Generic drugs are pharmaceutical products that are chemically identical to a brand-name drug but are typically sold at a much lower price after the original developer’s patent has expired. In 2026, generic drugs are the cornerstone of global healthcare, accounting for over $470 billion in market value and representing the vast majority of prescriptions filled worldwide.
1. What Defines a Generic Drug?
A generic drug must be bioequivalent to the brand-name (reference) drug. This means it has the same active ingredient, strength, dosage form, and route of administration.
“Same” vs. “Similar”: While the active pharmaceutical ingredient (API) is identical, generic drugs may use different inactive ingredients (excipients) like flavors, dyes, or binders.
Bioequivalence Testing: Generic manufacturers must prove that the drug releases the API into the bloodstream at the same rate and to the same extent as the original brand-name drug.
2. The 2026 Global Landscape & “Patent Cliff”
The year 2026 is a historic turning point for the generics industry due to a massive “Patent Cliff.”
Blockbuster Expiries: Between 2026 and 2028, patents for major blockbuster drugs (totaling approximately $40 billion in annual revenue) are expiring. This is fueling a surge in new generic launches in oncology, cardiology, and immunology.
Complex Generics: The industry is moving beyond simple pills. In 2026, there is a surge in Complex Generics, including drug-device combinations (like inhalers) and Generic Injectables, which require higher technical expertise to manufacture.
AI Integration: Manufacturers are now using Artificial Intelligence to model bioequivalence, which has shortened the development cycle from years to months, allowing generic versions to hit the market the day after a patent expires.
3. India: The “Pharmacy of the World” in 2026
India remains the global leader in generic production, with the domestic market reaching $30 billion in 2025 and projected to exceed $53 billion by 2034.
Manufacturing Scale: India graduates thousands of pharmaceutical scientists annually and operates over 10,000 manufacturing facilities, ensuring a massive, talent-backed supply chain.
Quality Supremacy: India holds the largest number of USFDA-approved plants outside the United States. In 2026, the focus has shifted from “low cost” to “low cost + high compliance,” ensuring that Indian generics are trusted by major health systems globally.
Backward Integration: To secure the supply chain in 2026, many Indian firms have achieved Backward Integration, producing their own raw materials (APIs) in-house through the government’s PLI (Production Linked Incentive) scheme.
4. Why Healthy Inc. is Your Strategic Generics Partner
In the competitive landscape of 2026, sourcing generics requires more than just a price list; it requires technical and regulatory reliability. Healthy Inc. serves as your professional gateway:
Strategic Sourcing Hub: We are associated with a network of specialized, WHO-GMP and USFDA compliant units. Whether you need high-volume analgesics, specialized oncology drugs, or complex injectables, we match you with the right facility.
Pharmacist-Led Technical Vetting: Led by professional pharmacists, we provide “straight answers” on stability and purity. We vet every batch to ensure the dissolution profiles and impurity limits match international standards exactly.
Regulatory & Dossier Mastery: We provide the full suite of CTD/ACTD dossiers and stability data for all climate zones, ensuring your products pass local health authority audits in any country.
Market Readiness: Through our network, we offer flexible Private Labeling (OEM), helping you build a high-quality brand backed by the world’s most robust manufacturing infrastructure.
