Showing all 9 results
Amlodipine besylate with Losartan potassium Tablets
FreeAmlodipine Tablets IP 5 mg
Each Uncoated tablet contains :
Amlodipine Besylate IP
Eq to Amlodipine 5 mgAmlodipine Tablets IP 10 mg
Each Uncoated Tablet Contains :
Amlodipine Besylate IP
Eq To Amlodipine 10 mgAmlodipine Besylate Tablets IP 2.5 mg
Each uncoated tablet contains :
Amlodipine Besylate IP
Eq. To Amlodipine 2.5 mgAmlodipine Besylate Tablets IP 2.5 mg
Each Film Coated Tablet Contains:
Amlodipine Besylate IP
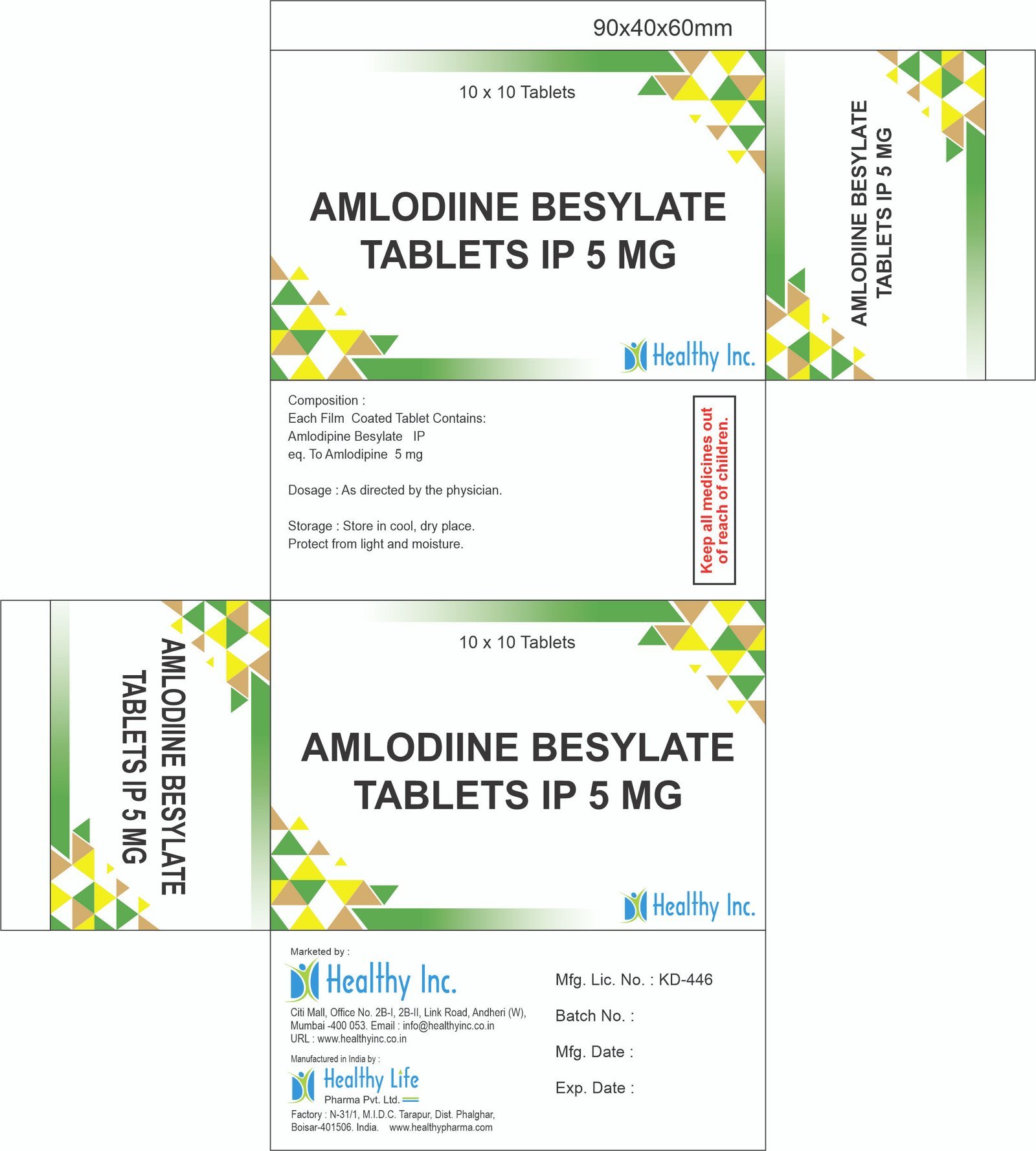
eq. To Amlodipine 2.5 mgAmlodipine Besylate Tablets IP 5 mg
Each Film Coated Tablet Contains:
Amlodipine Besylate IP
eq. To Amlodipine 5 mgAMLODIPINE & ENALAPRIL MALEATE TABLET
Each Film Coated Tablet Contains :
Amlodipine Besylate
Eq. To Amlodipine 5 mg
Enalapril Maleate 5 mg
Excipients q.sAMLODIPINE & LISINOPRIL TABLET
Each Film Coated Tablet Contains :
Amlodipine Besylate
Eq. To Amlodipine 5 mg
Lisinopril dihydrate
Eq. To Lisinopril 5 mg
Excipients q.sTRIPPLEACE AMLODIPINE -10 / AMLODIPINE
TABLETS 10 MG
Each uncoated tablet contains:
Amlodipine Besilate BP
Equivalent to Amlodipine 10 mg
Excipients Q.S.Usage: – Treat high blood pressure
Category: – Hypertensive / Cardiac Drugs
Therapeutic category: – hypertensive, Cardiovascular Agent,
Atenolol with Chlorthalidone tablets.
Freetenolol Tablets IP 25 mg Each Uncoated Tablet
contains :- Atenolol IP 25 mg
Excipients – QSAtenolol Tablets IP 50 mg
Each Uncoated Tablet contains :
Atenolol IP 50 mg
Excipients q.sOtenol 50
Atenolol Tablets BP
Each Uncoated tablet contains :
Atenolol BP 50 mgOtenol
Atenolol Tablets BP
Each Uncoated tablet contains :
Atenolol BP 100 mgUsage: – Treat high blood pressure and irregular heartbeat
Category: – Hypertensive Cardiac drugs
Therapeutic category: – Cardiovascular Agent,Anti hypertensive
Clindipine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Light-Shielded Cardiovascular Tablet, packed exclusively in highly secure, opaque Alu-Alu blister strips to ensure the absolute chemical stability of the highly photosensitive active ingredient.
