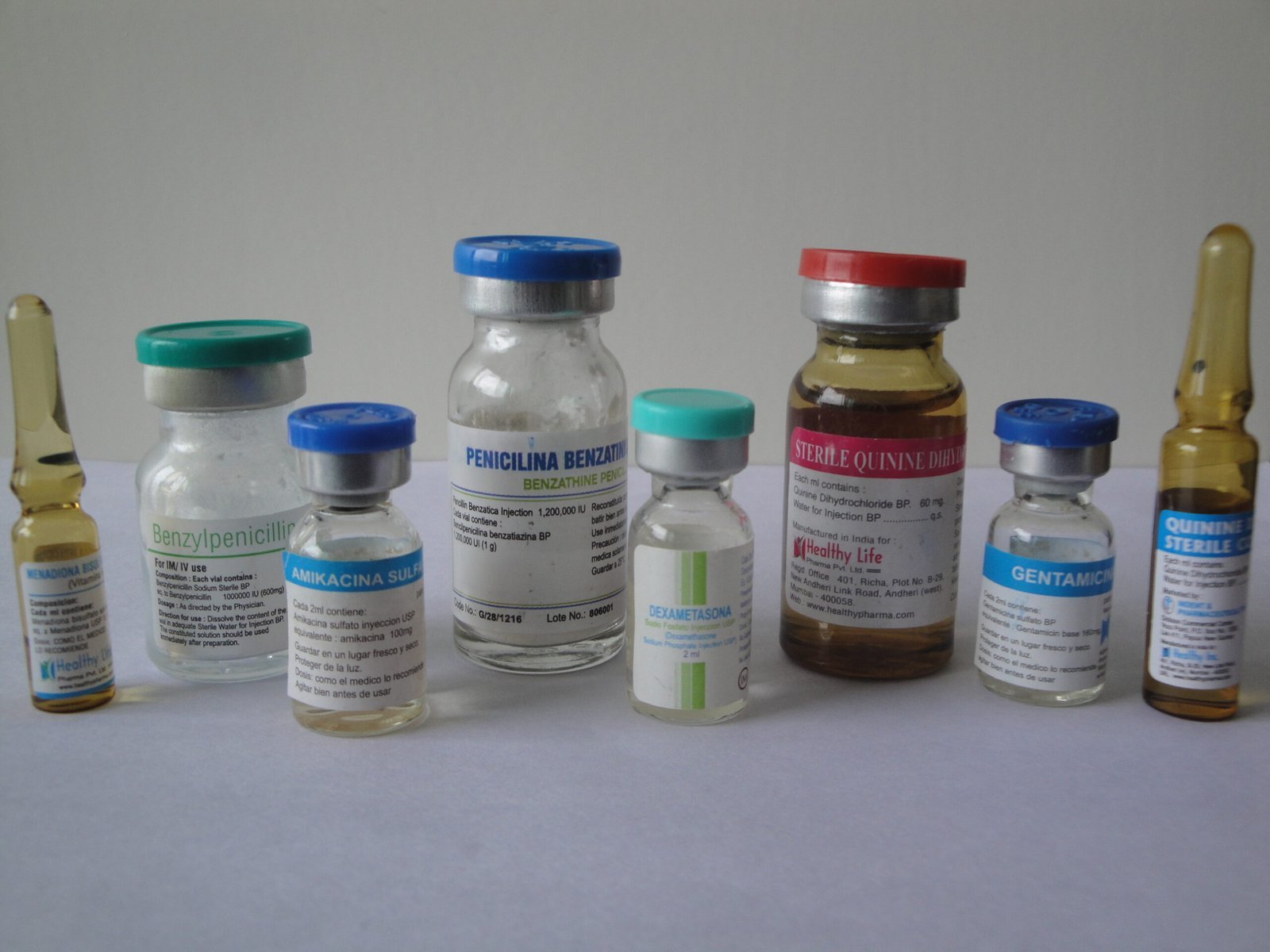
Description
Sterile Water for Injection (SWFI)
Healthy Inc is a specialized global supplier and exporter of essential pharmaceutical diluents and vehicles. We provide high-purity Sterile Water for Injection (SWFI) IP/BP/USP, sourced from WHO–GMP certified sterile facilities. This universal solvent is a critical export commodity to hospitals, pharmaceutical compounding centers, and government medical stores in Africa, LATAM, and Southeast Asia, serving as the primary vehicle for reconstituting dry powder antibiotics and diluting liquid concentrates.