Showing all 100 results
Calcium Carbonate with Vitamin D3 Tablets
FreeCalcium Carbonate With Vitamin D3
Each Uncoated Tablet contains :
Calcium Carbonate 1250 Eq Elemental Calcium IP (500 mg )
Cholecalciferol IP (250 IU)
Excipient (Inhouse ) (-QS)Calcium Carbonate With Vitamin D3Tablets
Each Uncoated Chewable Tablet Contains:
Calcium Carbonate IP 625 mg
Vitamin D3 IP 200 IUCalcium Gluconate Tablets IP 500 mg
Each Uncoated Tablet Contains:
Calcium Gluconate IP 500 mgCAL4U TABLETS
Calcium carbonate with Vitamin D3 Tablets
Each Film Coated Tablet Contains:
Calcium Carbonate IP 625 mg
eq. To Calcium 250 mg
Vitamin D3 125 IU
Shelf life: 1 yearSHREYCAL TABLETS
(Calcium, Minerals with Vitamin D3 Tablets )
Each Filmcoated Tablets Contains:
Calcium Citrate USP 1000 mg
Eq To Elemental Calcium 211 mg
Vitamin D3 IP 200 IU
Magnesium Hydroxide IP Eq To Elemental Magnesium 100 mg
Zinc Sulphate USP Eq To Elemental Zinc 4 mg
Colour : Titanium Dioxide IP
Shelf Life: 2 years
Dosage: As Directed by PhysicianCalcium Gluconate Tablets IP 500 mg
Each Uncoated Tablet Contains:
Calcium Gluconate IP 500 mgUsage: – Treat by low calcium levels
Category: – Generic Drugs
Therapeutic category: – Generic Drugs
Calcium Gluconate Injection
FreeCalcium gluconate injection is a sterile, supersaturated solution primarily used to treat acute symptomatic hypocalcemia. It is most commonly available as a 10% solution (100 mg/mL)
Calcium Gluconate Tablets
FreeCalcium Carbonate With Vitamin D3
Each Uncoated Tablet contains :
Calcium Carbonate 1250 Eq Elemental Calcium IP (500 mg )
Cholecalciferol IP (250 IU)
Excipient (Inhouse ) (-QS)Calcium Carbonate With Vitamin D3Tablets
Each Uncoated Chewable Tablet Contains:
Calcium Carbonate IP 625 mg
Vitamin D3 IP 200 IUCalcium Gluconate Tablets IP 500 mg
Each Uncoated Tablet Contains:
Calcium Gluconate IP 500 mgCAL4U TABLETS
Calcium carbonate with Vitamin D3 Tablets
Each Film Coated Tablet Contains:
Calcium Carbonate IP 625 mg
eq. To Calcium 250 mg
Vitamin D3 125 IU
Shelf life: 1 yearSHREYCAL TABLETS
(Calcium, Minerals with Vitamin D3 Tablets )
Each Filmcoated Tablets Contains:
Calcium Citrate USP 1000 mg
Eq To Elemental Calcium 211 mg
Vitamin D3 IP 200 IU
Magnesium Hydroxide IP Eq To Elemental Magnesium 100 mg
Zinc Sulphate USP Eq To Elemental Zinc 4 mg
Colour : Titanium Dioxide IP
Shelf Life: 2 years
Dosage: As Directed by PhysicianCalcium Gluconate Tablets IP 500 mg
Each Uncoated Tablet Contains:
Calcium Gluconate IP 500 mgUsage: – Treat by low calcium levels
Category: – Generic Drugs
Therapeutic category: – Generic Drugs
Celecoxib Capsules
FreeCelecoxib capsules are selective non-steroidal anti-inflammatory drugs (NSAIDs) used to manage pain and inflammation. They are commonly available in strengths of 50 mg, 100 mg, 200 mg, and 400 mg.
Clindamycin Injection
FreeClindamycin injection (brand names Cleocin Phosphate, Dalacin C) is a sterile aqueous solution of the antibiotic clindamycin phosphate. The exact composition of inactive ingredients depends on whether it is a single-dose or multi-dose formulation.
Diclofenac Potassium Tablets
FreeDiclofenacPotassium Tablets 50 mg
Each Sugarcoated tablet contains:
Diclofenac Potassium BP 50 mgDiclofenac Potassium and Paracetamol Tablets
Each uncoated Tablet contains: Diclofenac Potassium BP 50 mg
Paracetamol BP 650 mg
colour: Tartrazine –
Excipient – QSUsage: – Muscle aches, backaches, dental pain, menstrual cramps, and sports injuries
Category: – Anti Analgesic Painkiller Drugs
Therapeutic category: – NSAID, Arthritis, Painkiller
Digoxin Tablets
FreeDigoxin Tablets IP 0.25 mg
Each Uncoated tablet contains:
Digoxin IP 0.25 mgDIGOXIN TABLETS IP 62.5 MCG Each uncoated tabelt contiains: digoxin tablets IP 62. 5 mcg excipients q.s.
