Dolutegravir Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Polymorphically Stabilized Microdose Tablet, packed exclusively in highly secure, moisture-resistant heavy-gauge HDPE bottles or Alu-Alu blister strips to ensure the absolute chemical survival of the Dolutegravir Sodium salt.
Active Ingredient Strength Primary Clinical Function Dolutegravir (as Sodium) USP/Ph.Eur. 50 mg The Global INSTI Anchor: The definitive therapeutic payload required to force immediate, systemic viral suppression and maintain an undetectable viral load. Excipients D-Mannitol / Microcrystalline Cellulose / Sodium Starch Glycolate / Povidone K-30 / Sodium Stearyl Fumarate / Premium Polymeric Film Diluent / Binder / Superdisintegrant / Glidant / Heavy Film-Coating (Engineered specifically utilizing strict low-moisture granulation to prevent the active Dolutegravir Sodium from undergoing polymorphic shifts during compression, guaranteeing absolute pharmacokinetic bioequivalence) *Pack Sizes: Heavy-Gauge HDPE Bottles of 30 or 10×10 Alu-Alu Blisters (Optimized specifically for massive NGO distribution and chronic daily dispensing regimens).
Efavirenz ,Emtricitabine & Tenofovir Disoproxil Fumarate Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Hydrolytically Shielded Mega-Dose FDC Tablet, packed exclusively in highly secure, moisture-resistant heavy-gauge HDPE bottles to ensure the absolute chemical survival of the moisture-sensitive Tenofovir and light-sensitive Efavirenz.
Active Ingredient Strength Primary Clinical Function Efavirenz USP/Ph.Eur. 600 mg The NNRTI Anchor: The massive, highly lipophilic non-nucleoside payload required to force immediate, systemic viral suppression. Emtricitabine (FTC) USP/Ph.Eur. 200 mg The Cytidine Analogue: Provides profound, long-acting intracellular viral suppression to reinforce the backbone. Tenofovir Disoproxil Fumarate (TDF) USP/Ph.Eur. 300 mg The Nucleotide Backbone: The heavy-hitting chain terminator required to completely halt viral DNA elongation. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Hydroxypropyl Cellulose / Sodium Lauryl Sulfate / Magnesium Stearate / Premium Polymeric Film Diluent / Superdisintegrant / Binder / Surfactant / Heavy Film-Coating (Engineered utilizing High-Compression Bi-Layer Technology to fuse 1,100mg of active API into a single, aerodynamically shaped tablet that remains easy for patients to swallow, heavily coated to lock out destructive tropical humidity) *Pack Sizes: Heavy-Gauge HDPE Bottles of 30 (Optimized specifically for massive NGO distribution and strict 30-day chronic daily dispensing regimens).
Efavirenz Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, High-Density Macrodose Tablet, packed exclusively in highly secure, moisture-resistant heavy-gauge HDPE bottles or Alu-Alu blister strips to ensure the absolute chemical survival of the highly lipophilic active ingredient.
Active Ingredient Strength Primary Clinical Function Efavirenz USP/Ph.Eur. 600 mg The Global NNRTI Macrodose: The massive therapeutic payload required to force immediate, systemic viral suppression when combined with a dual-NRTI backbone. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Sodium Lauryl Sulfate / Povidone K-30 / Magnesium Stearate / Premium Polymeric Film Diluent / Superdisintegrant / Surfactant / Binder / Heavy Film-Coating (Engineered specifically utilizing powerful surfactants like Sodium Lauryl Sulfate to drastically increase the solubility of the notoriously lipophilic Efavirenz molecule, ensuring rapid and complete gastrointestinal absorption) *Pack Sizes: Heavy-Gauge HDPE Bottles of 30 or 10×10 Alu-Alu Blisters (Optimized specifically for massive NGO distribution and chronic daily dispensing regimens).
Emtricitabine & Tenofovir Disoproxil Fumarate Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Hydrolytically Shielded Macrodose FDC Tablet, packed exclusively in highly secure, moisture-resistant heavy-gauge HDPE bottles or Alu-Alu blister strips to ensure the absolute chemical survival of the moisture-sensitive Tenofovir.
Active Ingredient Strength Primary Clinical Function Emtricitabine (FTC) USP/Ph.Eur. 200 mg The Cytidine Analogue: Provides profound, long-acting intracellular viral suppression with an exceptionally favorable safety profile. Tenofovir Disoproxil Fumarate (TDF) USP/Ph.Eur. 300 mg
(Eq. to 245 mg Tenofovir Disoproxil)The Nucleotide Backbone: The heavy-hitting reverse transcriptase inhibitor required to anchor the ARV regimen and prevent viral breakthrough. Excipients Microcrystalline Cellulose / Croscarmellose Sodium / Lactose Monohydrate / Magnesium Stearate / Premium Polymeric Film (Opadry) Diluent / Superdisintegrant / Binder / Lubricant / Heavy Film-Coating (Engineered specifically utilizing dry-granulation and heavy film-coating to protect the API from atmospheric moisture, ensuring the massive 500mg active payload remains structurally intact and easily swallowable) *Pack Sizes: Heavy-Gauge HDPE Bottles of 30 (Optimized specifically for massive NGO distribution and strict 30-day chronic daily dispensing regimens).
Indinavir sulphate Capsules
FreeIndinavir sulphate capsules contain the active antiviral drug and several inactive ingredients used to stabilize the medication for the treatment of HIV-1 infection.
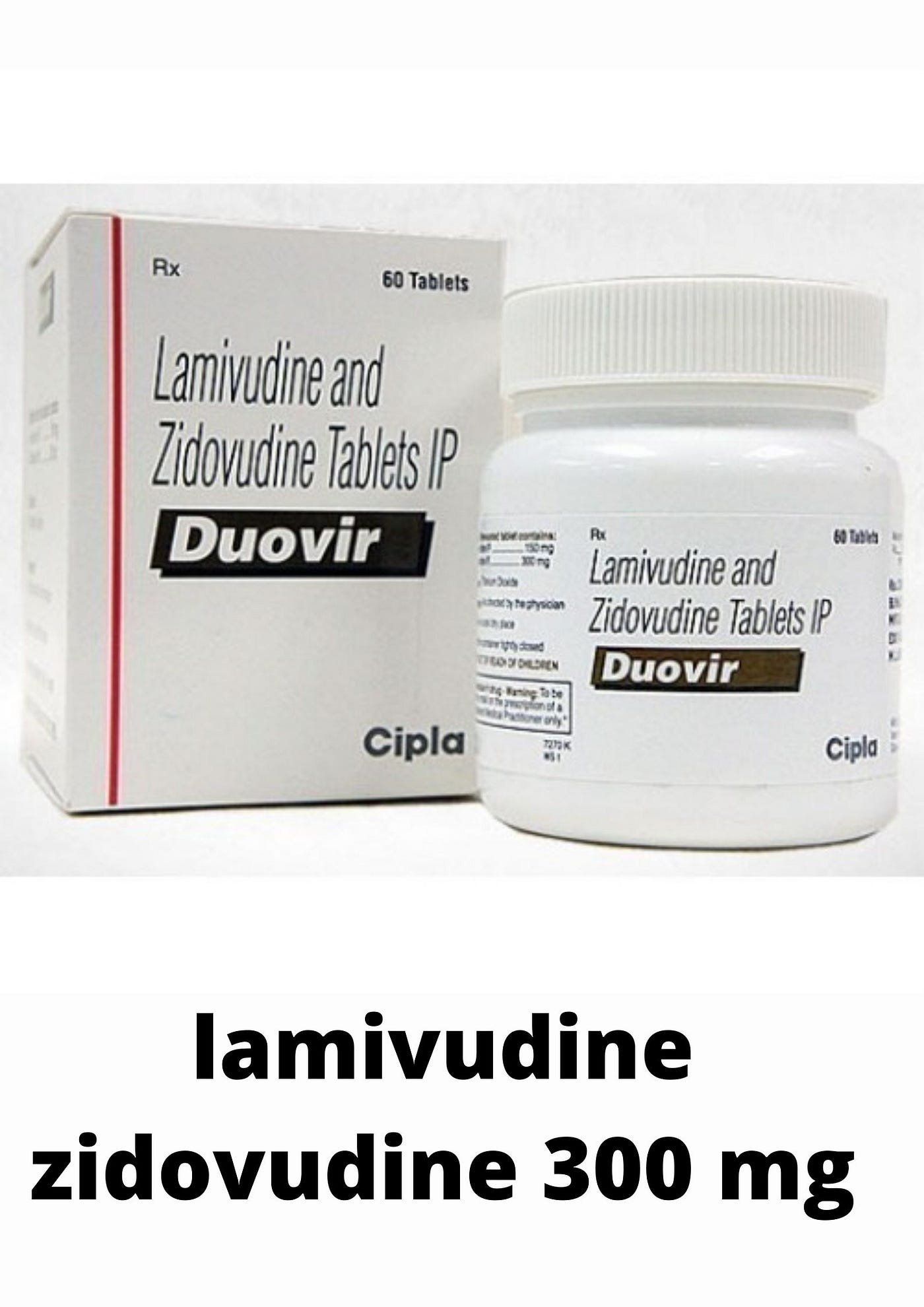
Lamivudine & Zidovudine tablets
FreeLamivudine Tablets IP 100 mg
Each Filmcoated tablet contains :
Lamivudine IP 100 mg exceipents Q.S.Lamivudine Tablets IP 150 mg
Each Filmcoated tablet contains :
Lamivudine IP 150 mgLamivudine 150 mg + Zidovudine 300 mg + Nevirapine 200 mg tablets IP Each Film Coated Tablets Contains: Lamivudine IP 150 mg Zidovudine IP 300 mg Nevirapine IP 200 mg Excipients Q.S.
Lamuzy Tablets Lamivudine and Zidovudine Tablets IP
Each Film Coated tablet contains :
Zidovudine IP 300 mg
Lamivudine IP 150 mgCirolam 150 Tablets
Lamivudine Tablets 150 mg
Each Film Coated tablet contains :
Lamivudine USP 150 mgUsage: – Hepatitis B infection
Category: – HIV Anti Viral drugs
Therapeutic category: – Antiviral, Anti HIV, Hepatitis Treatment
Nevirapine Tablets
FreeNevirapine tablets IP 200 mg
Each Film Coated Tablet Contains:
Nevirapine IP 200 mgUsage: – Prevents human immunodeficiency virus (HIV)
Category: – HIV Anti viral Drugs
Therapeutic category: – Antiviral, Anti HIV
Tenofovir 300mg + Lamivudine 300mg +Dolutegravir 50mg tablets
FreeA fixed-dose combination tablet of Tenofovir (300mg) + Lamivudine (300mg) + Dolutegravir (50mg), often abbreviated as TLD, is a first-line antiretroviral therapy (ART) used to treat HIV-1 infection.
Showing all 8 results