Product Overview
This product consists of Water for Injection that has been sterilized and packaged in single-dose containers.
The “Universal Diluent” Specialist:
- Purity: Manufactured via advanced multi-column distillation to remove all dissolved mineral solids and organic impurities.
- Non-Pyrogenic: Strictly controlled for bacterial endotoxins to ensure no fever reaction (pyrogenic response) occurs upon injection.
- Preservative-Free: Contains no bacteriostats, antimicrobial agents, or added buffers, making it safe for use with almost all injectable drugs without chemical incompatibility.
- Versatility: The standard diluent for reconstituting Beta-lactams (Ceftriaxone, Penicillin), dissolving anesthetics, and diluting concentrated electrolytes.
Product Composition & Strength
We supply this sterile solvent in Glass Ampoules, Plastic (FFS) Ampoules, or Vials.
| Active Ingredient | Specification | Description | Role |
|---|---|---|---|
| Sterile Water for Injection | 100% v/v | Clear, Colorless, Odorless | Solvent / Vehicle |
| pH Range | 5.0 to 7.0 | — | Neutral |
| Osmolality | 0 mOsmol/kg | Hypotonic | Must add solute before IV use |
*Pack Sizes: 2ml, 5ml, 10ml, 20ml Ampoules (Glass/Plastic) or 100ml Glass Vials.
Technical & Logistics Specifications
Critical data for Pharmaceutical Importers and Distributors.
| HS Code | 3004.90.99 (Other Medicaments) |
| Dosage Form | Sterile Liquid (Solvent) |
| Packaging | Glass Ampoule (Type I) or FFS (Form-Fill-Seal) Plastic Ampoule |
| Storage | Store below 30°C. Do not freeze. |
| Certificates | WHO-GMP, COPP, Free Sale Certificate |
Manufacturing Authority
Marketed and Distributed by Healthy Inc from WHO-GMP & ISO 9001:2015 certified units.
- FFS Technology: We utilize advanced Form-Fill-Seal (FFS) technology for plastic ampoules, ensuring the container is formed, filled, and sealed in a single sterile operation without human contact.
- Conductivity Testing: Rigorous testing ensures conductivity is < 1.3 µS/cm, guaranteeing low ionic contamination.
Therapeutic Indications (Human & Veterinary Use)
Indicated purely as a pharmaceutical aid:
- Reconstitution: Dissolving sterile dry powder drugs (Antibiotics, Hormones, Lyophilized biologics) prior to administration.
- Dilution: Diluting concentrated drug solutions to the desired concentration for infusion.
Dosage & Administration
Usage Instructions (Strictly as per Physician/Pharmacist):
- Volume: The volume used depends entirely on the drug manufacturer’s instructions for the solute being dissolved.
- Inspection: Always inspect for particulate matter or leaks before use. Discard if any particles are visible.
- Single Use: Preservative-free. Any unused portion must be discarded immediately.
Safety Warnings:
- HEMOLYSIS DANGER: NEVER INJECT PLAIN SWFI INTRAVASCULARLY. Sterile Water is hypotonic (0 osmolarity). Injecting it directly into a vein without a drug/solute will cause red blood cells to burst (hemolysis), leading to kidney failure or death. It must be mixed with a drug or salt to become isotonic.
- Incompatibility: Ensure the drug being dissolved is compatible with water (some drugs require saline or dextrose).
Commercial Inquiries
For hospital tenders, bulk export, or distributor pricing, please contact our export team.
WhatsApp/Call: +91 7710003340
Email: info@healthyinc.co.in