat the symptoms of schizophrenia
Category: – Anti Anxiety drugs
Therapeutic category: – Antipsychotic, Antidepressant, CNS Drugs, Anti Anxiety
Triflupromazine veterinary injection
FreeComposition
Triflupromazine Hydrochloride VET INJ. 5 ML
Usage: –Here are some key points about triflupromazine hydrochloride for veterinary use
Category: – veterinary injection
Therapeutic category: – Additionally, veterinarians
Trigger Capsules
FreeThe term Trigger Capsules is most commonly associated with a pharmaceutical product containing Sildenafil Citrate, used primarily for erectile dysfunction. However, “Trigger” is also used for a specific ayurvedic supplement and a combination heart medication.
Trihexyphenidyl Hydrochloride Tablets
FreeBenzhexol Tablets IP 2 mg
Trihexyphenidyl Hydrochloride tablets IP
Each Uncoated tablet contains:
Benzhexol hydrochloride IP 2 mgBenzhexol Hydrochloride Tablets IP 5 mg
Trihexyphenidyl Hydrochloride Tablets IP
Each Uncoated tablet contains :
Benzhexol Hydrochloride IP 5 mgBenzhexol Tablets BP 2 mg
Trihexyphenidyl Hydrochloride tablets BP 2 mg
Each Uncoated tablet contains:
Benzhexol hydrochloride BP 2 mgBenzhexol Tablets BP 5 mg
Trihexyphenidyl Hydrochloride Tablets
Each Uncoated tablet contains :
Benzhexol Hydrochloride BP 5 mgUsage: – stiffness, tremors, spasms, poor muscle control
Category: – Antiparkinson drugs
Therapeutic category: –
Trimethoprim & Sulphamethoxazole veterinary injection
FreeComposition
Trimethoprim & Sulphamethoxazole VET I/V INJ. 30ML
Usage: –Used to treat respiratory infections in both small and large animals
Category: – veterinary injection
Therapeutic category: – veterinary medicine
Tugain
FreeProduct Composition & Strengths
We supply this product in various concentrations and base formulations (Alcohol-based or Alcohol-free/Propanediol-based).
Active Ingredient Strength Primary Clinical Target Minoxidil IP/BP/USP 2% w/v Female Pattern Hair Loss: Optimized for women. Minoxidil IP/BP/USP 5% w/v Male Pattern Baldness: The standard clinical dose. Minoxidil IP/BP/USP 10% w/v Advanced Alopecia: High-potency specialist use. Solvent Matrix Ethanol / Propylene Glycol The Vehicle: Ensures deep follicular penetration. Ulipristal Acetate Tablets
FreeProduct Composition & Strength
We supply this product as a Film-Coated or Uncoated Tablet, packed in highly secure, moisture-resistant Alu-PVC or opaque Alu-Alu blister strips, optimized for both single-dose retail dispensing and chronic gynecological therapy.
Active Ingredient Strength Primary Clinical Use Ulipristal Acetate INN/Ph.Eur. 30 mg Emergency Contraception: Single-dose therapy taken within 120 hours of unprotected intercourse. Ulipristal Acetate INN/Ph.Eur. 5 mg Uterine Fibroids: Chronic daily dosing for pre-operative management of moderate-to-severe fibroid symptoms. Excipients Lactose Monohydrate / Povidone / Croscarmellose Sodium Diluent / Binder / Superdisintegrant (Ensures rapid gastric dispersion for urgent absorption) Ursodeoxycholic Tablet
FreeProduct Composition & Strength
We supply this product as Film Coated or Sustained Release (SR) Tablets. We use Micronized API for enhanced absorption.
Product Strength Active Ingredient (UDCA) Release Type Common Name 150 mg 150 mg Immediate Release Pediatric / Low Dose 300 mg 300 mg Immediate Release Standard Dose 450 mg 450 mg Sustained Release UDCA SR 600 mg 600 mg Sustained Release High Potency / Forte Vancomycin HCl injection
FreeComposition
Each vial contains :
Vancomycin HCl IP eq to Vancomycin 1000 mg
Each vial contains :
Vancomycin HCl IP eq to Vancomycin 500 mg
Pcd pharma franchise:-
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Vancomycin Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder (White to Off-White Plug/Cake) in glass vials, requiring two-step reconstitution and dilution prior to intravenous administration.
