Antiasthmatic medications are a specialized class of pharmaceuticals designed to prevent, manage, and treat the symptoms of asthma. In 2026, the global respiratory market is estimated at over $35 Billion, with the industry shifting toward “Smart Inhalers” and targeted biologic therapies to achieve better long-term control.
1. Major Classifications of Antiasthmatics
Asthma treatment is generally divided into two main categories: “Relievers” for immediate symptoms and “Controllers” for long-term maintenance.
| Category | Purpose | Common Examples |
| Short-Acting Beta-Agonists (SABA) | Relievers: Rapidly opens airways during an attack. | Salbutamol (Albuterol), Levosalbutamol. |
| Inhaled Corticosteroids (ICS) | Controllers: Reduces long-term airway inflammation. | Budesonide, Fluticasone, Beclomethasone. |
| Leukotriene Modifiers | Oral tablets that block inflammatory chemicals. | Montelukast, Zafirlukast. |
| Long-Acting Beta-Agonists (LABA) | Maintenance therapy (usually combined with ICS). | Salmeterol, Formoterol. |
| Biologics (Monoclonal Antibodies) | Targets specific immune pathways in severe asthma. | Omalizumab, Mepolizumab, Dupilumab. |
2. Significance of Antiasthmatic Manufacturing in India (2026)
As of 2026, India is a global leader in respiratory care, particularly in the production of affordable, high-quality inhalation devices.
Inhalation Technology Mastery: Manufacturing Metered Dose Inhalers (MDIs) and Dry Powder Inhalers (DPIs) requires high-precision engineering. Indian firms like Cipla and Lupin are global benchmarks for inhalation technology.
Transition to Green Propellants: In 2026, Indian manufacturers are leading the global switch to low-GWP (Global Warming Potential) propellants in MDIs, meeting the latest international environmental standards without compromising drug delivery.
Smart Inhaler Innovation: Indian CDMOs are currently pioneering “Smart Inhalers” equipped with Bluetooth sensors to track patient adherence and inhalation technique, a major trend in 2026 digital health.
Affordable Biologics: With the 2026 surge in biosimilars, India is making expensive biologic asthma treatments accessible to emerging markets at a fraction of the cost of innovator brands.
3. Why Healthy Inc. is Your Strategic Sourcing Partner
Navigating the complex world of respiratory devices and sterile inhalants requires a partner who understands Aerosol Science. Healthy Inc. acts as your technical bridge:
Strategic Sourcing Hub: We are associated with multiple state-of-the-art manufacturing units featuring dedicated Respiratory Blocks. Whether you need HFA-Inhalers, Respirator Solutions for Nebulization, or Montelukast tablets, we match you with the right facility.
Pharmacist-Led Technical Vetting: We provide “straight answers” on Fine Particle Fraction (FPF) and dose consistency. Our team vets every batch to ensure the drug reaches the deep lungs effectively, which is critical for patient safety.
Regulatory & Dossier Mastery: We simplify international registration by providing full CTD/ACTD dossiers, stability data for various climate zones, and bioequivalence reports required for global health tenders.
End-to-End Solutions: Through our network, we offer flexible Private Labeling (OEM) for inhalers and specialized packaging to ensure your brand stands out in the competitive respiratory market.
Showing all 19 results
Chlorpheniramine Tablets
FreeChlorpheniramine Maleate Tablets IP 4 mg
Each Uncoated tablet contains :
Chlorpheniramine Maleate IP 4 mgUsage: – Uses Hay fever, and the common cold
Category: – Anti Allergic drugs
Therapeutic category: – Antiasthmatic
Etofylline Theophylline Tablets
FreeEt Phylline Retard Plus
Etofylline & Theophylline Tablets
Each Sugar Coated Sustained Release Tablet Contains:
Etofylline BP 231 mg
Theophylline BP 69 mgET Phyllin Retard
Etofylline & Theophylline Tablets
Each Sustained release sugarcoated tablet contains:
Etofylline IP 115 mg
Theophylline IP 35 mgET Phyllin
Etofylline & Theophylline Tablets
Each sugarcoated tablet contains:
Etofylline IP 77 mg
Theophylline IP 23 mgUsage: – Treatment and prevention of asthma and chronic obstructive pulmonary disease
Category: – Anti Allergic Drugs
Therapeutic category: – Antiasthmatic
Salbutamol Inhaler
FreeSalbutamol Inhaler Metered-Dose Inhaler
Product Composition & Strength
We supply this product in standardized Metered-Dose Inhaler (MDI) formats, optimized for direct pulmonary delivery.
Component Strength (per actuation) Primary Clinical Function Salbutamol (as Sulfate) IP/BP 100 mcg The Rescue Agent: Instantly opens airways. Propellant HFA-134a (CFC-Free) The Carrier: Ensures ozone-friendly delivery. Formulation Pressurized Aerosol The Standard: 200 metered doses per canister. Salbutamol Tablets
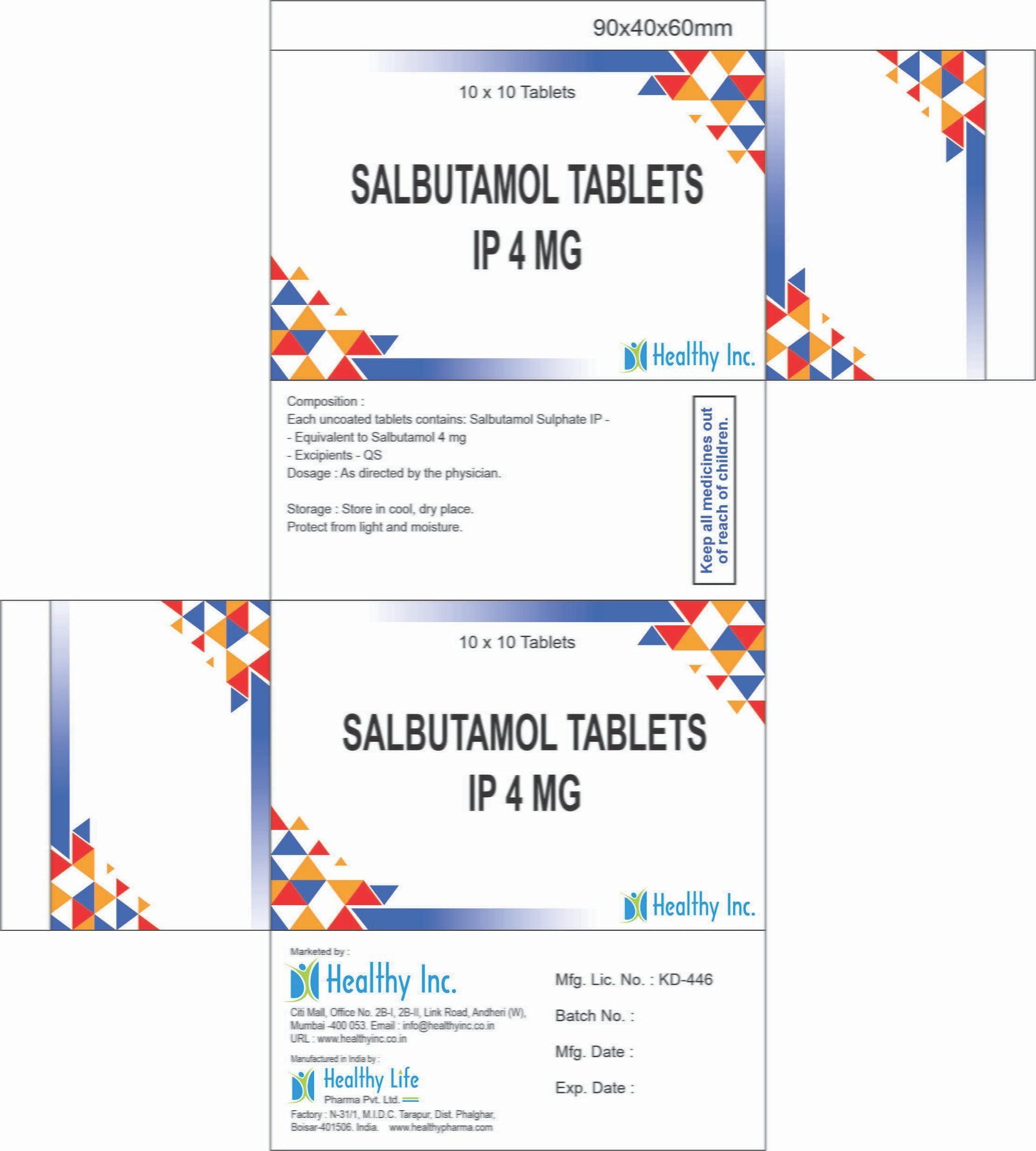
FreeSalbutamol Tablets IP 2 mg Each Uncoated Tablet contains: Salbutamol Sulphate IP – Equivalent to Salbutamol 2 mg
Excipients – QSSalbutamol Tablets IP 4 mg Each uncoated tablets contains: Salbutamol Sulphate IP –
Equivalent to Salbutamol 4 mg
Excipients – QSSalbutamol Tablets BP
Each Uncoated tablet contains:
Salbutamol Sulphate BP
Eq. to Salbutamol 4 mgUsage: – treat wheezing and shortness of breath caused by breathing problems
Category: – Antacid & Anti ulcer drugs
Therapeutic category: – Antiasthmatic
Theophyline Tablet
FreeTheophyline Tablets 200 mg
Each Uncoated tablet contains :
Theophyline IP 200 mUsage: – Prevent and treat wheezing, shortness of breath, and chest tightness caused by asthma
Category: – Anti Allergic Drugs
Therapeutic category: – Antiasthmatic, Antitussive, Bronchodilator