Active Ingredient Strength Primary Clinical Function Cilnidipine USP/Ph.Eur. 5 mg The Geriatric & Titration Anchor: The definitive starting dose utilized for elderly patients or those with mild essential hypertension to prevent sudden hypotensive episodes. Cilnidipine USP/Ph.Eur. 10 mg The Global Maintenance Standard: The standardized daily therapeutic payload utilized for the vast majority of adult patients requiring chronic 24-hour blood pressure control. Cilnidipine USP/Ph.Eur. 20 mg The Severe Refractory Macrodose: Utilized strictly for severe, uncontrolled hypertension where the 10mg dose has failed to achieve target blood pressure metrics. Excipients Microcrystalline Cellulose / Lactose Monohydrate / Croscarmellose Sodium / Hypromellose / Magnesium Stearate / Opaque Polymeric Film (Titanium Dioxide base) Diluent / Densifier / Superdisintegrant / Binder / Lubricant / Protective Light Shield (Engineered specifically utilizing a dense, titanium dioxide-heavy film coat. Cilnidipine is intensely sensitive to UV and visible light; this opaque coating guarantees 100% of the active molecule survives to reach the patient’s systemic circulation) *Pack Sizes: 10×10 Opaque Alu-Alu Blisters (Optimized specifically for strict 30-day chronic cardiology dispensing regimens).
Metoprolol Tablets
FreeMETOPROLOL TARTRATE TABLET IP Each Tablet Contains: Metoprolol tartrate IP 50 MG
XYPROL – T TABLETS
Metoprolol Tartrate Tablet IP
Each Tablet Contains:
Metoprolol Tartrate IP 50 mgUsage: – Treat high blood pressure
Category: – Anti Cardiovascular drugs
Therapeutic category: – Cardiovascular Agent, Anti hypertensive
Nebivolol Hydrochlorothiazide Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended, Dual-Active Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu or high-density PVC/PVDC blister strips to ensure the absolute chemical stability of the active ingredients.