Usage: – treat heart failure
Category: – Generic drugs and Medicine
Therapeutic category: – Generic Drugs
Disulfiram Tablets
FreeDisulfiram Tablet IP 500 MG Each uncoated tablets cotains: Diulfiram IP 500 mg Excipient -QS
Usage: – Side effects and safety, interactions
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Doxepin Hydrochloride Capsules
FreeDoxepin Hydrochloride capsules are tricyclic antidepressants (TCAs) used to treat depression, anxiety, and sleep disorders (insomnia).
Dutasteride Capsules
FreeDutasteride capsules are most commonly available as 0.5 mg soft gelatin capsules. They contain the active drug dissolved in a liquid medium to ensure proper absorption
Ergometrine Tablets
FreeErgometrine Tablets IP 250 mcg
Each Sugarcoated Tablet Contains:
Ergometrine Maleate IP 250 mcgUsage: – Postpartum haemorrhage and post abortion hemorrhage in patients with uterine atony
Category: – Generic Drugs and Medicine
Therapeutic category: –
Etoricoxib Tablets
FreeEtoricoxib tablets 90 mg
Each film coated tablet contains:
Etoricoxib 90 mgUsage: – Pain relief
Category: – Anti Analgesic Painkiller Drugs
Therapeutic category: – NSAID, Analgesic, Generic Drugs
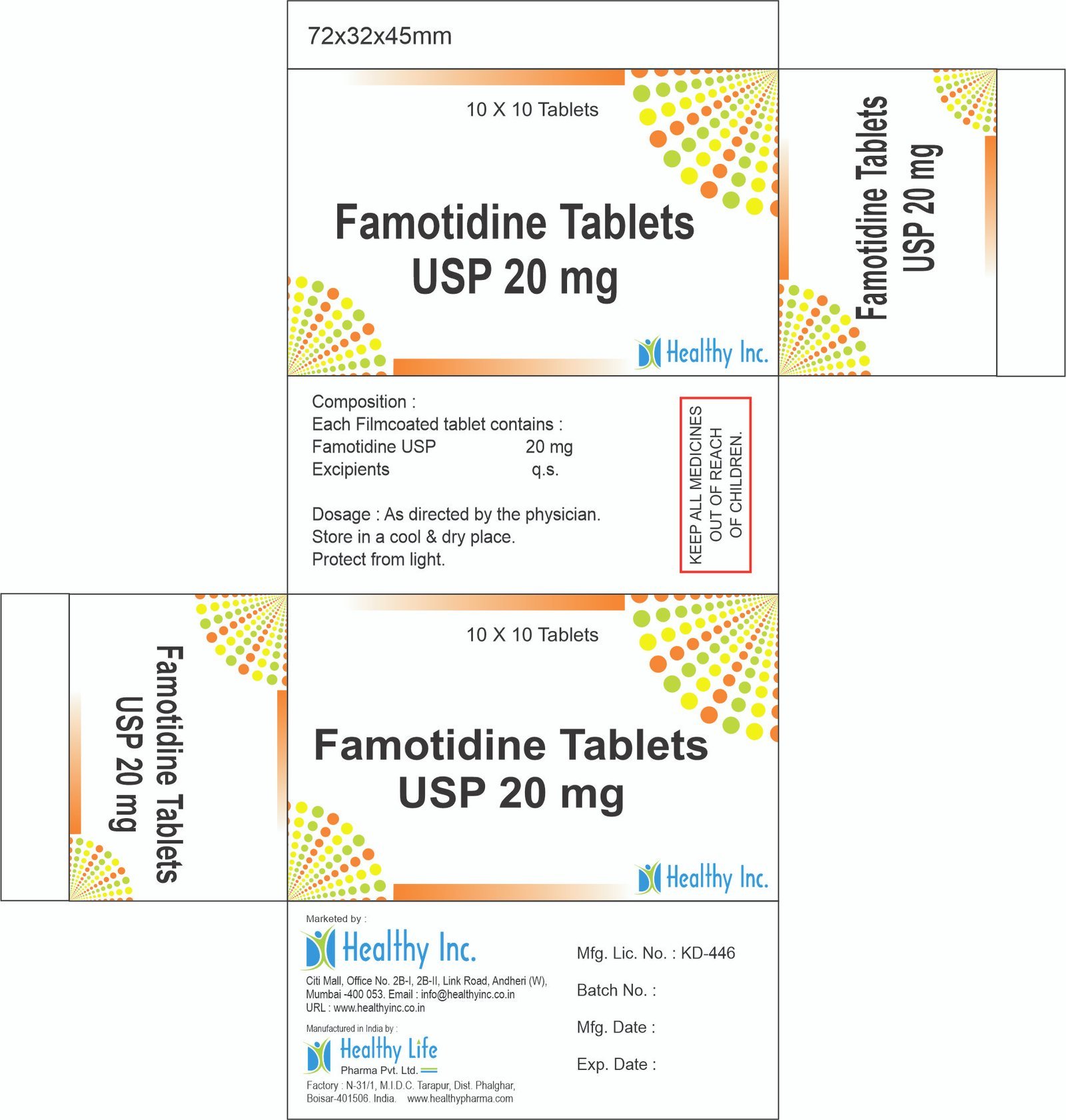
Famotidine Tablets
FreeFamotidine Tablets USP 20 mg
