Showing all 4 results
Methylprednisolone Sodium Succinate Injection
FreeProduct Composition & Strength
We supply this product as a Sterile Lyophilized Powder requiring reconstitution with the provided solvent.
Active Ingredient Strength Standard Use Methylprednisolone Sod. Succ. 40 mg Local Injection / Minor Allergic Reaction Methylprednisolone Sod. Succ. 125 mg Acute Asthma / Status Asthmaticus Methylprednisolone Sod. Succ. 500 mg / 1000 mg (1 gm) Pulse Therapy (Lupus / Transplant Rejection) Diluent Bacteriostatic Water Provided with Act-O-Vial or Separate Ampoule *Pack Sizes: Single Vial with Diluent.
Salbutamol & Theophylline Tablets
FreeProduct Composition & Strength
We supply this product as a Precision-Blended Tablet, packed exclusively in highly secure, moisture-resistant Alu-PVC or Alu-Alu blister strips to ensure the absolute chemical stability of both active pharmaceutical ingredients.
Active Ingredient Strength Primary Clinical Function Salbutamol Sulphate BP/Ph.Eur. 2 mg (Equivalent to Salbutamol base) Rapid-onset beta-2 agonism for immediate smooth muscle relaxation. Theophylline (Anhydrous) BP/Ph.Eur. 100 mg Global Clinical Standard: PDE inhibition for sustained bronchodilation and mild respiratory anti-inflammation. Excipients Microcrystalline Cellulose / Maize Starch / Purified Talc / Magnesium Stearate Diluent / Binder / Glidant / Lubricant (Engineered for strict, synchronized gastric dissolution) *Pack Sizes: 10×10 Blisters (Optimized specifically for high-compliance 30-day chronic pulmonology dispensing regimens).
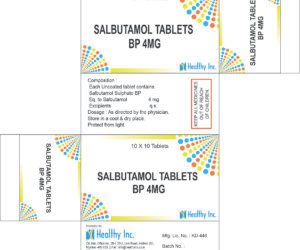
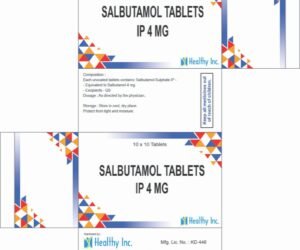
Salbutamol Tablets
FreeSalbutamol Tablets IP 2 mg Each Uncoated Tablet contains: Salbutamol Sulphate IP – Equivalent to Salbutamol 2 mg
Excipients – QSSalbutamol Tablets IP 4 mg Each uncoated tablets contains: Salbutamol Sulphate IP –
Equivalent to Salbutamol 4 mg
Excipients – QSSalbutamol Tablets BP
Each Uncoated tablet contains:
Salbutamol Sulphate BP
Eq. to Salbutamol 4 mgUsage: – treat wheezing and shortness of breath caused by breathing problems
Category: – Antacid & Anti ulcer drugs
Therapeutic category: – Antiasthmatic