Cardiovascular agents are a broad class of pharmaceuticals used to treat medical conditions associated with the heart or the circulatory system (blood vessels). In the 2026 pharmaceutical landscape, these agents are the primary defense against cardiovascular diseases (CVDs), which remain the leading cause of global mortality.
These medications work by altering the rate and intensity of cardiac contractions, modifying blood vessel diameter, managing blood volume, or regulating cholesterol and clotting factors.
1. Major Classifications of Cardiovascular Agents
Cardiovascular therapy is highly specific, with different drug classes targeting different physiological mechanisms:
| Category | Primary Function | Common Examples |
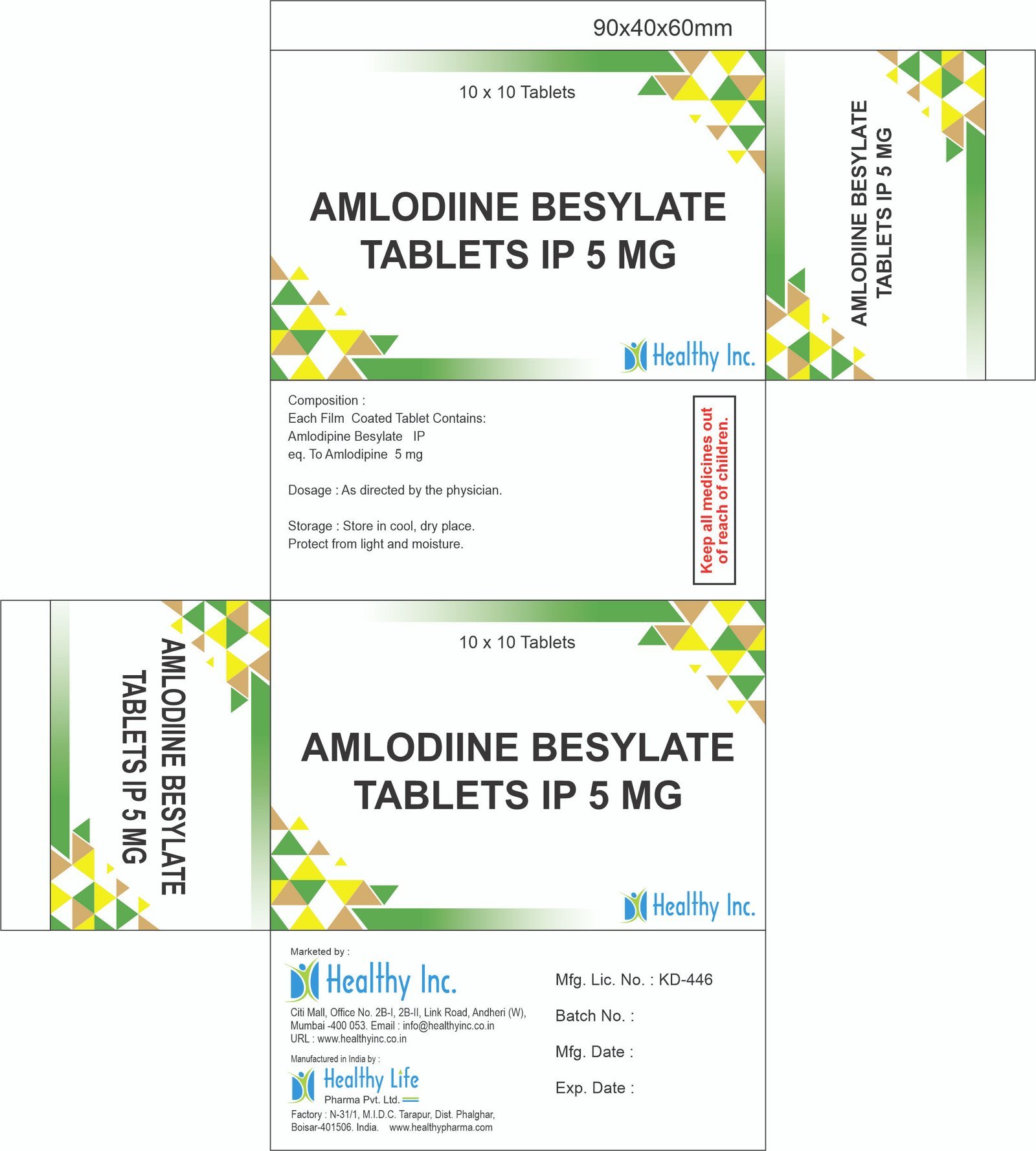
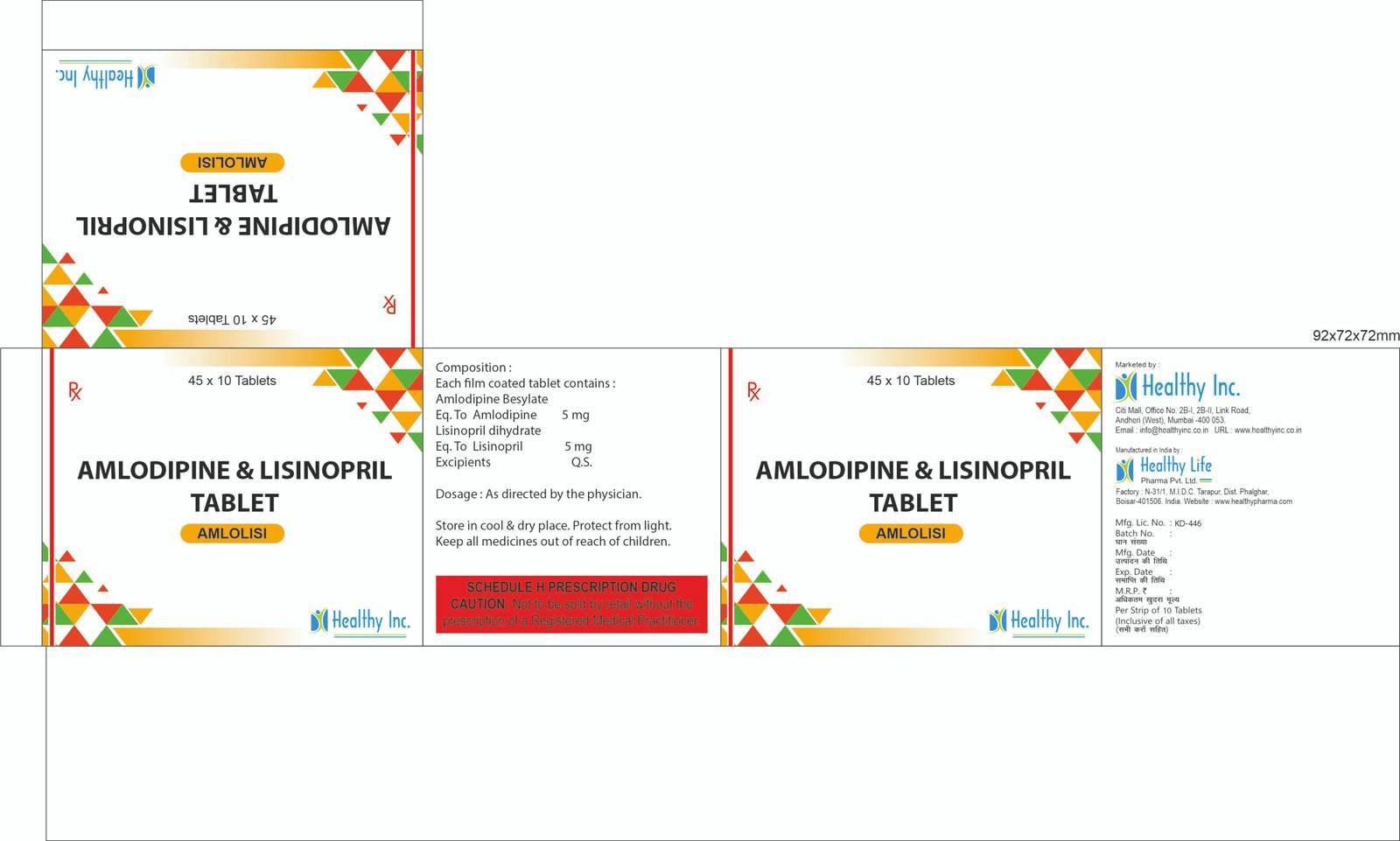
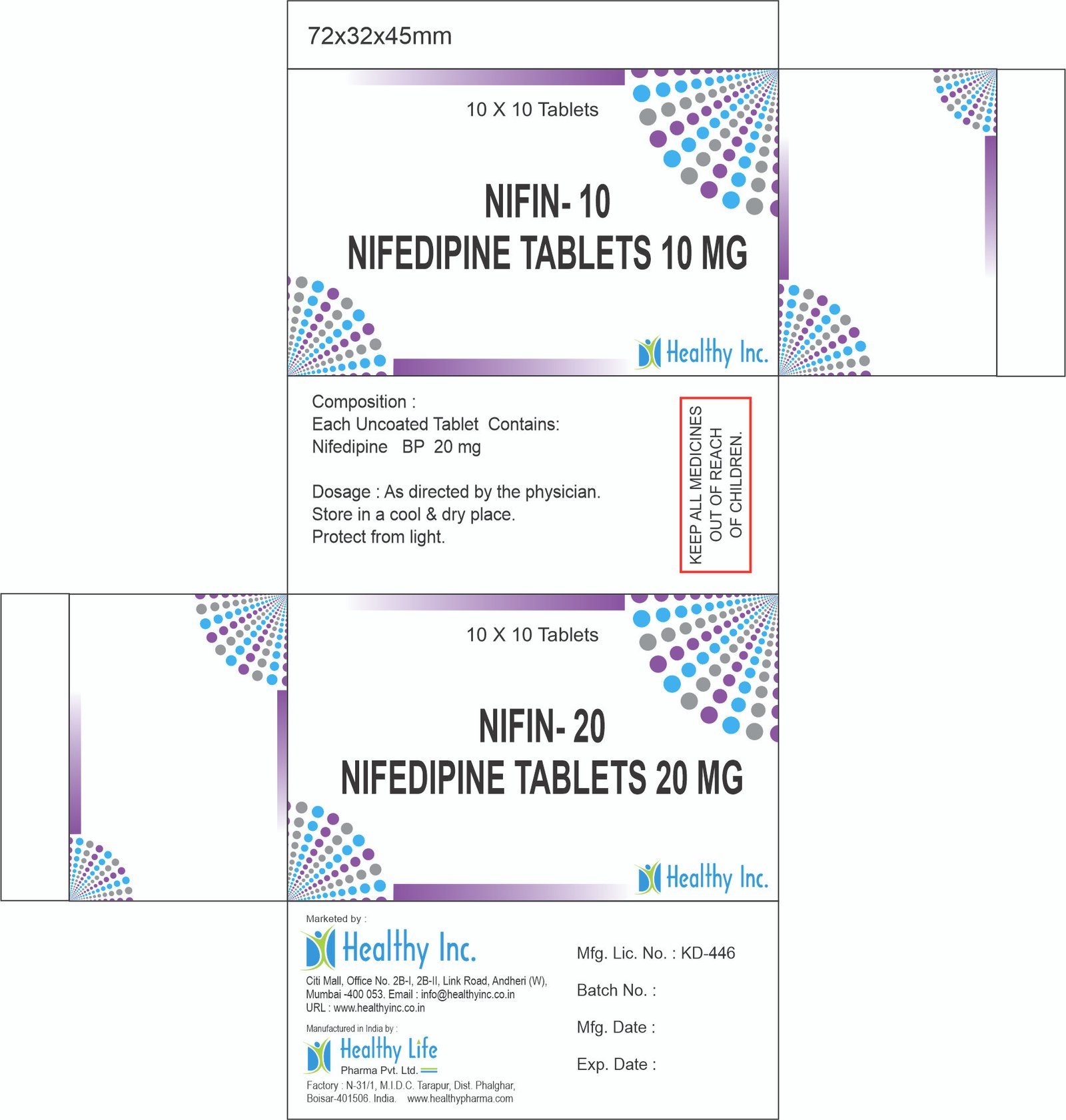
| Antihypertensives | Reduces blood pressure to prevent stroke and heart failure. | Amlodipine (CCB), Telmisartan (ARB), Enalapril (ACEI). |
| Antihyperlipidemics | Lowers “bad” LDL cholesterol and triglycerides to prevent plaque buildup. | Atorvastatin, Rosuvastatin, Ezetimibe. |
| Antithrombotics | Prevents or dissolves blood clots (includes anticoagulants and antiplatelets). | Aspirin, Clopidogrel, Warfarin, Rivaroxaban (DOAC). |
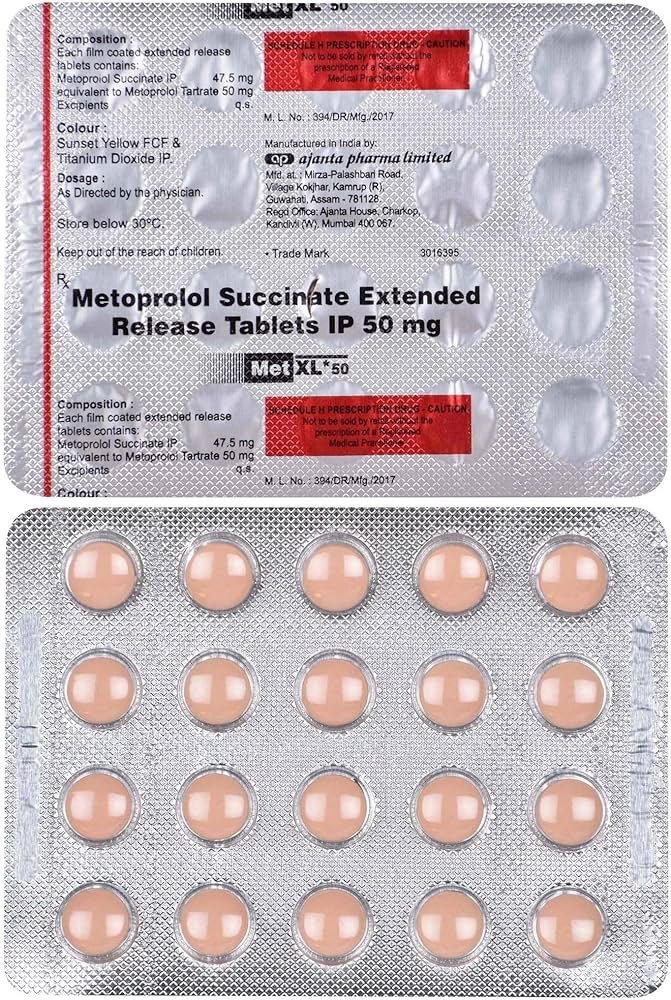
| Antiarrhythmics | Corrects irregular heartbeats (arrhythmias) by stabilizing electrical signals. | Amiodarone, Digoxin, Beta-blockers (Metoprolol). |
| Diuretics | Helps the kidneys remove excess salt and water, reducing blood volume. | Furosemide (Loop), Spironolactone (Potassium-sparing). |
2. 2026 Industry Trends & Innovations
The year 2026 marks a shift toward precision cardiology and long-acting delivery systems:
Nanomedicine & Targeted Delivery: Emerging 2026 research focuses on using nanoparticles to deliver drugs directly to atherosclerotic plaques or damaged heart tissue, minimizing systemic side effects.
Fixed-Dose Combinations (FDCs): There is a massive trend toward “Polypills”—combining an antihypertensive, a statin, and an antiplatelet into one tablet to improve patient compliance.
PCSK9 Inhibitors: While statins remain the baseline, 2026 has seen a surge in injectable PCSK9 inhibitors for patients who are resistant to traditional cholesterol therapy.
AI in Predictive Manufacturing: Manufacturers now use AI to predict demand for chronic medications, ensuring zero stock-outs for life-saving drugs like anticoagulants.
3. Significance of Cardiovascular Manufacturing in India (2026)
India is the global backbone for affordable heart care. As of 2026, the Indian cardiac drug market is projected to hit INR 50,000 crores.
Global Export Hub: India exports over $25 billion in pharmaceuticals annually, with cardiovascular generics serving as a primary pillar for markets in over 100 countries.
WHO-GMP Compliance: Leading Indian facilities in states like Maharashtra (Mumbai) and Gujarat operate high-tech, WHO-GMP certified blocks dedicated to cardiac formulation, ensuring international reliability.
The PCD Pharma Model: A unique 2026 growth driver in India is the PCD (Propaganda Cum Distribution) model, which democratizes access to cardiac care in rural areas by granting monopoly rights to local franchisees.
Advanced Formulation Tech: India leads in producing complex formats like Extended-Release (ER) tablets and sterile Cardiovascular Injections at a fraction of the cost found in Western markets.
4. Why Healthy Inc. is Your Strategic Sourcing Partner
Navigating the high-volume cardiac market requires a partner who understands the “Zero-Interruption” necessity of heart medication. Healthy Inc. acts as your technical bridge:
Strategic Sourcing Hub: We are associated with multiple state-of-the-art manufacturing units featuring dedicated Cardiac Blocks. Whether you need high-purity Statins, ACE Inhibitors, or Anti-arrhythmic Injections, we match you with the right facility.
Pharmacist-Led Technical Vetting: We provide “straight answers” on bioequivalence and stability. Our team vets every batch to ensure precise dissolution rates, which is critical for maintaining stable blood pressure and heart rhythm.
Regulatory & Dossier Mastery: We simplify international registration by providing full CTD/ACTD dossiers, stability data for all climate zones, and WHO-GMP certifications required for global tenders.
Custom Branding (OEM): Through our network, we offer flexible private labeling, allowing you to launch high-quality cardiac brands with customized packaging and professional detailing.
Showing all 31 results