Active Ingredient Strength Initial Reconstitution Requirement Vancomycin Hydrochloride USP/BP 500 mg (0.5 g) Requires 10 ml of Sterile Water for Injection Vancomycin Hydrochloride USP/BP 1000 mg (1.0 g) Requires 20 ml of Sterile Water for Injection Further Dilution (CRITICAL) IV Infusion Matrix Must be further diluted in at least 100 ml to 200 ml of 0.9% Normal Saline or 5% Dextrose before infusing. *Pack Sizes: Single Vial or Tray of 10 Vials.
Varenicline Tablets
FreeProduct Composition & Strength
We supply this product as a Film-Coated Tablet, packed in highly secure, moisture-resistant Alu-Alu blister strips. We specifically offer the globally mandated “Titration Starter Packs” to ensure patient compliance during the critical first two weeks of therapy.
Active Ingredient Strength Primary Clinical Use Varenicline Tartrate INN/USP 0.5 mg Mandatory dose-titration initiation (Days 1 through 7 of the quit attempt). Varenicline Tartrate INN/USP 1 mg Standard Adult Maintenance Dose (Day 8 through Week 12 or 24). Excipients Microcrystalline Cellulose / Anhydrous Dibasic Calcium Phosphate / Croscarmellose Sodium Diluent / Superdisintegrant (Ensures rapid, uniform gastric dispersion) Verapamil Tablet
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets (Immediate Release) or Sustained Release (SR) Tablets.
Product Strength Active Ingredient Release Type Role 40 mg Verapamil HCl 40 mg Immediate Release Low Dose / Titration 80 mg Verapamil HCl 80 mg Immediate Release Standard Dose 120 mg SR Verapamil HCl 120 mg Sustained Release Once/Twice Daily 240 mg SR Verapamil HCl 240 mg Sustained Release High Dose Maintenance Verapamil Tablets
FreeProduct Composition & Strength
We supply this product as Sugar Coated Tablets (Immediate Release) or Film Coated Tablets (Sustained Release – SR).
Active Ingredient Strength (Standard) Therapeutic Role Verapamil HCl USP/BP 40 mg / 80 mg Immediate Release (Arrhythmia) Verapamil HCl USP/BP 120 mg (SR) Sustained Release (Hypertension) Verapamil HCl USP/BP 240 mg (SR) High Dose / Once Daily Excipients Q.S. Sodium Alginate / HPMC SR Matrix Former Vildagliptin & Metformin Tablets
FreeProduct Composition & Strength
We supply this product as a highly advanced Film-Coated Tablet (Available in Immediate or Bilayer Sustained-Release formats), packed strictly in high-barrier Alu-Alu blister strips. Alu-Alu packaging is completely non-negotiable, as Vildagliptin is intensely sensitive to moisture degradation.
Active Ingredient Strength & Format Primary Clinical Use Vildagliptin INN/Ph.Eur. 50 mg (Immediate Release Layer) Rapid dissolution to block DPP-4 and manage post-prandial incretin levels. Metformin Hydrochloride USP/BP 500 mg / 850 mg / 1000 mg (IR or SR) Provides continuous baseline hepatic glucose suppression and peripheral sensitization. Excipients Hypromellose (HPMC) / Microcrystalline Cellulose / Titanium Dioxide SR Polymer Matrix / Diluent / Film-Coating (Engineered for precise kinetic release) Vildagliptin Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Uncoated or Film-Coated Tablet, packed strictly in high-barrier Alu-Alu blister strips to guarantee absolute protection from moisture and environmental degradation.
Active Ingredient Strength Primary Clinical Use Vildagliptin INN/Ph.Eur. 50 mg Standard Adult Dose for monotherapy or adjunctive combination therapy. Excipients Microcrystalline Cellulose / Anhydrous Lactose / Magnesium Stearate Diluent / Lubricant (Engineered for optimal compressibility and rapid gastric dispersion) Vinblastine Sulphate Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder (White to Off-White) or as a Ready-to-Use Liquid Solution in glass vials.
