Anti-HIV & ARVs: The Global Mission from India (2026)
In the pharmaceutical landscape of 2026, Anti-HIV medications, or Antiretrovirals (ARVs), represent more than just treatment—they are a global public health priority. India remains the indispensable “Pharmacy of the World” for HIV care, producing over $1.8 Billion worth of ARVs annually and supplying the vast majority of life-saving regimens to the Global South through programs like NACO and the Global Fund.
1. What are ARVs (Antiretrovirals)?
Antiretrovirals (ARVs) are a specialized class of antiviral drugs used to treat HIV (Human Immunodeficiency Virus). While they do not cure HIV, they prevent the virus from replicating in the body. This reduces the viral load to “undetectable” levels, allowing the immune system to recover and preventing the transmission of the virus.
Key 2026 Drug Classes:
Integrase Inhibitors (INSTIs): The current “Gold Standard” (e.g., Dolutegravir). They block the integrase enzyme, which HIV uses to insert its genetic material into human cells.
NRTIs & NNRTIs: (e.g., Tenofovir, Lamivudine, Efavirenz). These block the reverse transcriptase enzyme, stopping the virus from “writing” its code into the cell.
Capsid Inhibitors: (e.g., Lenacapavir). A 2026 breakthrough class that disrupts the virus at multiple stages of its life cycle.
Fixed-Dose Combinations (FDCs): Multiple drugs combined into a single “Three-in-One” pill (like TLD) to simplify treatment and ensure patient compliance.
2. Significance of Indian ARV Manufacturing in 2026
The year 2026 marks a major technological shift in Indian ARV production, moving from daily pills to “Long-Acting” solutions.
Transition to Long-Acting Injectables: India is now a central hub for Long-Acting ARVs that can be administered once every two or six months. This shift from “pill-a-day” to “shot-a-season” is revolutionizing treatment adherence worldwide.
WHO-Prequalification Leadership: Indian facilities lead the world in WHO-Prequalified ARVs. This certification allows Indian manufacturers to participate in massive international tenders, ensuring that high-quality HIV care is affordable even in low-income regions.
Paediatric Innovation: India has pioneered “strawberry-flavored” and “dispersible” ARV tablets for children. Companies like Aurobindo, Hetero, and Mylan (Viatris) have specialized lines to ensure that infants and children receive the precise, palatable doses they need.
PrEP & PEP Dominance: Beyond treatment, India is the primary producer of PrEP (Pre-Exposure Prophylaxis). Affordable generic PrEP from India is the backbone of global HIV prevention strategies in 2026.
3. Why Healthy Inc. is Your Strategic ARV Sourcing Partner
Sourcing ARVs requires a partner who understands the high stakes of Data Integrity and Sterile Compliance. Healthy Inc. serves as your technical gateway to India’s premier ARV units.
Strategic Sourcing Hub: We are associated with multiple state-of-the-art, WHO-GMP and USFDA compliant manufacturing units. Whether you need the latest Dolutegravir combinations or specialized Pediatric ARVs, we match you with the facility holding the best regulatory track record.
Pharmacist-Led Technical Vetting: We provide “straight answers” on Bioequivalence (BE) studies and dissolution profiles. Our team vets every batch to ensure the exact therapeutic window is maintained, which is critical for preventing viral resistance.
Regulatory & Dossier Mastery: We simplify international registration by providing full CTD/ACTD dossiers, stability data for Zone IVb (hot/humid climates), and all necessary certifications for global health tenders.
Future-Ready Portfolio: Through our network, we provide access to 2026-era innovations, including long-acting injectables and capsid inhibitors, giving your brand a competitive edge in the evolving mental and public health market.
