Description
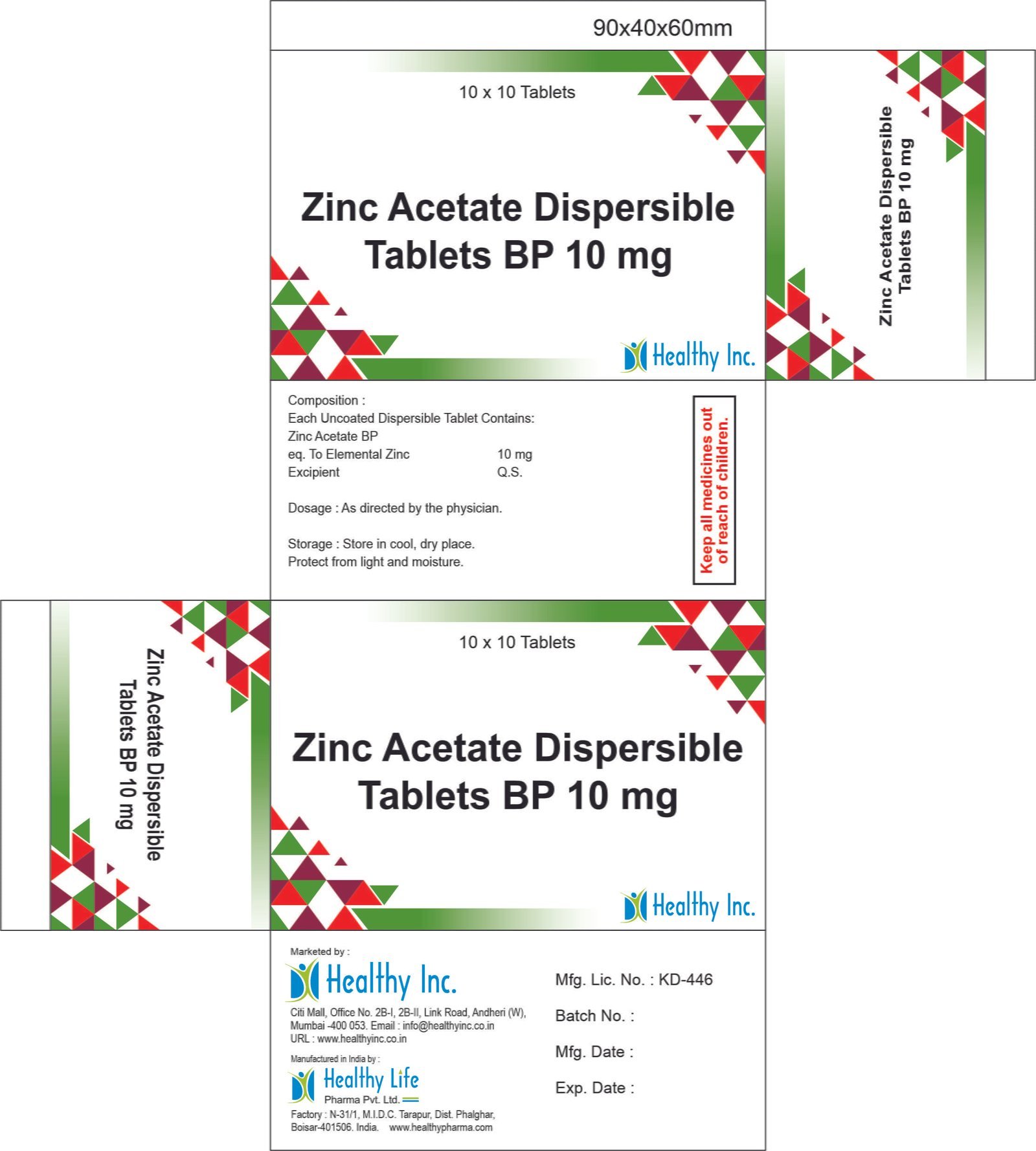
Zinc Dispersible Tablet
Zinc Sulphate Dispersible Tablets (20 mg)
Healthy Inc is a specialized global supplier and exporter of advanced pediatric, gastrointestinal, and public health therapeutics. We provide ultra-high-purity, kinetically stable Zinc Dispersible Tablets, manufactured in WHO–GMP certified, high-precision pharmaceutical facilities. This “Essential Trace Mineral” is a massive-volume strategic export to National Health Programs, NGO relief agencies (UNICEF/WHO/Red Cross), and international B2B distributors, serving as the globally preferred intervention for reducing childhood mortality related to diarrheal diseases.