Active Ingredient Strength Primary Clinical Function Nebivolol (as HCl) Ph.Eur./USP 5 mg Cardioselective & Vasodilatory Base: Reduces cardiac workload and provides active Nitric Oxide-driven peripheral vasodilation. Hydrochlorothiazide (HCTZ) Ph.Eur./USP 12.5 mg / 25 mg Thiazide Diuretic Driver: Forces the excretion of excess sodium and water, massively amplifying the blood-pressure-lowering effect of the beta-blocker. Excipients Lactose Monohydrate / Microcrystalline Cellulose / Croscarmellose Sodium / Polysorbate 80 / Magnesium Stearate / Premium Opadry Film Diluent / Superdisintegrant / Wetting Surfactant / Moisture-Barrier Coating (Engineered specifically to protect the highly delicate racemic Nebivolol balance while ensuring rapid, synchronized gastric dissolution of the diuretic) *Pack Sizes: 10×10 Blisters or 28/30-Tablet Calendar Packs (Optimized specifically for strict 30-day chronic cardiology dispensing regimens).
Olmesartan Medoxomil Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the highly volatile ester prodrug.
Active Ingredient Strength Primary Clinical Function Olmesartan Medoxomil Ph.Eur./USP 20 mg Initiation / Global Standard: Standard adult starting and maintenance dose for the continuous control of essential hypertension. Olmesartan Medoxomil Ph.Eur./USP 40 mg Severe Refractory Standard: Maximum adult maintenance dose for patients requiring further reduction in blood pressure. Excipients Microcrystalline Cellulose / Lactose Monohydrate / Hydroxypropyl Cellulose / Magnesium Stearate / Titanium Dioxide Low-Moisture Diluent / Binder / Lubricant / Premium Moisture-Barrier Film-Coating (Engineered specifically to lock out ambient humidity and prevent the premature hydrolysis of the medoxomil group prior to ingestion) *Pack Sizes: 10×10 Alu-Alu Blisters or 3×10 Unit-Dose Blisters (Optimized specifically for strict 30-day chronic cardiology dispensing regimens).
RAMIPRIL & HYDROCLORTHIAZIDE TABLETS
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Film-Coated Tablet or Capsule, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the highly degradable Ramipril molecule.
Active Ingredient Strength Primary Clinical Function Ramipril Ph.Eur./USP 5 mg (or 2.5 mg / 10 mg) ACE inhibition for vasodilation, RAAS blockade, and critical potassium retention. Hydrochlorothiazide Ph.Eur./USP 12.5 mg / 25 mg Global Clinical Standard: Thiazide diuretic for sodium excretion and sustained blood volume reduction. Excipients Microcrystalline Cellulose / Sodium Starch Glycolate / Sodium Bicarbonate / Hypromellose / Magnesium Stearate Diluent / Superdisintegrant / Alkaline Stabilizing Agent (Engineered specifically to protect Ramipril from acidic degradation and moisture-induced cyclization) *Pack Sizes: 10×10 Alu-Alu Blisters or 3×10 Unit-Dose Blisters (Optimized specifically for strict, high-compliance 30-day chronic cardiology dispensing regimens).
Ramipril and Amlodipine Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Capsule or Film-Coated Tablet, packed exclusively in highly secure, moisture-resistant Alu-Alu blister strips to ensure the absolute chemical stability of the highly degradable Ramipril molecule.
Active Ingredient Strength Primary Clinical Function Ramipril Ph.Eur./USP 5 mg / 10 mg ACE inhibition for vasodilation, neurohormonal blockade, and venous pressure equalization. Amlodipine (as Besilate) Ph.Eur./USP 5 mg / 10 mg Global Clinical Standard: Sustained CCB for profound, 24-hour arterial vasodilation. Excipients Microcrystalline Cellulose / Crospovidone / Sodium Stearyl Fumarate / Hypromellose / Iron Oxide Red Diluent / Superdisintegrant / Alkaline Stabilizing Lubricant (Engineered to protect Ramipril from acidic degradation and moisture) *Pack Sizes: 10×10 Alu-Alu Blisters or 3×10 Unit-Dose Blisters (Optimized specifically for strict, high-compliance 30-day chronic cardiology dispensing regimens).