Anti-anxiety medications, scientifically known as anxiolytics, are a class of pharmaceuticals designed to inhibit anxiety and manage disorders such as Generalized Anxiety Disorder (GAD), panic attacks, and social phobia. In the fast-evolving landscape of 2026, these medications have shifted from broad-spectrum sedatives to targeted, precision therapies.
1. Major Classifications of Anti-Anxiety Drugs
The pharmaceutical industry categorizes anxiolytics based on their neurotransmitter targets and duration of action:
| Category | Mechanism of Action | Common Examples |
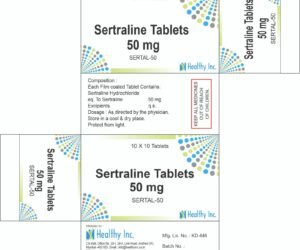
| SSRIs & SNRIs | First-line treatment; increases serotonin/norepinephrine levels. | Escitalopram, Sertraline, Venlafaxine. |
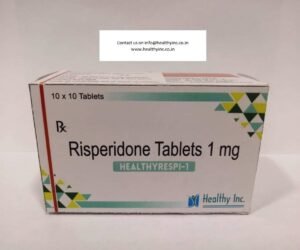
| Benzodiazepines | Fast-acting; enhances GABA (calming neurotransmitter) activity. | Diazepam, Alprazolam, Lorazepam. |
| Azapirones | Non-sedating; acts on serotonin receptors without dependence risk. | Buspirone. |
| Beta-Blockers | Manages physical symptoms (tremors, racing heart) during performance anxiety. | Propranolol. |
| Novel Anxiolytics | 2026-era neurosteroids and fast-acting nasal sprays for acute panic. | Brexanolone analogs, Intranasal GAD-modulators. |
2. Significance of Anti-Anxiety Manufacturing in India (2026)
In 2026, the global anxiety drug market has reached $22.50 billion, with India serving as a critical volume and innovation hub.
Global Supply Leadership: India manufactures approximately 20% of the world’s generic psychiatric medications, ensuring that high-quality mental health treatments remain affordable across 190+ countries.
Advanced Delivery Platforms: Indian CDMOs are currently pioneering Nanoparticle-based drug delivery. These systems allow medications to cross the blood-brain barrier (BBB) more efficiently, reducing side effects and enabling lower dosages.
Precision Manufacturing: Facilities in India have integrated AI-driven production monitoring to ensure 100% batch consistency for highly potent molecules, adhering to updated WHO-GMP and USFDA 2026 guidelines.
Strategic Shift to Biologics: Beyond traditional pills, Indian firms are exploring the production of neuro-peptides and biosimilars for complex psychiatric conditions.
3. Why Healthy Inc. is Your Strategic Sourcing Partner
Sourcing psychiatric medications requires a partner who understands the sensitivity of “Central Nervous System” (CNS) products. Healthy Inc. acts as your technical bridge:
Strategic Sourcing Hub: We are associated with multiple state-of-the-art manufacturing units specializing in CNS therapies. We match your specific needs—from Extended-Release (ER) tablets to Oral Suspensions—with the facility holding the best stability data.
Pharmacist-Led Technical Vetting: We provide “straight answers” on bioavailability and pharmacokinetics. Our team vets every batch to ensure precise dissolution rates, which is critical for maintaining steady drug plasma levels in anxiety patients.
Regulatory & Dossier Mastery: We simplify international registration by providing full CTD/ACTD dossiers, stability studies for all climate zones, and bioequivalence reports.
Innovative Product Portfolio: Through our network, we offer the latest 2026-era formulations, including fast-dissolving mouth strips and pediatric-safe oral liquids, providing your brand with a competitive edge in patient compliance.
Showing all 11 results