Active Ingredient Strength & Format Reconstitution / Dilution Vinblastine Sulphate USP/BP 10 mg / 10 ml (Liquid) Ready-to-use solution (1 mg/ml). Requires further dilution in 0.9% NaCl before infusion. Vinblastine Sulphate USP/BP 10 mg (Lyophilized Powder) Requires exactly 10 ml of Bacteriostatic 0.9% Sodium Chloride to yield 1 mg/ml. Excipients Sodium Chloride / Water for Injection Isotonicity / Solvent System *Pack Sizes: Single Vial encased in specialized protective cytotoxic shrink-wrap
Vincristine Sulphate Injection
FreeVincristine sulphate injection (often known by the brand Oncovin) is a sterile, preservative-free solution of a vinca alkaloid chemotherapy agent derived from the periwinkle plant. It is a clear, colourless liquid typically supplied in 1 mg or 2 mg vials.
Vitamin A Capsules 100000 IU
FreeVitamin A capsules with a 100,000 IU strength are high-potency supplements typically used to treat severe vitamin A deficiency or as part of large-scale public health immunization programs. Because vitamin A is fat-soluble, these capsules are almost always formulated as soft gelatin capsules containing an oily liquid.
Vitamin A Capsules 200000 IU
FreeVitamin A capsules with a 200,000 IU strength are ultra-high-potency supplements typically used in international health programs to treat severe Vitamin A deficiency (VAD) in children aged 12–59 months. Like the 100,000 IU version, these are almost exclusively soft gelatin capsules containing an oily liquid.
Vitamin A injection Veterinary
FreeProduct Composition & Strength
We supply this sterile oil-based injection in amber glass vials.
Active Ingredient Strength (High Potency) Base Role Vitamin A Palmitate IP/BP/USP 6,00,000 I.U. / mL Oil Base Nutritional Supplement Vitamin E (Alpha Tocopherol) (Optional / Q.S.) Antioxidant Stabilizer & Absorption Aid Ethyl Oleate / Vegetable Oil Q.S. Vehicle Depot Carrier Vitamin B Complex Tablets with Vitamin B12
FreeProduct Composition & Strength
We supply this product as Sugar Coated or Film Coated Tablets (Yellow/Red/Orange) in blister packs or bulk jars.
- Vitamin B1 1mg + Vitamin B2 0.5mg + Vitamin B6 0.5mg + Calcium Pantothenate 1mg + Niacinamide 10mg + Vitamin B12 0.5 mcg
- Vitamin A 800 iu + Cholecalciferol 100 iu + Thiamine Mononitrate vit b1 1.0mg + Riboflavin vit b2 0.5mg + Pyridoxine hydrochloride vit b6 0.5mg + Calcium Pantothenate 1.0mg + Niacinamide 10mg + folic acid 0.1mg
- Thiamine HCL 10mg + Riboflavine 10mg + Pyridoxine HCL 3mg + Cynocobalamine 15mg
- Thiamine HCL 10mg + Riboflavine 10mg + Pyridoxine HCL 3mg + Cynocobalamine 15mg
- Vitamin A 800 IU + Vitamin D 200 IU + Vitamin E 10mg + Vitamin B1 1.4mg + Vitamin B2 1.4mg + Vitamin B6 1.9mg + Vitamin B12 2.6 mcg + Vitamin C 70.0mg + Nicotinamide 18.0mg + Folic Acid
400.0 mcg + Copper Sulphate 2.0mg + Ferrous sulphate 30.0mg + Selenium dioxide 65.0 mcg + Zinc Sulphate 15.0mg + Potassium Iodide 150.0 mcg
- Vitamin b1 1.0mg + vitamin b2 0.5mg + vitamin b6 0.5mg + calcium Pantothenate 1.0mg + Niacinamide 10mg + folic acid 0.1mg
- Vitamin b1 5mg + vitamin b2 5mg + vitamin b6 2mg + Niacinamide 50mg + calcium Pantothenate 5mg
- Thiamine Mononitrate 3mg + riboflavin 1.0mg + pyridoxine hydrochloride 0.5mg + calcium Pantothenate 3mg + Niacinamide 15mg
- Vitamin b1 Mononitrate 0.5mg + vitamin b2 0.5mg + Niacinamide b3 7.5mg
- Thiamine hcl 10mg + riboflavin 10mg + pyridoxine hcl 3mg + Cynocobalamine 15mg
- Vitamin b1 1mg + vitamin b2 0.5mg + vitamin b6 0.5mg + calcium Pantothenate 1.0mg + Niacinamide 10mg + vitamin b12 0.5 mcg
Active Ingredient Strength (B-Complex Forte) Role Vitamin B1 (Thiamine Mononitrate) 10 mg Nerve Function / Energy Vitamin B2 (Riboflavin) 10 mg Cellular Respiration / Vision Vitamin B3 (Niacinamide) 45 mg Skin Health / DNA Repair Vitamin B5 (Calcium Pantothenate) 50 mg Hormone Synthesis Vitamin B6 (Pyridoxine HCl) 3 mg Neurotransmitter Synthesis Vitamin B12 (Cyanocobalamin) 15 mcg RBC Formation / DNA Synthesis Vitamin B-Complex + Folic Acid Tablets
FreeProduct Composition & Strength
We supply this product as Sugar Coated or Film Coated Tablets (typically Yellow or Orange). The coating protects the photosensitive vitamins and masks the characteristic odor.
