Showing all 8 results
Esomeprazole Capsule
FreeEsomeprazole capsules primarily contain Esomeprazole Magnesium (as a dihydrate or trihydrate) as the active ingredient. Because esomeprazole is “acid-labile” (broken down by stomach acid), these capsules are formulated as delayed-release or gastro-resistant systems containing enteric-coated pellets.
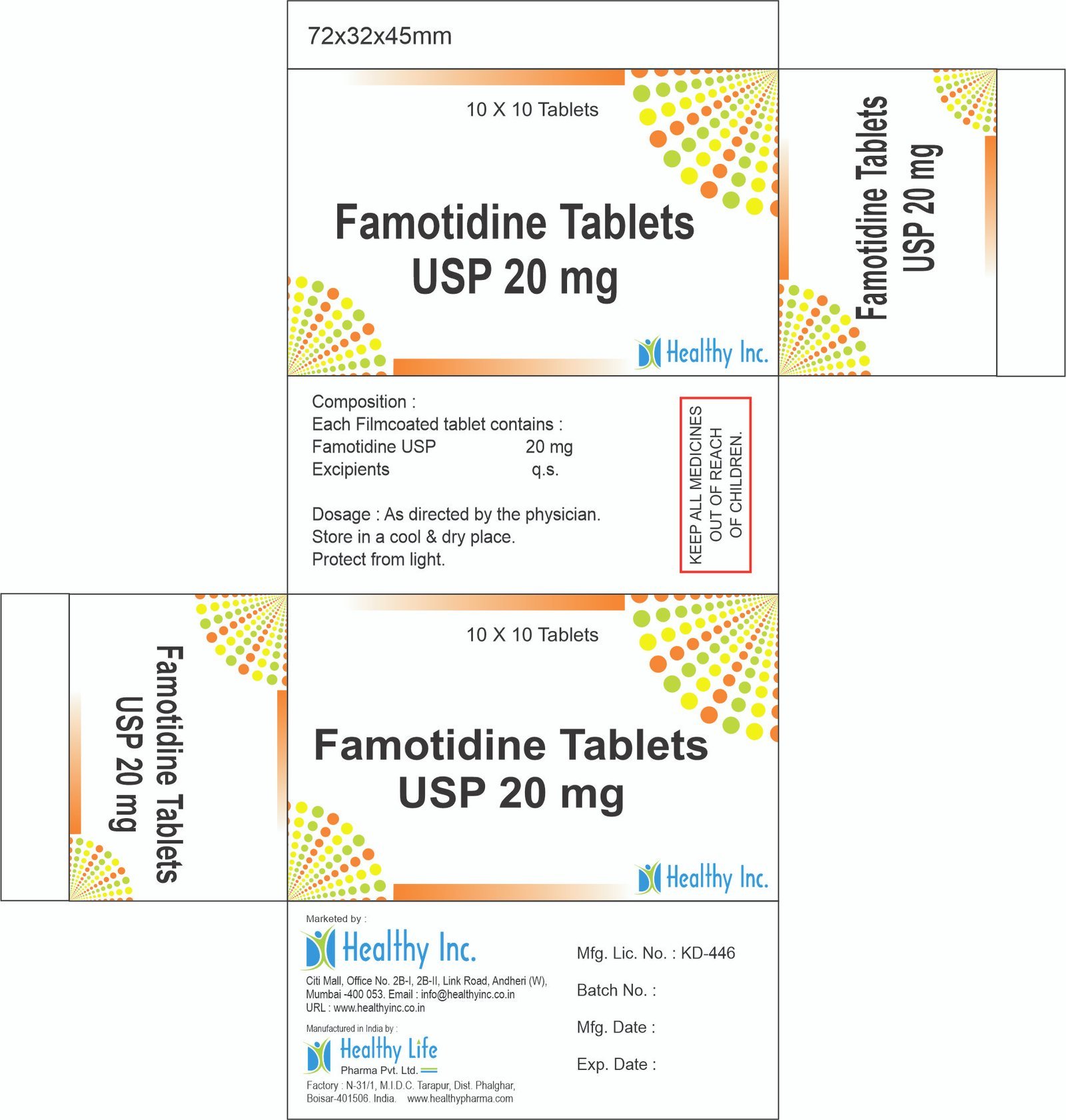
Famotidine Tablets
FreeFamotidine Tablets USP 20 mg
Each Filmcoated tablet contains :
Famotidine USP 20 mgUsage: – Prevent and treat heartburn due to acid indigestion and sour stomach
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Omeprazole Capsules
FreeOmeprazole capsules typically contain enteric-coated pellets to prevent the stomach’s acid from breaking down the medication before it can be absorbed in the small intestine.
Omeprazole Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder (White to Off-White Cake) in clear glass vials, typically accompanied by a specialized sterile solvent.
Active Ingredient Strength Reconstitution Requirement Omeprazole Sodium USP/BP 40 mg Requires provided 10 ml solvent or specific diluent Excipients Sodium Hydroxide / Disodium Edetate Alkalizing Agent / Stabilizer *Pack Sizes: Single Vial + 10 ml Solvent Ampoule, or Tray of 10 Vials.
Pantoprazole Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder (White to Off-White Cake) in clear glass vials.
Active Ingredient Strength Reconstitution Requirement Pantoprazole Sodium USP/BP 40 mg Requires 10 ml of 0.9% Sodium Chloride Excipients Sodium Hydroxide / Edetate Disodium Alkalizing Agent / Stabilizer *Pack Sizes: Single Vial or Tray of 10 Vials. (Often packed with a 10 ml ampoule of 0.9% NaCl diluent).
Propanthaline Tablets
FreeXyproline Tablets
Propanthaline Tablets IP 15 mg
Each Coated tablet contains :
Propanthaline Bromide IP 15 mgUsage: – treat ulcers
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Rabeprazole Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder (White to Pale Yellow Cake) in clear glass vials.
Active Ingredient Strength Reconstitution Requirement Rabeprazole Sodium USP/BP 20 mg Requires 5 ml of Sterile Water for Injection Excipients Mannitol / Sodium Hydroxide Bulking Agent / Alkalizing Stabilizer *Pack Sizes: Single Vial or Tray of 10 Vials. (Often packed with a 5 ml diluent ampoule).
Ranitidine Injection
FreeProduct Composition & Strength
We supply this product as a Sterile, Clear, Aqueous Solution in glass ampoules.
Active Ingredient Concentration Total Content & Format Ranitidine Hydrochloride USP/BP 25 mg / ml 50 mg in 2 ml Glass Ampoule Excipients Phenol / Potassium Di-hydrogen Phosphate Preservative / pH Buffer System (pH 6.7 to 7.3) *Pack Sizes: Tray of 10 Ampoules, 50 Ampoules, or 100 Ampoules.