Product Overview
This formulation operates as a “Cellular Integrity Restorer.” It ensures patient safety by physically strengthening the gut barrier and chemically activating the enzymes required for immune defense.
Mechanism 1 (Intestinal Epithelial Repair): Zinc physically promotes the Regeneration of Enterocytes (gut cells). This chemically improves the absorption of water and electrolytes, physically firming the stool and reducing the frequency of diarrhea.
Mechanism 2 (Immune Modulation): It physically assists in the production of T-lymphocytes and Natural Killer cells. This chemically enhances the body’s ability to clear the underlying infection (bacterial or viral) that triggered the diarrhea.
Mechanism 3 (Enzyme Activation): Zinc acts as a physical cofactor for over 300 enzymes involved in protein synthesis and cell division, chemically ensuring rapid recovery and growth following a period of illness.
Product Composition & Strength
We supply this product as Dispersible Tablets (DT), specifically engineered for pediatric ease of use where swallowing whole pills is not possible.
| Active Ingredient | Strength (Elemental Zinc) | Primary Clinical Function |
| Zinc Sulphate Monohydrate | 20 mg | The Gut Restorer: Essential for diarrhea recovery. |
| Excipients | Pharma Grade | The Dispersant: Ensures tablet dissolves in seconds. |
| Flavoring | Orange / Vanilla / Pineapple | The Palatable Base: Ensures pediatric compliance. |
Technical & Logistics Specifications
HS Code: 3004.50.39 (Medicaments containing vitamins/minerals / Others)
CAS Number: 7446-19-7 (Zinc Sulphate Monohydrate)
Dosage Form: Dispersible Oral Tablet
Packaging: 10 Tablets per Blister in high-barrier Alu-Alu (Cold-Form Foil). Our specialized packaging ensures 36-month stability in Zone IVb tropical climates.
Manufacturing Authority & Compliance
Rapid-Dispersible Engineering: Pediatric patients physically cannot swallow whole tablets. Our manufacturing process utilizes Super-Disintegrant Technology, physically ensuring the tablet dissolves completely in 5ml of water or breast milk in under 60 seconds.
Precise Elemental Calibration: Because elemental Zinc levels are physically critical, we utilize Automated High-Precision Granulation, physically ensuring that every 20mg dose is chemically accurate to protect against deficiency or overdose.
Moisture-Barrier Stability: Zinc Sulphate is physically hygroscopic (absorbs moisture). We utilize Dehumidified Production Suites and Alu-Alu blister packaging, physically preventing chemical degradation in high-humidity export markets.
Primary Indications
Pediatrics: Management of Acute and Persistent Diarrhea (used in conjunction with Oral Rehydration Salts).
Public Health: Treatment and prevention of Zinc Deficiency in malnourished populations.
Immunology: Adjunctive therapy for respiratory infections and wound healing.
Usage Instructions
Dissolution: Place the tablet physically in a small spoon with 5ml of clean water or breast milk.
Administration: Allow it to physically dissolve completely (do not crush) before giving it to the child.
The 14-Day Protocol: It is physically critical to take Zinc once daily for 14 consecutive days, even if the diarrhea stops sooner. This chemically ensures the body’s Zinc stores are replenished to prevent physical relapse.
Safety Warning: For Pediatric/Adult Use. Vomiting Note: If the child physically vomits within 30 minutes of taking the dose, the dose should be chemically repeated. Copper Note: Long-term high-dose Zinc can physically interfere with copper absorption. Contraindication: Do not use in patients with known hypersensitivity to Zinc Sulphate or its components.
Global Export & Contract Manufacturing
Healthy Inc is a premier Pediatric and Public Health Pharmaceutical Exporter in India. As a verified Medicine Supplier in Mumbai, we offer flexible Third Party Manufacturing (Contract Manufacturing) for high-volume Zinc Dispersible Tablets. Whether you are a National Ministry of Health in Africa or a B2B Pharma Marketplace partner in the CIS or SE Asia, we ensure secure, WHO-GMP compliant delivery.
Commercial Inquiries:
WhatsApp/Call: +91 7710003340
Email: info@healthyinc.co.in