Active Ingredient Strength (Standard) Therapeutic Role Vitamin B1 (Thiamine Mononitrate) 5 mg / 10 mg Nerve / Carbo Metabolism Vitamin B2 (Riboflavin) 5 mg / 10 mg Skin / Mucosa / Vision Vitamin B6 (Pyridoxine HCl) 2 mg / 3 mg Protein Metabolism Niacinamide (Vitamin B3) 45 mg / 50 mg Cellular Energy / Skin Calcium Pantothenate (Vitamin B5) 50 mg Wound Healing / Hormones Folic Acid (Vitamin B9) 1 mg / 1.5 mg DNA Synthesis / Anemia Excipients Q.S. Talc / Sucrose / Oxide Colors Coating Agents Vitamin B-Complex Tablets
FreeProduct Composition & Strength
We supply this product as Sugar Coated or Film Coated Tablets (typically Yellow, Red, or Brown). The coating protects the photosensitive vitamins and masks the strong odor of B-complex.
Active Ingredient Strength (Standard Forte) Therapeutic Role Vitamin B1 (Thiamine Mononitrate) 10 mg / 100 mg Nerve Function Vitamin B2 (Riboflavin) 10 mg Energy / Vision / Skin Vitamin B6 (Pyridoxine HCl) 3 mg / 100 mg Neurotransmitter Synthesis Vitamin B12 (Cyanocobalamin) 15 mcg / 500 mcg Myelin Repair / RBCs Niacinamide (Vitamin B3) 45 mg / 50 mg Cellular Respiration Calcium Pantothenate (Vitamin B5) 50 mg Coenzyme A Precursor Excipients Q.S. Talc / Shellac / Titanium Dioxide Coating Agents Vitamin B-complex veterinary injection
FreeComposition
Vitamin B-complex (I.V.) INJ. I/V 30 ML VET INJECTION
Vitamin B-complex (I.V.) INJ. I/V 100 ML VET INJECTION
Usage: –B-complex injections are often used to provide nutritional support to animals with deficiencies in these vitamins
Category: – veterinary injection
Therapeutic category: – veterinary medicine
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Vitamin B1 B6 B12 (B-Complex) Injection
FreeProduct Composition & Strength
We supply this product as a Sterile, Clear, Red Aqueous Solution in glass ampoules.
Active Ingredient Strength (per 3 ml Ampoule) Primary Function Vitamin B1 (Thiamine Hydrochloride) 100 mg Nerve Conduction & Energy Vitamin B6 (Pyridoxine Hydrochloride) 100 mg Neurotransmitter Synthesis Vitamin B12 (Cyanocobalamin) 1000 mcg (1 mg) Myelin Sheath Repair Excipients Benzyl Alcohol / Water for Injection Preservative / Solvent Base *Pack Sizes: Tray of 5 or 10 Ampoules. (Often presented in larger bulk boxes for clinic use).
Vitamin B12 (Cyanocobalamin) Tablets
FreeVitamin B12 (Cyanocobalamin) 500 mcg tablets contain cyanocobalamin as the active medicinal ingredient and various inactive excipients used to form the tablet structure. Cyanocobalamin is a synthetic, stable form of Vitamin B12 that is commonly used in dietary supplements to treat or prevent deficiency.