Showing all 18 results
Enalapril Tablets
FreeEnalapril Maleate Tablets IP 2.5 mg
Each Uncoated Tablet Contains:
Enalapril Maleate IP 2.5 mgENALAPRIL MALEATE TABLETS IP 5 MG
Each uncoated Tablet Contains:
Enalapril Maleate IP 5 mg Excipients q.s.Enalapril Maleate tablets IP 10 mg
Each Uncoated Tablet contains :
Enalapril maleate IP 10 mgOELPRIL -5 ENALAPRIL TABLETS BP 5 MG EACH UNCOATED TABLET CONTAINS : ENALAPRIL MALEATE BP 5 MG
Oepril – 10
Enalapril Tablets USP
Each Uncoated tablet contains :
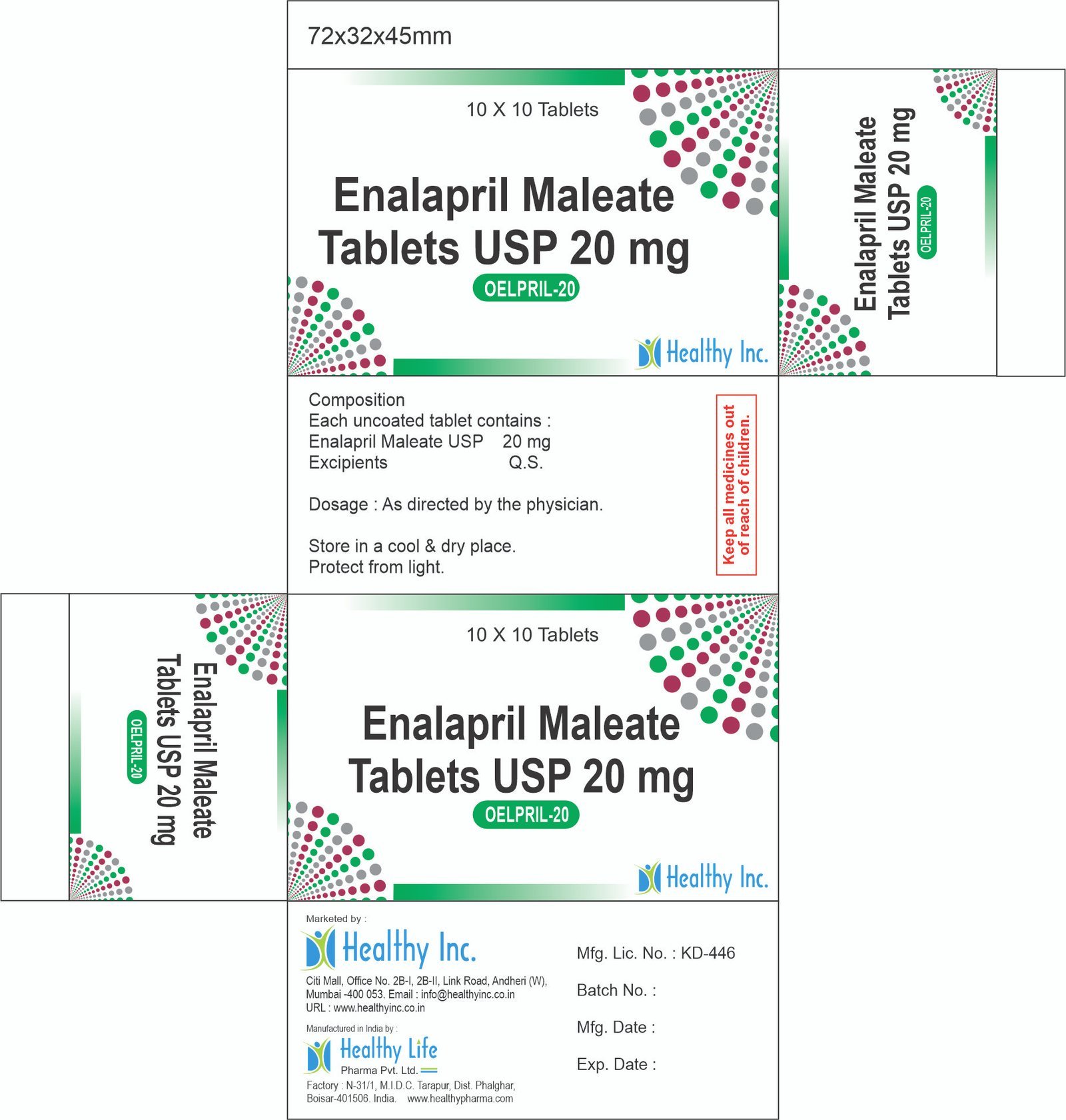
Enalapril Maleate USP 10 mgOepril – 20
Enalapril Maleate Tablets USP
Each Uncoated tablet contains :
Enalapril Maleate USP 20 mg.Usage: – Reduce high blood pressure and to prevent or treat heart failure
Category: – Anti Hypertensive / Cardiac Drugs
Therapeutic category: – Antihypertensive