Showing all 7 results
Enalapril Tablets
FreeEnalapril Maleate Tablets IP 2.5 mg
Each Uncoated Tablet Contains:
Enalapril Maleate IP 2.5 mgENALAPRIL MALEATE TABLETS IP 5 MG
Each uncoated Tablet Contains:
Enalapril Maleate IP 5 mg Excipients q.s.Enalapril Maleate tablets IP 10 mg
Each Uncoated Tablet contains :
Enalapril maleate IP 10 mgOELPRIL -5 ENALAPRIL TABLETS BP 5 MG EACH UNCOATED TABLET CONTAINS : ENALAPRIL MALEATE BP 5 MG
Oepril – 10
Enalapril Tablets USP
Each Uncoated tablet contains :
Enalapril Maleate USP 10 mgOepril – 20
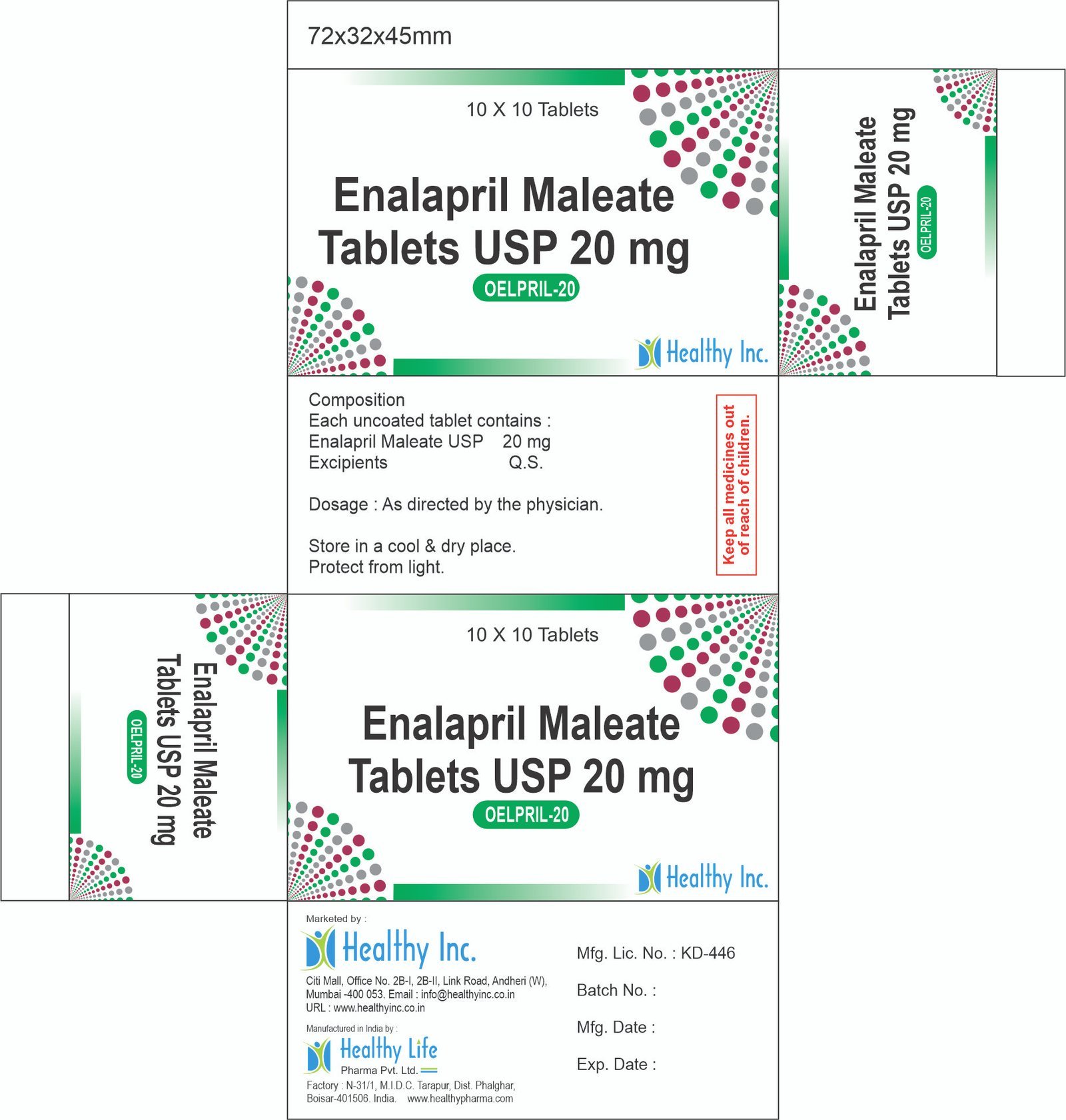
Enalapril Maleate Tablets USP
Each Uncoated tablet contains :
Enalapril Maleate USP 20 mg.Usage: – Reduce high blood pressure and to prevent or treat heart failure
Category: – Anti Hypertensive / Cardiac Drugs
Therapeutic category: – Antihypertensive
Imidapril Hydrochloride Tablets
FreeIMAPRIL TABLETS 5 MG
IMIDAPRIL HYDROCHLORIDE 5 MG
EACH FILM COATED TABLET CONTAINS :
IMIDAPRIL HYDROCHLORIDE 5 MGIMAPRIL TABLETS 10 MG
IMIDAPRIL HYDROCHLORIDE 10 MG
EACH FILM COATED TABLET CONTAINS :
IMIDAPRIL HYDROCHLORIDE 10 MGUsage: – reduce high blood pressure
Category: – Hypertensive / Cardiac Drugs
Therapeutic category: – Hypertensive
METOPROLOL + RAMIPRIL TABLETS
FreeProduct Composition & Strength
We supply this product as a Precision-Engineered, API-Segregated Bilayer Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the highly degradable Ramipril molecule.