Vitamin B6 Pyridoxine Tablet
FreeProduct Composition & Strength
We supply this product as Uncoated or Film Coated Tablets in blister packs or bulk jars.
Active Ingredient Strength (Standard) Role Pyridoxine Hydrochloride IP/BP/USP 10 mg Maintenance / Prophylaxis Pyridoxine Hydrochloride IP/BP/USP 25 mg / 40 mg Therapeutic Dose Pyridoxine Hydrochloride IP/BP/USP 50 mg / 100 mg High Potency / TB Adjunct Excipients Q.S. Binder / Filler Vitamin K1 Injection
FreeProduct Composition & Strength
We supply this product as a Sterile, Clear, Yellow to Amber Aqueous Dispersion in glass ampoules.
Active Ingredient Strength & Format Target Patient Population Phytomenadione (Vitamin K1) USP/BP 1 mg / 0.5 ml Neonates / Infants: Prophylaxis of VKDB Phytomenadione (Vitamin K1) USP/BP 10 mg / 1 ml Adults: Anticoagulant Antidote / Hypoprothrombinemia Excipients Polyethoxylated Castor Oil / Propylene Glycol Solubilizer System / Co-solvent *Pack Sizes: Tray of 10 Ampoules or Box of 100 Ampoules.
Vitamine Sterile veterinary injection
FreeComposition
Vitamine AD3 E inj. Sterile DE INJ. 10 ML VET INJECTION
Vitamine AD3 E H inj. DE+H INJ. 10 ML VET INJECTION
Usage: – commonly used in animals for various purposes. Here are some common usages
Category: – veterinary injection
Therapeutic category: – performance of animals.
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Voglibose & Metformin Tablets
FreeProduct Composition & Strength
We supply this product as a highly advanced Bilayer Film-Coated Tablet, packed in highly secure, moisture-resistant Alu-Alu blister strips. Alu-Alu packaging is completely non-negotiable, as Voglibose is highly sensitive to moisture degradation.
Active Ingredient Strength & Format Release Profile Function Voglibose USP/BP/JP 0.2 mg / 0.3 mg (Immediate Release Layer) Instantly dissolves in the gut to block carbohydrate absorption during the meal. Metformin Hydrochloride USP/BP 500 mg (Sustained Release Matrix) Slow-release polymer matrix providing continuous 24-hour hepatic suppression. Excipients Hypromellose (HPMC) / Microcrystalline Cellulose SR Polymer Matrix / Diluent (Engineered to control precise dissolution rates) Voglibose Tablet
FreeVoglibose tablets are oral anti-diabetic medications primarily composed of the active ingredient Voglibose, a synthetic alpha-glucosidase inhibitor. They are typically available in strengths of 0.2 mg or 0.3 mg.
Voriconazole Tablets
FreeVoriconazole tablets primarily contain voriconazole as the active antifungal ingredient, typically available in strengths of 50 mg or 200 mg.
Warfarin Tablets
FreeProduct Composition & Strengths
We supply this product in standardized, color-coded strengths to ensure precise dose titration, which is physically critical for this medication.
Active Ingredient Strength Primary Clinical Target Warfarin Sodium IP/BP/USP 1 mg Micro-Titration: For fine-tuned adjustments. Warfarin Sodium IP/BP/USP 2 mg Standard Maintenance: For stable anticoagulation. Warfarin Sodium IP/BP/USP 5 mg High Dose: For initial loading or resistant cases. Water for Injection
FreeProduct Specifications & Quality
We supply WFI that strictly complies with IP/BP/USP standards for parenteral use.
Parameter Specification (USP/BP) Primary Clinical Benefit Purity Sterile & Pyrogen-Free The Safety: Prevents infection and fever. Conductivity < 1.3 The Inertness: No mineral interference. Total Organic Carbon < 500 ppb The Cleanliness: No organic contaminants. Packaging Glass Ampoules / Plastic Vials The Integrity: Tamper-proof and airtight. Zaleplon Capsules
FreeZaleplon capsules (commonly known by the brand name Sonata) are nonbenzodiazepine hypnotics used for the short-term treatment of insomnia. They are formulated to help patients fall asleep quickly.