Each Filmcoated tablet contains :
Famotidine USP 20 mgUsage: – Prevent and treat heartburn due to acid indigestion and sour stomach
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Glucosamine Sulphate Chondroitin Sulphate Tablets
FreeGlucosamine Sulphate And Chondroitin Sulphate Tablets
Each Film Coated Tablet Contains:
Glucosamine Sulphate 500 mg
added as Glucosamine Sulphate Sodium Chloride
Chondroitin Sulphate 400 mg
added as Chondroitin Sulphate SodiumUsage: – slow or prevent the degeneration of joint cartilage
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
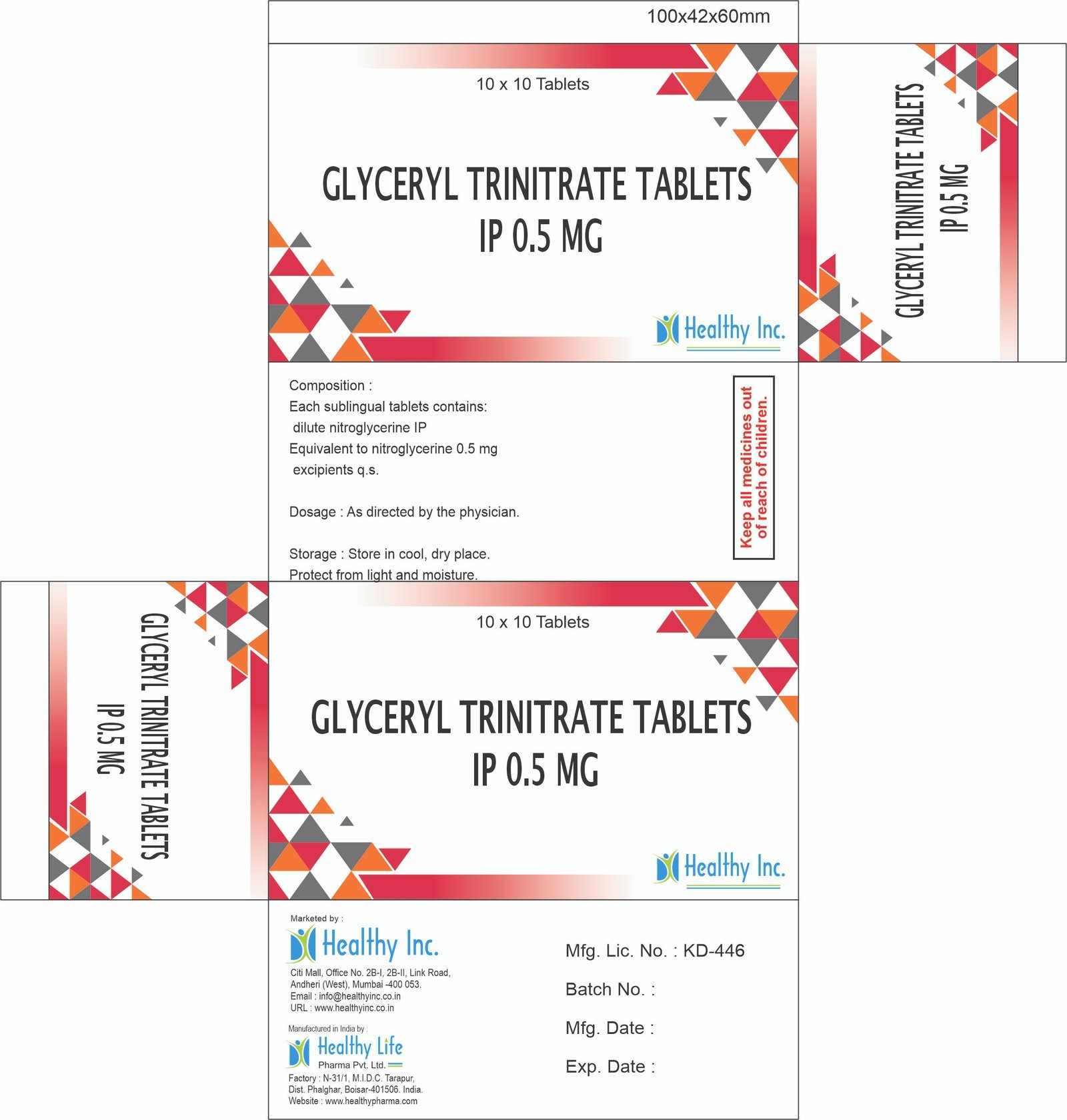
Glyceryl Trinitrate Tablets
FreeGLYCERYL TRINITRATE TABLETS IP 0.5 MG
Each sublingual tablets contains: Dilute Nitroglycerin IP Equivalent to Nitroglycerin 0.5 mg
Excipients q.s.Usage: – Treat angina (chest pain)
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic
Heparin Sodium Injection
FreeHeparin Sodium injection is a sterile, aqueous solution of a glycosaminoglycan derived from porcine (pig) intestinal mucosa, used as an anticoagulant to prevent and treat blood clots.
Hyoscine Butylbromide Injection
FreeHyoscine Butylbromide injection (brand name Buscopan) is a sterile, clear, colourless solution used as an antispasmodic to treat acute spasms of the gastrointestinal, biliary, and genito-urinary tracts.
Iron Folic Acid Tablets
FreeIRON WITH FOLIC ACID TABLETS IP
Each Enteric Coated Tablet Contains: –
Dried Ferrous Sulphate 335 mg
Eq. to Elemental iron IP (100 mg)
Folic Acid IP (0.5 mg)
Indigo Caramine (- )
Excipients (- )Iron with Folic acid Tablets IP
Each sugarcoated tablet contains:Ferrous Sulphate IP (200 mg)
Eq. to Elemental Iron IP (60 mg)
Folic Acid IP (0.5 mg)
Ponceau 4R (In House) (- )
Excipients (- QS)Usage: – Treat or prevent low iron in the body
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs / Medicine
Itopride Tablets
FreeItopride Tablets
Each Film Coated Tablet Contains:
Itopride Hydrochloride 50 mgUsage: – treatment of heartburn and nausea
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Lidocaine Cartridges
FreeA standard Lidocaine and Adrenaline (also known as Lignocaine and Epinephrine) dental cartridge contains a sterile solution designed for local anesthesia. The most common formulation is 2% Lidocaine with 1:80,000 Adrenaline
Lidocaine Injection
FreeLidocaine Hydrochloride (HCl) injection is a sterile, aqueous solution containing the active local anaesthetic drug along with specific excipients to ensure it is safe for parenteral administration.
Lignocaine Adrenaline Dental Cartridges
FreeLignocaine (Lidocaine) and Adrenaline dental cartridges are standard local anaesthetics used for oral procedures. A typical 2% Lignocaine with 1:80,000 Adrenaline cartridge contains
Magnesium Sulphate Injection
FreeMagnesium Sulphate injection is a sterile, concentrated solution used primarily to treat magnesium deficiency, manage pre-eclampsia/eclampsia, and control certain cardiac arrhythmias
Modafinil Tablets
FreeMODAFINIL TABLETS USP 100 MG EACH UNCOATED TABLET CONTAINS: MODAFINIL BP 100 MG: – MODAFINIL BP 100 mg
EXCIPIENTS QSUsage: – Treat sleepiness caused by narcolepsy, shift work sleep disorder, or obstructive sleep apnea
Category: – Generic Drugs and Medicine
Therapeutic category: – Nootropic, CNS Drugs, Generic Drugs
Multivitamins & Multiminerals Capsules
FreeMultivitamin and multimineral capsules contain a broad spectrum of active nutrients designed to fill dietary gaps, along with inactive excipients that provide structure and stability to the capsule. While specific formulations vary by brand and target audience (such as men, women, or seniors), most contain a combination of 13 essential vitamins and several key minerals.
Naloxone Injection
FreeNaloxone Hydrochloride injection is a sterile, clear solution used as an opioid antagonist to rapidly reverse respiratory depression.
Neostigmine Injection
FreeNeostigmine Methylsulfate injection is a sterile, clear solution of a cholinesterase inhibitor used primarily to reverse the effects of non-depolarising neuromuscular blocking agents after surgery or to treat myasthenia gravis.
Nitroglycerin Injection
FreeNitroglycerin injection (also known as Glyceryl Trinitrate or GTN) is a sterile solution used for the rapid treatment of acute heart failure, unstable angina, and to control blood pressure during surgery.
Nitroglycerin Tablets
FreeNitroglycerin Tablets USP
Each Sublingual Tablet Contains :
Dilute Nitroglycerin USP
Eq.to Nitroglycerin 0.5 mg
Excipients qsUsage: – Treat episodes of angina (chest pain)
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Oral Rehydration Salts (Sachets)
FreeOral Rehydration Salts IP
Each 4.2 gm contains :
Sodium Chloride IP 0.52 gm
Potassium Chloride IP 0.30 gm
Sodium Citrate IP 0.58 gm
Anhydrous Glucose IP 2.70 gm
Excipients qs
Refreshing FlavourOral Rehydration Salts IP
Each 21 gm contains :
Sodium Chloride IP 2.6 gm
Potassium Chloride IP 1.5 gm
Sodium Citrate IP 2.9 gm
Anhydrous Dextrose IP 13.5 gm
Excipients qs
Refreshing Orange FlavourOral Rehydration Salts BP
Each 4.2 gm contains :
Sodium Chloride BP 0.52 gm
Potassium Chloride BP 0.30 gm
Sodium Citrate BP 0.58 gm
Anhydrous Glucose BP 2.70 gm
Refreshing FlavourOral Rehydration Salts BP
Each 21 gm contains :
Sodium Chloride BP 2.6 gm
Potassium Chloride BP 1.5 gm
Sodium Citrate BP 2.9 gm
Anhydrous Glucose BP 13.5 gm
Refreshing Orange FlavourORASEL / ORAL REHYDRATION SALT B.P. Each 21 gm sachet contains: Anhydrous glucose BP 13.5 g Sodium Chloride BP 2.6 g Sodium citrate BP 2.9 g Potassium Chloride BP 1.5 g Refreshing orange Flavour
HEALTHY ORS /ORAL REHYDRATION SALTS BP EACH 20.5 GM CONTAINS:: – POTASSIUM CHLORIDE BP 1.5 GM
– SODIUM CITRATE BP 2.9 GM
– ANHYDROUS GLUCOSE BP 13.5 GM
– SODIUM CHLORIDE BP BP 2.6 GM– EXCIPIENTS QS
Usage: – To replace salts and water that the body loses when you have dehydration caused by gastroenteritis, diarrhea, or vomiting
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Prednisolone Tablets
FreePrednisolone Tablets IP 5 mg
Each uncoated tablet contains:
Prednisolone IP 5 mgPrednisolone Tablets IP 10 mg
Each uncoated tablet contains:
Prednisolone IP 10 mgPrednisolone Tablets IP 20 mg
Each uncoated tablet contains:
Prednisolone IP 20 mg
Excipients q.sPrednisolone Tablets IP 10 mg
Each uncoated tablet contains:
Prednisolone IP 10 mgProgesterone Capsules
FreeProgesterone capsules are hormone replacement medications typically containing natural micronized progesterone, which is chemically identical to the hormone produced by the human body. They are most commonly available in 100 mg and 200 mg strengths.Propanthaline Tablets
FreeXyproline Tablets
Propanthaline Tablets IP 15 mg
Each Coated tablet contains :
Propanthaline Bromide IP 15 mgUsage: – treat ulcers
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Sulfasalazine Tablet
FreeSalacir Tablets
Sulfasalazine Table
Each Uncoated tablet contains :
Sulfasalazine USP 500 mgUsage: – treat bowel inflammation, diarrhea
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Tacrolimus Tablets
FreeTacrolimus Tablets 1 MG film coated tablet: Anhydrous Tacrolimus 1 MG
Usage: – to prevent rejection of a kidney, heart, or liver transplant
Category: – Generic drugs & Medicine
Therapeutic category: – Generic
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Tadalafil 10mg and Dapoxetine 30 mg Tablets
FreeA tablet containing Tadalafil 10mg + Dapoxetine 30mg is a combination medication designed to treat two common male sexual health issues: erectile dysfunction (ED) and premature ejaculation (PE).
Tadalafil Tablets
FreeKwiklis -10 Tablets
Tadalafil Tablets 10 mg
Each Filmcoated tablet contains :
Tadalafil 10 mgTHADAAL / TADALAFIL TABLETS BP 20 MG
Each Film coated Tablet Contains:
Tadalafil BP 20 mg
q.s. –Usage: – treat erection problems
Category: – Generic drugs & Medicine
Therapeutic category: – Generic Drugs, Erectile Dysfunction
Temazepam tablet manufacturer supplier exporter
FreeKwiklis -10 Tablets
Temazepam tablets, 15 mg
Each Filmcoated tablet contains :
Tadalafil 10 mgTemazepam / Temazepam tablets 15 MG
Each Film coated Tablet Contains:
Temazepam 15 mg q.s. –
Usage: – treat erection problems
Category: – Generic drugs & Medicine
Therapeutic category: – Generic Drugs, Erectile Dysfunction
Thiamine tablet manufacturer supplier exporter
FreeThiamine tablets 75 mg
Each Film coated tablet contains :
Thiamine tablets 175 mgThiamine / Thiamine tablets 75
Thiamine tablets 20 mg
q.s. –
Category: – Generic drugs & Medicine
Therapeutic
category: – Generic Drugs, Erectile Dysfunction
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Ursodeoxycholic Tablet
FreeUrsodeoxycholic Acid Tablets
Each Sugar Coated Tablet Contains :
Ursodeoxycholic Acid BP 150 mgUrsodeoxycholic Acid Tablets
Each Sugar Coated Tablet Contains:
Ursodeoxycholic Acid BP 300 mgUsage: – Primary biliary cirrhosis and gallbladder stones
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Pcd marketing:-
Manufacturer: – Healthy Life Pharma Pvt Ltd
Exporter: – Healthy Inc
Supplier: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Distributor: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Seller: – Healthy Life Pharma Pvt Ltd
Healthy Inc
Zinc Dispersible Tablet
FreeZinc Acetate Dispersible Tablets 10 mg
Each Uncoated Dispersible Tablet Contains:
Zinc Acetate BP
eq. To Elemental Zinc 10 mg
Approved Colour & Flavour usedZinc Acetate Dispersible Tablets 20 mg
Each Uncoated Dispersible Tablet Contains:
Zinc Acetate BP
eq. To Elemental Zinc 20 mg
Approved Colour & Flavour usedUsage: – treat Wilson’s disease
Category: – Vitamin supplement,Generic Drugs
Therapeutic category: – Generic Drugs
Zinc Sulphate Tablet
FreeZinc Sulphate tablets 10 mg
Each Filmcoated tablet contains :
Zinc Sulphate Monohydrate BP
Eq. To elemental Zinc 10 mgZinc Sulphate Tablets IP 20 mg
Each Uncoated tablet contains:
Zinc Sulphate IP Eq. To elemental Zinc 20 mgZinc Sulphate Tablets
Each Filmcoated tablet contains :
Zinc Sulphate BP
Eq. To elemental Zinc 50 mgZINC SULPHATE TABLETS USP 20 MG
EACH UNCOATED TABLET CONTAINS:
– ZINC SULPHATE MONOHYDRATE, EQUIVALENT TO ELEMENTAL ZINC IP 20 mgZinc Sulphate Tablet USP
Each Uncoated Dispersible Tablet Contains:
Zinc Sulphate Monohydrate USP
eq. To Elemental Zinc 20 mg Excipients q.s
Approved Colour & Flavour usedZinc Sulfate Tablets IP
Each dispersible tablet contains:
Zinc Sulfate Monohydrate IP
Eq. to elemental Zinc 20 mg
Excipients q.s Approved Flavour & Colour usedZinc Sulphate Tablet USP
Each Uncoated Dispersible Tablet Contains:
Zinc Sulphate Monohydrate USP
eq. To Elemental Zinc 20 mg Excipients q.s
Approved Colour & Flavour usedUsage: – treat and to prevent zinc deficiency
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Zinc tablet with Oral Rehydration Salts Tablet
FreeZRATE KIT ZINC + ORS CMB Each kit contains: A Oral Rehydration Salts BP
Each 21 gm Contains :
Sodium Chloride BP 2.6 gm
Potassium Chloride BP 1.5 gm
Sodium Citrate BP 2.9 gm
Anhydrous Glucose BP 13.5 gm
Approved Flavour Used
Excipients q.s Refreshing orange flavour B Zinc Sulfate Tablets USP
Each dispersible tablets contains:
Zinc Sulfate Monohydrate USP
Eq. to elemental Zinc USP 20 mgORASEL KIT / ZINC + ORS KIT CMB
Each kit contiains:
Orasel / Oral rehydration salt B.P.
Each 21 gm sachet contains:
Anhydrous Glucose BP 13.5 g
Sodium Chloride BP 2.6 g
Sodium Citrate BP 2.9 g
Potassium Chloride BP 1.5 g
Refreshing orange flavour
ZINC SULPHATE TABLETS USP
Each Dispersible tablets contains:
Zinc Sulphate monohydrate Eq. to elemental Zinc USP 20 mg
Excipients Q.S.
Approved Flavour and colour used
Kit Of Zinc Sulfate Tablets USP
& Oral Rehydration Salts IP
Each Kit Contains:
A Zinc Sulfate Tablets USP
Healthy Zinc 14 Tablets
Each uncoated dispersible tablets contains:
Zinc Sulfate Monohydrate USP
Eq. to elemental Zinc 20 mg
Approved colour & flavour
B Oral Rehydration Salts IP 2 X 21 gm
Each 21 gm pack contains:
Sodium Chloride IP 2.60 gm
Potassium Chloride IP 1.50 gm
Sodium Citrate IP 2.90 gm
Anhydrous Dextrose IP 13.50 gm
Excipients q.s.
Refreshing Orange FlavourKit Of Zinc Sulfate Tablets USP & Oral Rehydration Salts IP
Each Kit Contains:
A Zinc Sulfate Tablets USP
Healthy Zinc 14 Tablets
Each uncoated dispersible tablets contains:
Zinc Sulfate Monohydrate USP
Eq. to elemental Zinc 20 mg
Approved flavour
B Oral Rehydration Salts IP
Healthy ORS 2 X 21 gm
Each 21 gm pack contains:
Sodium Chloride IP 0.52 gm
Potassium Chloride IP 0.30 gm
Sodium Citrate IP 0.58 gm
Anhydrous Dextrose IP 2.70 gm
Excipients q.s.
Refreshing Orange FlavourUsage: – treat and to prevent zinc deficiency
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs
Zolpidem Tablet
FreeZoldem
Zolpidem Tartrate tablets
Each Filmcoated tablet contains:
Zolpidem Tartrate BP 10 mgUsage: – treat insomnia
Category: – Generic Drugs and Medicine
Therapeutic category: – Generic Drugs