Active Ingredient Strength Primary Clinical Function Metoprolol (as Succinate/Tartrate) Ph.Eur./USP 25 mg / 50 mg Cardioselective Base: Reduces heart rate, cardiac output, and myocardial oxygen demand; protects against sudden cardiac death. Ramipril Ph.Eur./USP 2.5 mg / 5 mg ACE Inhibitor Driver: Forces profound systemic vasodilation, protects the kidneys (renoprotection), and halts pathological cardiac remodeling. Excipients Microcrystalline Cellulose / Sodium Stearyl Fumarate / Hypromellose / Sodium Bicarbonate / Iron Oxide Colors Low-Moisture Diluent / Lubricant / Film-Coating / Polymeric Stabilizer (Engineered specifically to physically isolate the acidic Ramipril from cross-reacting with the Metoprolol salt, preventing the formation of toxic diketopiperazine impurities) *Pack Sizes: 10×10 Alu-Alu Blisters (Optimized specifically for strict 30-day chronic cardiology dispensing regimens).
RAMIPRIL & HYDROCLORTHIAZIDE TABLETS
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet or Capsule, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the highly degradable Ramipril molecule.
Active Ingredient Strength Primary Clinical Function Ramipril Ph.Eur./USP 5 mg (or 2.5 mg / 10 mg) ACE inhibition for vasodilation, RAAS blockade, and critical potassium retention. Hydrochlorothiazide Ph.Eur./USP 12.5 mg / 25 mg Global Clinical Standard: Thiazide diuretic for sodium excretion and sustained blood volume reduction. Excipients Microcrystalline Cellulose / Sodium Starch Glycolate / Sodium Bicarbonate / Hypromellose / Magnesium Stearate Diluent / Superdisintegrant / Alkaline Stabilizing Agent (Engineered specifically to protect Ramipril from acidic degradation and moisture-induced cyclization) *Pack Sizes: 10×10 Alu-Alu Blisters or 3×10 Unit-Dose Blisters (Optimized specifically for strict, high-compliance 30-day chronic cardiology dispensing regimens).
Ramipril and Amlodipine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Capsule or Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the highly degradable Ramipril molecule.
Active Ingredient Strength Primary Clinical Function Ramipril Ph.Eur./USP 5 mg / 10 mg ACE inhibition for vasodilation, neurohormonal blockade, and venous pressure equalization. Amlodipine (as Besilate) Ph.Eur./USP 5 mg / 10 mg Global Clinical Standard: Sustained CCB for profound, 24-hour arterial vasodilation. Excipients Microcrystalline Cellulose / Crospovidone / Sodium Stearyl Fumarate / Hypromellose / Iron Oxide Red Diluent / Superdisintegrant / Alkaline Stabilizing Lubricant (Engineered to protect Ramipril from acidic degradation and moisture) *Pack Sizes: 10×10 Alu-Alu Blisters or 3×10 Unit-Dose Blisters (Optimized specifically for strict, high-compliance 30-day chronic cardiology dispensing regimens).
Ramipril Tablets
FreeRamipril Tablets IP 2.5mg
Each Film coated tablet contains:
Ramipril IP 2.5 mgRamipril tablets IP 5 mg
Each Filmcoated tablet contains :
Ramipril IP 5 mgUsage: – high blood pressure and heart failure
Category: – Anti Cardiovascular drugs
Therapeutic category: – Cardiovascular Agent, Antihypertensive
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc