Showing all 17 results
Amlodipine & Enalapril Maleate Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets to protect the Enalapril Maleate from moisture-induced degradation and to ensure the physical integrity of the combination.
Active Ingredient Strength (Standard) Therapeutic Role Amlodipine (as Besylate) BP/USP 5 mg / 10 mg Calcium Channel Blocker (CCB) Enalapril Maleate BP/USP 5 mg / 10 mg / 20 mg ACE Inhibitor Excipients Q.S. Sodium Stearyl Fumarate / Lactose Lubricant & Stabilizer Amlodipine Besylate & Losartan Potassium Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets. The formulation is optimized for 24-hour efficacy, allowing for convenient once-daily dosing.
Active Ingredient Strength (Standard) Therapeutic Role Amlodipine Besylate BP/USP 5 mg Calcium Channel Blocker (CCB) Losartan Potassium BP/USP 50 mg Angiotensin Receptor Blocker (ARB) Excipients Q.S. Microcrystalline Cellulose / Starch Stabilizing Core Matrix Atenolol & Chlorthalidone Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets. The combination is engineered to maintain therapeutic plasma levels over a 24-hour period, supporting once-daily dosing.
Active Ingredient Strength (Standard) Therapeutic Role Atenolol BP/USP 50 mg / 100 mg Cardioselective Beta-Blocker Chlorthalidone BP/USP 12.5 mg / 25 mg Long-acting Diuretic Excipients Q.S. Magnesium Stearate / Povidone Stabilizing Core Matrix Hyoscine Butylbromide Tablets
FreeProduct Composition & Strength
We supply this product as Sugar Coated Tablets or Film Coated Tablets. The coating is essential to mask the naturally bitter taste of the active ingredient and ensure patient compliance.
Active Ingredient Strength (Standard) Therapeutic Role Hyoscine Butylbromide BP/USP 10 mg Muscarinic Antagonist (Antispasmodic) Excipients Q.S. Calcium Hydrogen Phosphate / Maize Starch Stabilizing Core Matrix Coating Agent Q.S. Sucrose / Talc / Acacia Traditional Sugar Coating Isoniazid & Pyridoxine Hydrochloride Tablets
FreeProduct Composition & Strength
We supply this product as Uncoated or Film Coated Tablets. The ratio of Isoniazid to Pyridoxine is carefully calibrated to meet international clinical guidelines for neuroprotection.
Active Ingredient Strength (Standard) Therapeutic Role Isoniazid BP/USP 300 mg Primary Antitubercular (Bactericidal) Pyridoxine Hydrochloride BP/USP 10 mg / 25 mg / 50 mg Neuroprotective Vitamin (B6) Excipients Q.S. Starch / Lactose / Magnesium Stearate Tablet Core Matrix Isoniazid & Pyridoxine Hydrochloride Tablets
FreeProduct Composition & Strength
We supply this product as Uncoated or Film Coated Tablets. The ratio of Isoniazid to Pyridoxine is carefully calibrated to meet international clinical guidelines for neuroprotection.
Active Ingredient Strength (Standard) Therapeutic Role Isoniazid BP/USP 300 mg Primary Antitubercular (Bactericidal) Pyridoxine Hydrochloride BP/USP 10 mg / 25 mg / 50 mg Neuroprotective Vitamin (B6) Excipients Q.S. Starch / Lactose / Magnesium Stearate Tablet Core Matrix Isosorbide Mononitrate Tablets
FreeProduct Composition & Strength
We supply this product as Immediate Release Tablets (20 mg). We also offer Extended Release (ER/SR) variants for once-daily dosing through our contract manufacturing division.
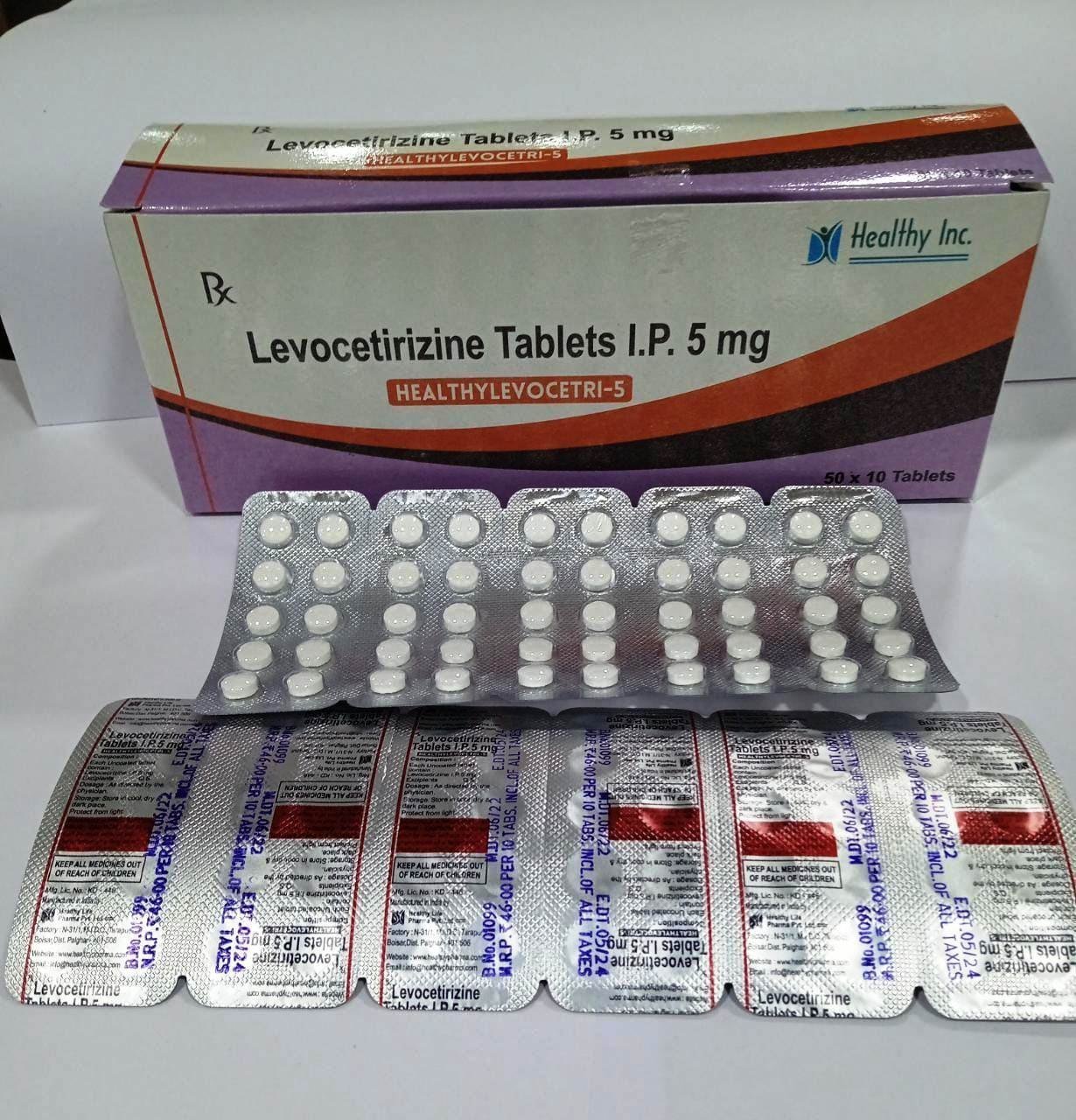
Active Ingredient Strength (Standard) Therapeutic Role Isosorbide Mononitrate BP/USP 20 mg Organic Nitrate / Vasodilator Excipients Q.S. Microcrystalline Cellulose / Lactose Stabilizing Matrix Levocetirizine Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets. The small, easy-to-swallow tablets are engineered for rapid disintegration to facilitate quick relief from acute allergic flares.
Active Ingredient Strength (Standard) Therapeutic Role Levocetirizine Dihydrochloride BP/USP 5 mg Potent H1-Receptor Antagonist Excipients Q.S. Microcrystalline Cellulose / Lactose Diluent & Disintegrant Coating Agent Q.S. Opadry® White Protective Film Coating Norfloxacin Tablets
FreeNorfloxacin Tablets IP 400 mg
Each Film Coated Tablet Contains:
Norfloxacin IP 400 mgFlonor Tablets
Norfloxacin Tablets BP 400 mg
Each Filmcoated tablet contains :
Norfloxacin BP 400 mgUsage: – Treat a variety of bacterial infections
Category: – Antibiotic Drugs
Therapeutic category: – Antibiotic
Rifampicin & Isoniazid Tablets
FreeTechnical & Manufacturing Specifications
Formulated for chemical stability and precise bioavailability of both active ingredients.
Technical Metric Specification Standard Active Composition Rifampicin (150mg/300mg) + Isoniazid (75mg/150mg) Dosage Form Film-Coated Tablet HS Code 3004.20.19 (Anti-TB Medicaments) Packaging Alu-Alu Blister (Crucial for Rifampicin stability) Shelf Life 24 to 36 Months (Subject to stability data) Rifampicin Isoniazide Pyrizinamide Tablets
FreeRifampicin, Isoniazide and Pyrizinamide Tab (Combi Pack)
(Rifampicin Tablets IP) + (Isoniazide Tablets IP) + (Pyrizinamide Tablets IP)
Each film Coated Tablet Contains :
Rifampicin IP 100 mg
Each coated Tablet Contains :
Isonoazide IP 50 mg
Each coated Tablet contains :
Pyrizinamide IP 250 mgRIP TABLETS
(Rifampicin, Isoniazid And Pyrazinamide Tablets Int.Ph.)
Each Filmcoated Tablet Contains:
Rifampicin BP/Int.Ph. 150 mg
Isoniazid BP/Int.Ph. 75 mg
Pyrazinamide BP/Int.Ph. 400 mgUsage: – treat tuberculosis (TB) infection
Category: – Antibiotic drugs
Therapeutic category: – Antibiotic, Antituberculosis
Rifampicin, Isoniazid, Pyrazinamide & Ethambutol Tablets
FreeProduct Composition & Strength
We supply this product as Film Coated Tablets (usually Brown or Reddish-Brown). We offer both the “Daily Regimen” and “Intermittent Regimen” strengths.
Active Ingredient Strength (WHO Daily FDC) Therapeutic Role Rifampicin IP/BP/USP 150 mg Sterilizing / Broad Spectrum Isoniazid IP/BP/USP 75 mg Early Bactericidal Pyrazinamide IP/BP/USP 400 mg Intracellular Sterilizing Ethambutol HCl IP/BP/USP 275 mg Resistance Prevention