Thyroid hormones are essential pharmaceuticals used to manage metabolic disorders and hormone deficiencies. In 2026, they remain a cornerstone of endocrine therapy, particularly in India, where thyroid dysfunction affects nearly 42 million people. These drugs function by mimicking or regulating the natural hormones—Thyroxine ($T_{4}$) and Triiodothyronine ($T_{3}$)—produced by the thyroid gland to control the body’s energy expenditure.
1. Major Classifications of Thyroid Drugs
In a professional pharmaceutical context, medications are divided into replacement therapies and antithyroid agents:
| Category | Primary Function | Common Examples |
| Replacement ($T_{4}$) | Synthetic version of thyroxine; standard for hypothyroidism. | Levothyroxine (e.g., Thyronorm, Eltroxin). |
| Replacement ($T_{3}$) | Faster-acting active form; often used for rapid response. | Liothyronine (Cytomel). |
| Combination Therapy | A fixed ratio of synthetic $T_{3}$ and $T_{4}$. | Liotrix (Thyrolar). |
| Antithyroid Agents | Inhibits hormone synthesis for hyperthyroidism/Graves’ disease. | Methimazole, Propylthiouracil (PTU). |
| Natural Thyroid | Derived from porcine (pig) glands; contains both $T_{3}$ and $T_{4}$. | Armour Thyroid. |
2. Emerging Trends in 2026: Precision & Home Care
The thyroid market in 2026 has shifted toward patient-centric monitoring and cellular-level optimization:
Home-Based Diagnostic Kits: A significant growth area in 2026 is the surge in smartphone-connected home test kits. Patients can now measure TSH, $T_{3}$, and $T_{4}$ levels with lab-grade accuracy at home, allowing for real-time dosage adjustments via telehealth.
Mitochondria-First Approach: New clinical models in 2026 emphasize that hormone levels alone may not be enough. Research now focuses on mitochondrial health to ensure that once hormones are administered, the cells have the energy capacity to actually utilize them.
Personalized Dosing: There is a move away from “one-size-fits-all” tablets toward personalized compounding and micro-dosing to avoid the side effects of over-replacement, such as heart palpitations or bone loss.
3. Significance of Thyroid Manufacturing in India (2026)
India is a global powerhouse for thyroid care, with the domestic diagnostic and drug market growing at a CAGR of 7.8% as of 2026.
Massive Scale of Treatment: With 1 in 10 Indian adults living with hypothyroidism, local manufacturers like Abbott India and GSK produce billions of doses of Levothyroxine annually to meet both domestic and export demands.
Affordability Hub: India remains the primary source for affordable, high-quality generic thyroid hormones for the Global South, ensuring that life-long therapy remains accessible.
Advanced Testing Infrastructure: Indian diagnostic chains are leaders in adopting Immunoassay-based sensitivity improvements, reducing false negatives in thyroid screenings.
Awareness Leadership: January 2026 (Thyroid Awareness Month) saw a major push by Indian hospitals for early screening in high-risk groups, such as pregnant women and those with a family history of autoimmune disorders like Hashimoto’s.
4. Why Healthy Inc. is Your Strategic Sourcing Partner
Managing a thyroid portfolio requires a partner who understands the high sensitivity of hormone stability. Healthy Inc. acts as your technical bridge:
Strategic Sourcing Hub: We are associated with state-of-the-art units specializing in Hormonal and Endocrine therapies. Whether you need high-volume Levothyroxine tablets or specialized Antithyroid agents, we match you with the right facility.
Pharmacist-Led Technical Vetting: We provide “straight answers” on Active Pharmaceutical Ingredient (API) purity and tablet stability. Our team vets every batch to ensure the narrow therapeutic index of these hormones is strictly maintained.
Regulatory & Dossier Mastery: We simplify international registration by providing full CTD/ACTD dossiers, including stability data for Zone IVb (hot/humid climates) where thyroid hormones are prone to degradation.
Supply Chain Continuity: For chronic conditions like hypothyroidism, treatment cannot be interrupted. Through our network, we ensure a reliable, WHO-GMP certified supply chain that keeps your market consistently stocked.
Showing all 2 results
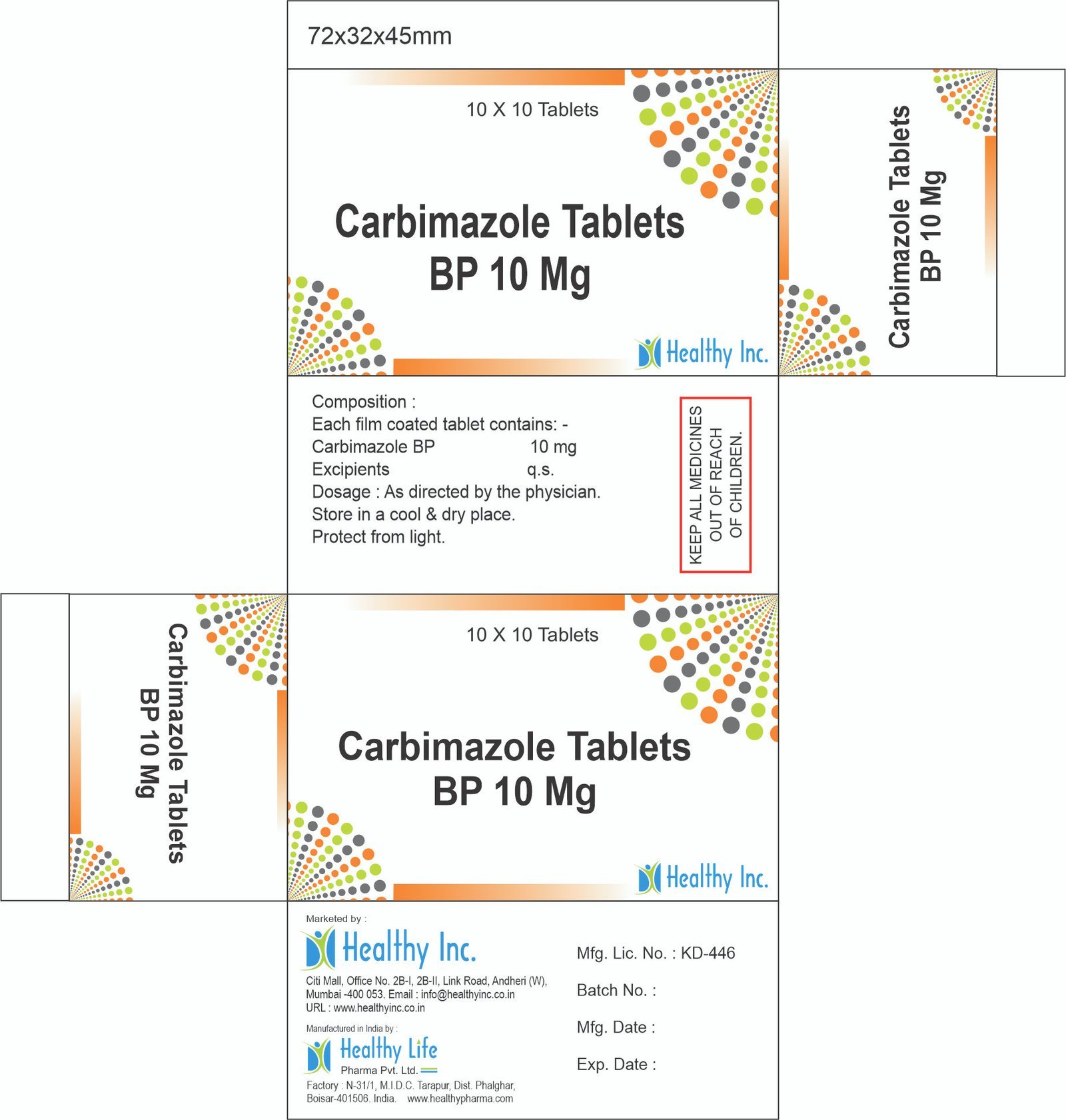
Carbimazole Tablets
FreeCarbimazole Tablets BP 10 Mg
Each film coated tablet contains: Carbimazole BP 10 mg Excipient – QS
Usage: – Treat an overactive thyroidCategory: – Thyroid Hormones drugs
Therapeutic category: – Thyroid Hormones
Levothyroxine Tablets
FreeThyrostim Tablets
Levothyroxine Sodium Tablets BP
(Thyroxine Sodium tablets BP)
Each Uncoated tablet contains :
Levothyroxine Sodium BP
Eq. To Anhydrous Levothyroxine Sodium 0.1 mgTHYROXINE SODIUM TABLETS BP (LEVOTHYROXINE SODIUM TABLETS BP 0.25 MG)
EACH UNCOATED TABLET CONTAINS:
LEVOTHYROXINE SODIUM EQ. TO ANHYDROUS LEVOTHYROXINE SODIUM BP 0.25 MG
EXCIPIENTS Q.S.
Levothyroxine Sodium Tablets IP 50 MCG Each Uncoated tablet contains:Levothyroxine Sodium IP Eq. to Anhydrous Levothyroxine Sodium IP (50 mcg)
EXCIPENT (- QS)
THYROXINE 25 MCG
EACH UNCOATED TABLET CONTAINS:
LEVOTHYROXINE SODIUM EQ TO ANHYDROUS LEVOTHYROXINE SODIUM IP 25 MCGLevothyroxine Tablets IP 25 mcg Each uncoated tablet contains:
LEVOTHYROXINE SODIUM EQ TO ANHYDROUS LEVOTHYROXINE SODIUM IP (25 mcg)
Excipient (- QS)Levothyroxine Sodium Tablets IP 50 MCG Each Uncoated tablet contains: Levothyroxine Sodium IP Eq. to Anhydrous Levothyroxine Sodium IP (50 mcg)
EXCIPENT (- QS)
Usage: – Treat hypothyroidismCategory: – Thyroid Hormones drugs
Therapeutic category: – Thyroid Hormones