Zinc Dispersible Tablet
FreeProduct Composition & Strength
We supply this product as Dispersible Tablets (DT), specifically engineered for pediatric ease of use where swallowing whole pills is not possible.
Active Ingredient Strength (Elemental Zinc) Primary Clinical Function Zinc Sulphate Monohydrate 20 mg The Gut Restorer: Essential for diarrhea recovery. Excipients Pharma Grade The Dispersant: Ensures tablet dissolves in seconds. Flavoring Orange / Vanilla / Pineapple The Palatable Base: Ensures pediatric compliance. Zinc Sulphate Tablets
FreeZinc Sulphate tablets 10 mg
Each Filmcoated tablet contains :
Zinc Sulphate Monohydrate BP
Eq. To elemental Zinc 10 mgZinc Sulphate Tablets IP 20 mg
Each Uncoated tablet contains:
Zinc Sulphate IP Eq. To elemental Zinc 20 mgZinc Sulphate Tablets
Each Filmcoated tablet contains :
Zinc Sulphate BP
Eq. To elemental Zinc 50 mgZINC SULPHATE TABLETS USP 20 MG
EACH UNCOATED TABLET CONTAINS:
– ZINC SULPHATE MONOHYDRATE, EQUIVALENT TO ELEMENTAL ZINC IP 20 mgZinc Sulphate Tablet USP
Each Uncoated Dispersible Tablet Contains:
Zinc Sulphate Monohydrate USP
eq. To Elemental Zinc 20 mg Excipients q.s
Approved Colour & Flavour usedZinc Sulfate Tablets IP
Each dispersible tablet contains:
Zinc Sulfate Monohydrate IP
Eq. to elemental Zinc 20 mg
Excipients q.s Approved Flavour & Colour usedZinc Sulphate Tablet USP
Each Uncoated Dispersible Tablet Contains:
Zinc Sulphate Monohydrate USP
eq. To Elemental Zinc 20 mg Excipients q.s
Approved Colour & Flavour usedUsage: – treat and to prevent zinc deficiency
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Zinc Tablet With Oral Rehydration Salts Tablet
FreeProduct Composition & Strength
We supply this as a **Combi-Pack** containing 2 Sachets of ORS and 1 Blister of Zinc DT (10s or 14s).
Component 1: Zinc Dispersible Tablet (DT)
Active Ingredient Strength Form Zinc Sulfate Monohydrate Eq. to Elemental Zinc 20 mg Dispersible Tablet (Fruit Flavor) Component 2: ORS Sachet (WHO Reduced Osmolarity)
Ingredients (Per Sachet) Quantity (g) Role Sodium Chloride 2.6 g Sodium replacement Glucose (Anhydrous) 13.5 g Absorption carrier Potassium Chloride 1.5 g Potassium replacement Trisodium Citrate 2.9 g Corrects Acidosis Total Weight 20.5 g (For 1 Liter Water) Zolpidem Tartrate Tablets
FreeZoldem
Zolpidem Tartrate tablets
Each Filmcoated tablet contains:
Zolpidem Tartrate BP 10 mgUsage: – treat insomnia
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Zonisamide Capsules
FreeZonisamide capsules are antiepileptic medications used as adjunctive therapy for adults and adolescents with partial-onset seizures. They are chemically classified as sulfonamides.
ZyHCG
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder (White Plug/Cake) in glass vials, accompanied by a specific sterile diluent.
Active Ingredient Strength (International Units) Clinical Application Focus Highly Purified hCG USP/BP 2,000 IU Male TRT Adjunct / Luteal Phase Support Highly Purified hCG USP/BP 5,000 IU Standard Ovulation Trigger / Hypogonadism Highly Purified hCG USP/BP 10,000 IU High-Dose IVF Trigger Protocol Reconstitution Requires 1 ml 0.9% Sodium Chloride Isotonic Diluent for IM/SC Injection *Pack Sizes: 1 Vial of Lyophilized Powder + 1 Ampoule of Diluent (1 ml) per box.
α – β Arteether Injection
FreeComposition
α-β Arteether 150 mg / 2ml
Each ml Containsα – β Arteether I.P.………… 75 mg
In Oily Base …………………. Q.S. 1 ml, 2 mlPcd pharma franchise:-
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc