Showing all 3 results
Clonazepam Tablets
FreeClonazepam Tablets IP 0.5 mg
Each Uncoated Tablet contains :
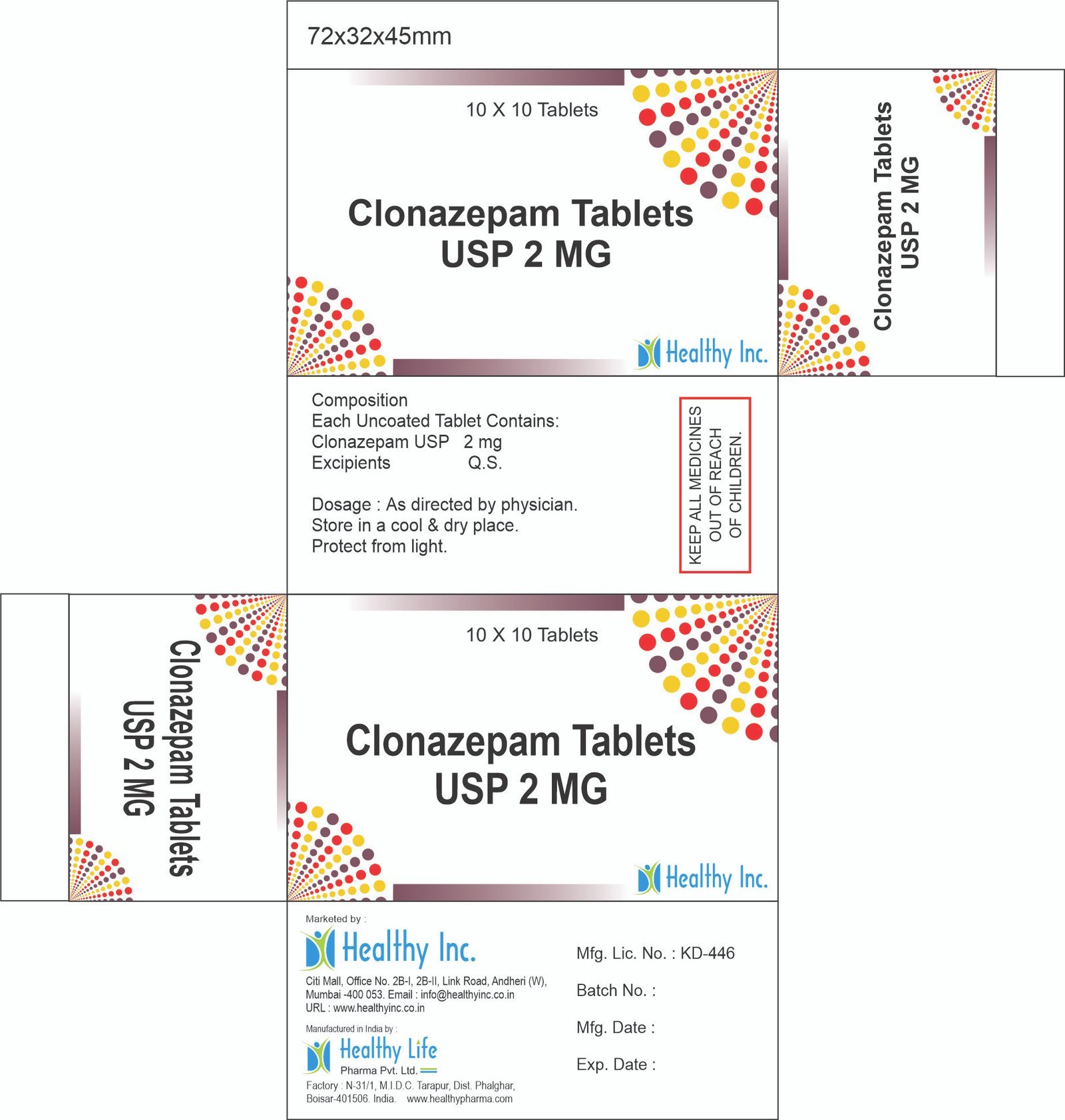
Clonazepam IP 0.5 mgClonazepam Tablets USP 2 MG
Each Uncoated Tablet Contains:
Clonazepam USP 2 mgUsage: – Control seizures or fits due to epilepsy, involuntary muscle spasms, panic disorder and sometimes restless legs syndrome
Category: – Anti Hypertensive / Cardiac drugs
Therapeutic category: – Anti convulsant , Antiepileptic